Case Reports in Clinical Medicine
Vol.3 No.8(2014), Article
ID:48576,7
pages
DOI:10.4236/crcm.2014.38098
Epstein-Barr Virus Positive Inflammatory Pseudotumor of the Spleen: Case Report and Review of the Literature
Slim Jarboui1, Abelwaheb Hlel1, Med Ali Bekkay1, Habib BelHaadj Khalifa1, Moncef Mokni2*
1Department of general surgery of Sidi Bouzid Hospital-Tunisia, University of Medicine, Sousse, Tunisia
2Department of Anatomopathlogy, Farhat Hached Hospital, Sousse, Tunisia
Email: *drslimjarboui@yahoo.fr
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 1 June 2014; revised 30 June 2014; accepted 20 July 2014
ABSTRACT
Inflammatory Pseudotumor (IPT) forms a group of etiologically, histologically, and biologically heterogeneous tumefactive lesions that are histologically characterized by prominent inflammatory infiltrates. It has been described in various organs including the lungs, bladder, liver, spleen, heart, and others. It may mimic a malignant tumor clinically and radiologically. Splenic IPT are most frequently detected incidentally. We report a case of 38-year-old women admitted for recurrent abdominal pain. Physical examination was unremarkable. Contrast enhanced CT showed a well hypodense, circumscribed lesion, measuring 5 × 3.5 cm. A differential diagnosis of lymphoma, teratomas, angioma or angiosarcoma was considered. The patient underwent splenectomy. Histologically, the tumor consisted of an inflammatory infiltrate composed predominantly of diffusely lymphoplasmocytic cells and stromal fibroblasts. Epstein-Barr virus was detected on in situ hybridization exclusively in the epitheloid and spindles cells. The optimal management of this tumor and eventually in asymptomatic patients is still controversial. IPT of the spleen should be remembered when evaluating single or multiple nodules in spleens. The clinical and pathological features of previously reported cases are reviewed in this paper.
Keywords:Inflammatory Pseudotumor, Spleen, Splenectomy, Epstein-Barr Virus

1. Introduction
The term Inflammatory Pseudotumor (IPT) has historically been applied to heterogeneous group of mass-forming lesions characterized by a prominent inflammatory infiltrate as the predominant cellular component. They have been observed in various parts of the body including the orbit, respiratory tract, gastrointestinal tract, and soft tissues, lymph nodes and liver. IPT of the spleen is extremely rare and is frequently misdiagnosed as malignant neoplasms or other benign tumors.
In this report, we report an IPT of the spleen, and pay particular attention via a review of the literature to the diagnosis modalities, differentiated diagnosis and treatment of such tumors.
2. Case Report
A 38-year-old woman admitted to our department for a six-month history of upper left side pain of an intermediate severity. There was no history of constitutional symptom, overseas travel, and trauma or alcohol abuse. She did not have any other clinical symptom such as fever, appetites loss or weight loss. Physical examination showed no hepatomegaly, splenomegaly or lymphadenopathy. Laboratory findings were unremarkable except for slightly elevated white blood cell count (11,300/mm3) and C-reactive protein level (135 mg/ml). Abdominal Ultrasonography showed a hypoechoic splenic mass and enhanced abdominal computed tomography revealed the presence of a low density, hypovascular, centrally, well defined mass measuring 5 × 3.5 cm (Figure 1(a)). The contrast enhancement of the mass was heterogeneous with suspected necrotic areas within the tumor (Figure 1(b)). There was no evidence of lymphogenic or hamatogenic metastases.
Under diagnosis of a malignant tumor like lymphoma or atypical angiosarcoma, laparotomic splenectomy was performed. Grossly, the specimen of the spleen measured 11 × 7.5 cm in size and weighed 210 gr (Figure 2). Macroscopically, the mass was round, well circumscribed, solid medullary and measured 5 × 3 × 3 cm (Figure 3). The remainder of the spleen was compressed but otherwise grossly unremarkable. Histological findings showed that the mass was infiltrated by many lymphocytes, plasma cell with fibrous changes. No abnormal infiltrated lymphocytes were detected (Figure 4). Most of the tumor was hypercellular but punctuated in areas by zones of sclerosis and necrosis. In situ Hybridation was positive for the search of Epstein Barr Virus (EBV RNA1 and RNA2) exclusively within epitheloid and spindle cells through the tumor (Figure 5), and immunohistochemical staining was negative for follicular dendritic lesion especially CD21 and CD35, roiling out the diagnosis of IPTlike follicular dendritic cell-Tumors.
The postoperative course was uneventful and the patient fit well six months after surgery with no sign of recurrence or lymphoproliferative disorder.
3. Discussion
IPT are relatively rare lesions characterized by chronic infiltration of inflammatory cells and areas of fibrosis. They have been observed in various parts of the body including the orbit, respiratory tract, gastrointestinal tract, and soft tissues, lymph nodes and liver [1] -[7] . IPT of the spleen are extremely rare and are frequently misdiagnosed as malignant neoplasms or other benign tumors. Until 2008, less than 85 cases have been reported in the literature and this since the initial report of two cases by Cotetingam and Jaffe in 1984 [1] [3] [5] -[8] .
The clinical signs and symptoms are most divers, with pain in the left flank or hypochondrium like in our case. Pain can be associated in some cases with fever or splenomegaly, while in other cases, patients were
 (a)
(a) (b)
(b)
Figure 1. (a) Axial computed tomography without contrast injection showed a well-defined, medullary, low-density tumor over the spleen; (b) Axial computed tomography with contrast injection showed a little increase in the density of the splenic lesion respectively in arterial and portal phase.
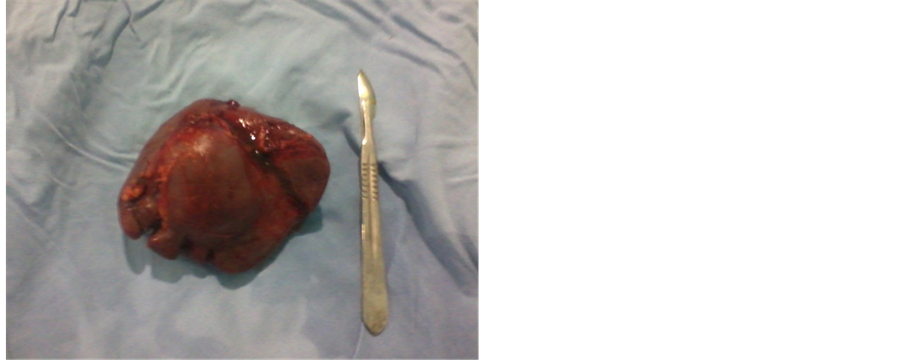
Figure 2. Grossly aspect of the spleen. The specimen measured 11 × 7.5 cm and weighed 210 gr.

Figure 3. Macroscopic view of the specimen: a well circumscribed, solid and medullary mass, measured 5 × 3 × 3 cm. the major part of the lesion showed yellowish appearance (arrow) with some stippling red spots.
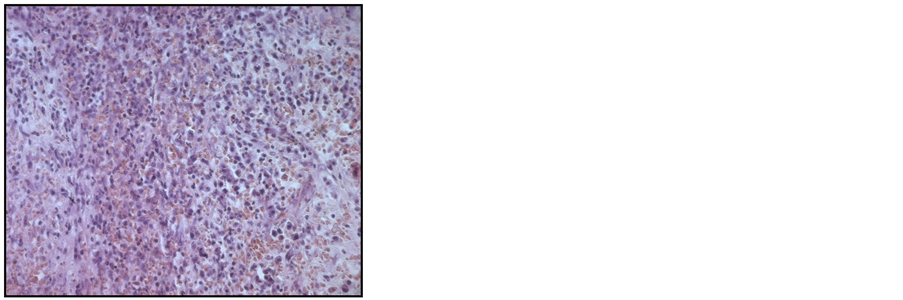
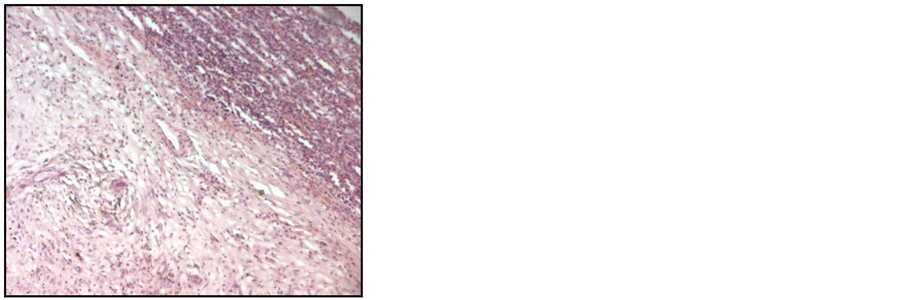
Figure 4. Histological findings for the spleen via hematoxylin and Eosin staining showing infiltration by lymphocytes and plasma cells (original magnification ×200 and 400).
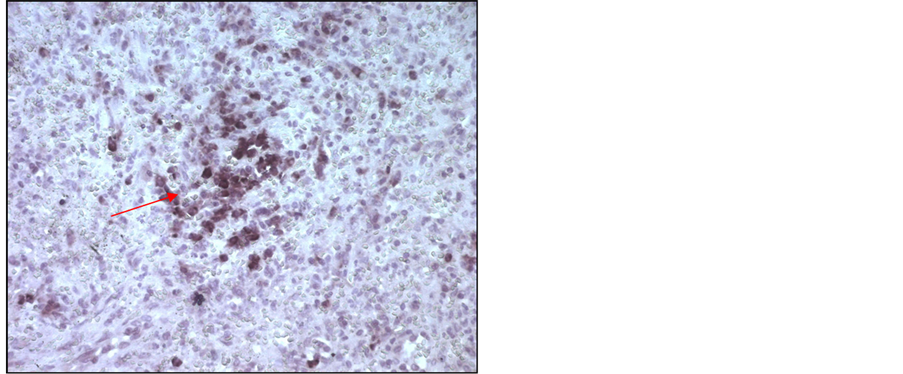
Figure 5. in situ hybridization for Epstein-Barr Virus-encoded nuclear RNA1 and RNA2 shows strong staining in the epitheloid and spindles cells (×200).
asymptomatics [1] [3] [5] [8] -[11] .
Biological findings are also no specifics and often normal. In some cases, laboratory studies showed anemia and leucocytosis due to the chronic inflammation [1] -[3] . Hypercalcemia, monoclonal peaks in the proteinogram and polyclonal hypergammglobulinemia that disappeared after splenectomy have also been reported [2] [5] .
Recent advances in imaging are helpful in the identification of space occupying lesions of the spleen but, no radiological findings have been found that are characteristics of IPT. This may be because the proportion in the distribution of inflammatory cells and fibrosis differs according to the cause and the period of inflammation [1] [4] [5] [8] . Ultrasonography shows a low echoic lesion in most cases [1] [2] [4] -[6] [9] . CT is the radiological test that most often demonstrates and this like in our patient, a low-density mass in both the non-enhanced and enhanced modes [1] [3] [5] [7] [11] -[20] . Franquet et al. [21] reported that the presence of a central satellite area corresponding to a fibrosis plaque on CT examination after contrast administration is strongly suggestive of an IPT. In several cases, magnetic resonance imaging (MRI) has been applied to spleen IPT, but the reported findings also seen insufficient to diagnose this benign entity. Typically, the lesion showed low to isointensity on T1-weighted images and high intensity with surrounding low intensity on T2 weighted images. The surrounding low-intensity area on the T2 weighted images corresponded to a focal area of fibrosis [1] [5] [9] [11] [14] [15] [16] -[20] . The dynamic MRI showed that the lesion enhanced from outside and later than benign splenic parenchyma. Fluorodeoxyglucose positron emission (FDG PET/CT) can be semiquantitatively measured using an SUV, which is generally higher in malignant tumors than in inflammatory disease. However, this method of differentiation is of limited value for IPT as the FDG uptake varies with the proportion of fibrosis and inflammatory cell infiltration [3] .
Although CT and MRI are quite sensitive, they are not specific in differentiating between splenic IPT and other malignancies. There are some reports emphasizing the importance of undertaking percutaneous fine-needle biopsy in cases of hepatic or splenic IPT to avoid unnecessary surgery [1] [2] [4] [11] [13] [20] [22] . But needle biopsy has some disadvantages such as uncertainness of detection of the disease, risk of metastases if the mass is a malignant neoplasm and potential hemorrhagic complications of the procedure. Few reports have also shown that percutaneous image guided biopsy of the spleen is both safe and effective. Kawaguchi et al reported a case of IPT involving the liver and the spleen which was confirmed following a percutaneous liver and spleen biopsy [1] [11] [13] [20] . Keogan et al. [22] performed image-guided biopsy on 20 patients; they reported less than 2% of complications rate. In addition, needle-seeding is an extremely rare complication, with an estimated rate of less than 0.01% following percutaneous abdominal fine-needle biopsy.
Splenic IPT frequently mimic the splenic neoplasms clinically and radiologically. Radiological findings as described above are not sufficiently specific to differentiate this type of lesion from other neoplasm including lymphoma, teratoma, angioma, angiosarcoma, or abscess [1] [2] [5] [6] [9] [10] [13] [14] [17] . The lack of calcification and fatty elements argues against splenic hemangioma or angiosarcoma, whereas the lack of a cystic configuration argues against lymphangioma. The most specific modality to differentiate from angiosarcoma is angiography. Angiographic findings of angiosarcoma show a hypervascular tumor with contrast pooling, and that of splenic IPT usually show vascular or hypovascular tumor [1] [5] . Splenic abscess are seen as heterogeneous hypodense mass with fluid attenuation and peripheral ring enhancement. However, primary splenic lymphoma or hamartoma remain in the differential diagnosis [1] [5] [10] [23] . A definitive diagnosis often requires Histopathological examination. In case of splenic IPT, almost all final diagnosis requires surgery since splenectomy is not only diagnostic tool but can also be curative. Laparoscopic splenectomy has been introduced in clinical setting, making it easier to perform [1] [4] [24] -[27] .
Gross features in reported cases of splenic IPT are described as a solitary well-circumscribed lesion with white or yellow cut surface that compresses the adjacent splenic parenchyma which is consistent with the findings in our case [1] -[6] [8] [9] [11] . The microscopic findings are characteristic and the diagnosis can be made by identifying the reactive nature of the cells. They show some resemblance to granulation tissue and normal lymphocytes and plasma cells are constant features, although in variable mixtures and numbers. The dominant growth pattern may be compact spindle cells, collagenous stroma, or xanthogranulomatous. The spindle cells show smooth-muscle differentiation and are generally positive for the immunohistochemical marker smoothmuscle actin. The majority of lymphocytes are T cells, with fewer numbers of B cells. The plasma cells are polyclonal, ruling out a plasma cell neoplasm [1] [4] -[6] [8] [9] . IPT should be distinguished from two other tumors: the IPT-like follicular dendrites cell-Tumor (FDC); and an inflammatory myofibroblastic tumor. These two lesions are neoplastic and therefore have a potentially worse prognosis than IPT [2] [12] .
The etiology and pathogenesis of this entity are unknown. Infections, vascular causes and autoimmune disorders have been hypothesized in their pathogenesis. Infection is one hypothesis [2] [4] [6] [9] [11] -[13] [28] -[33] . Some cases were reported to be Epstein-Barr virus (EBV)-positive-inflammatory follicular dendrites cell-Tumor (FDC). Association between EBV and IPT of the spleen and the liver has been well established [12] . In our case, no acid fast organisms, fungi, parasites, bacteria, could be identified, but EBV was detected on in situ hybridization. Like in our case, Lewis at al had reported a case of IPT of the spleen with positive clonal EBV in the epitheloid and spindle cells but negative for Follicular Dendritic cells markers. He suggests that IPT with clonal positive EBV DNA should be considered as true neoplasm rather than reactive or reparative processes. Vascular cause is another hypothesis and it is believed by some that theses lesions may be due to intraparenchymatous hemorrhage secondary to traumatism or coagulopathy. The high content of plasma cells in this lesion suggests the theory of an immunological origin as another hypothesis [2] [4] [9] [13] . Our patients did not show any signs of predisposition for auto immune disease and no acute or chronic infection was known. Furthermore, immunohistochemical examination of our specimen was negative for IgG, excluding then the diagnosis of IgG 4-related lesion. Thomas et al. had in their study of eight cases of PST, they reported an association with idiopathic thrombocytopenic purpura and they found some architectural similarities with Riedel’s thyroiditis and idiopathic retroperitoneal fibrosis, suggesting then an autoimmune pathogenesis [34] .
The prognosis of IPT has generally been considered favorable after splenectomy according to the previous published cases [2] [5] [9] [13] . However, careful follow-up is necessary, since some patients with IPT of the liver are reported probably to have died as a result of the disease.
4. Conclusion
In conclusion, we report a rare case of splenic IPT confirmed by histological examination of the spleen. It is currently a relatively rare pathology not associated with specific clinical findings. No imaging techniques allow preoperative diagnosis. The place and indication of percutaneous biopsy are debated. Most often, only splenectomy and histopathological study of the specimen allow diagnosis and treatment of this entity. A subset of these tumors harbors the EBV in epitheloid and spindle cells, and they must be distinguished from other IPT like tumors such IPT like FDC tumor and inflammatory myofibroblastic tumor because these two lesions are neoplastic and can show a worse prognosis.
References
- Yarmohammadi, H., Nakamoto, D., Faulhaber, P.F., Mieldler, J. and Azar, N. (2001) Inflammatory Pseudotumor of the Spleen: Review of Clinical Presentation and Diagnostic Methods. Journal of Radiology Case Reports, 5, 16-22.
- Takamoto, K., Midorikawa, Y., Minagawa, M. and Makuuchi, M. (2007) Inflammatory Pseudotumor of the Spleen: Clinical Impact in Surgical Treatment. Bioscience Trends, 1, 113-116.
- Cotelingham, J.D. and Jaffe, E.S. (1984) Inflammatory Pseudotumor of the Spleen. American Journal of Surgical Pathology, 8, 375-380. http://dx.doi.org/10.1097/00000478-198405000-00006
- Kawaguchi, T., Mochizuki, K., Kizu, T., Miyazaki, M., Yakushijin, T., Tsutsui, S., Morii, E. and Takehara, T. (2012) Inflammatory Pseudotumor of the Liver and Spleen Diagnosed by Percutaneous Needle Biopsy. World Journal of Gastroenterology, 18, 90-95.
- Hsu, C., Lin, C., Yang, T. and Chang, H. (2008) Splenic Inflammatory Pseudotumor Mimicking Angiosarcoma. World Journal of Gastroenterology, 14, 6421-6424. http://dx.doi.org/10.3748/wjg.14.6421
- McMahon, R.F.T. (1988) Inflammatory Pseudotumor of the Spleen. Journal of Clinical Pathology, 41, 734-736. http://dx.doi.org/10.1136/jcp.41.7.734
- Erkan, N., Yildirim, M., Yilmaz, C. and Yagei, A. (2004) Inflammatory Pseudotumor as an Unusual Cause of Colonic Obstruction: A Case Report. Acta Chirurgica Belgica, 104, 462-464.
- Celebi, A., Kayacan, S.M., Keskin, S., Temeloglu, E., Koç, D., Ercan, V., Esin, D., Ekizoglu, I., Vatansever, S., Gulluoglu, M.G. and Erk, O. (2006) A Case of Inflammatory Pseudotumor in the Spleen. Southern Medical Journal, 99, 774-776. http://dx.doi.org/10.1097/01.smj.0000217486.13651.f6
- Noguchi, H., Kondo, H., Kondo, M., Shiraiwa, M. and Monobe, Y. (2000) Inflammatory Pseudotumor of the Spleen: A Case Report. Japanese Journal of Clinical Oncology, 30, 196-203. http://dx.doi.org/10.1093/jjco/hyd048
- Safran, D., Welch, J. and Rezuke, W. (1991) Inflammatory Pseudotumor of the Spleen. Archives of Surgery, 126, 904-908. http://dx.doi.org/10.1001/archsurg.1991.01410310114018
- Yan, J., Peng, C., Yang, W., Wu, C., Ding, J., Shi, T. and Li, H. (2008) Inflammatory Pseudotumor of the Spleen: Report of 2 Cases and Literature Review. Archive of Canadian Journal of Surgery, 51, 75-76.
- Lewis, J.T., Gaffney, R.L., Casey, M.B., Farrell, M.A., Morice, W.G. and Macon, W.R. (2003) Inflammatory Pseudotumor of the Spleen Associated with a Clonal Epstein-Barr Virus Genome. Case Report and Review of the Literature. American Journal of Clinical Pathology, 120, 56-61. http://dx.doi.org/10.1309/BUWNMG5RV4D09YYH
- Wells, I.T.P. and Dodds, N.I. (2010) An Unusual Perisplenic Mass. The British Journal of Radiology, 83, 268-270.http://dx.doi.org/10.1259/bjr/22454963
- Kaza, R.K, Azar, S., Al-Hawary, M.M. and Francis, I.R. (2010) Primary and Secondary Neoplasms of the Spleen. Cancer Imaging, 10, 173-182.
- Okura, N., Mori, K., Moriscita, Y., Oda, T., Tanoi, T. and Minami, M. (2012) Inflammatory Pseudotumor of the Intrapancreatic Accessory Spleen: Computed Tomography and Magnetic Resonance Imaging Findings. Japanese Journal of Radiology, 30, 171-175. http://dx.doi.org/10.1007/s11604-011-0021-z
- Pfeifer, L., Agaimy, A., Janka, R., Boxberger, F., Wein, A., Neurath, M.F. and Siebler, J. (2011) Complete Long-Term Remission of an Inflammatory Pseudotumor under Corticosteroid Therapy. Case Reports in Oncology, 4, 304-310.http://dx.doi.org/10.1159/000329415
- Bhatt, S., Simon, R. and Dogra, V.S. (2008) Radiologic-Pathologic Conferences of the University of Rochester School of Medicine: Inflammatory Pseudotumor of the Spleen. American Journal of Roentgenology, 191, 1147-1479.http://dx.doi.org/10.2214/AJR.08.1011
- Hamdi, I., Marzouk, I., Toulali, L., Amara, H., Bellara, I., Ridene, I., Abdelkader, A.B., Khila, M. and Kraeim, C.H. (2006) Inflammatory Pseudotumor of the Spleen and Radiopathologic Correlation. Journal de Radiologie, 87, 1894-1896. http://dx.doi.org/10.1016/S0221-0363(06)74172-5
- Ma, P.C., Hsieh, S.C., Chien, J.C., Lao, W.T. and Chan, W.P. (2007) Inflammatory Pseudotumor of the Spleen: CT and MRI Findings. International Surgery, 92, 119-122.
- Handa, U., Tiwari, A., Singhal, N., Mohan, H. and Kaur, R. (2011) Utility of Ultrasound-Guided Fine-Needle Aspiration in Splenic Lesions. Diagnostic Cytopatholog, 41, 1038-1042.
- Franquet, T., Montes, M., Aizcorbe, J., Barberena, J., De Aazua, Y.R. and Cobo, F. (1989) Inflammatory Pseudotumor of the Speen: Ultrasound and Computed Tomography Findings. Gastrointestinal Radiology, 14, 181-183.http://dx.doi.org/10.1007/BF01889191
- Keogan, M.T., Freed, K.S., Paulson, E.K., Nelson, R.C. and Dodd, L.G. (1999) Imaging-Guided Percutaneous Biopsy of Focal Splenic Lesions: Update on Safety and Effectiveness. American Journal of Roentgenology, 172, 933-937.http://dx.doi.org/10.2214/ajr.172.4.10587123
- Krishnan, J. and Frizzera, G. (2003) Two Splenic Lesions in Need of Clarification: Hamartoma and Inflammatory Pseudotumor. Seminars in Diagnostic Pathology, 20, 94-104. http://dx.doi.org/10.1016/S0740-2570(03)00014-5
- Makrin, V., Avital, S., White, I., Sagie, B. and Szuld, A. (2008) Laparoscopic Splenectomy for Solitary Splenic Tumors. Surgical Endoscopy, 22, 2009-2012. http://dx.doi.org/10.1007/s00464-008-0024-8
- Shapiro, A.J. and Adams, E.D. (2006) Inflammatory Pseudotumor of the Spleen Managed Laparoscopically. Can Preoperative Imaging Establish the Diagnosis? Case Report and Literature Review. Surgical Laparoscopy, Endoscopy & Percutaneous Techniques, 16, 357-361. http://dx.doi.org/10.1097/01.sle.0000213738.77654.02
- Ushida, H., Ohta, M., Shibata, K., Endo, Y., Iwaki, K., Tominaga, M., Ishio, T. and Kitano, S. (2006) Laparoscopic Splenectomy in Patients with Inflammatory Pseudotuomor of the Spleen: Report of 2 Cases and Review of the Literature. Surgical Laparoscopy, Endoscopy & Percutaneous Techniques, 16, 182-186. http://dx.doi.org/10.1097/00129689-200606000-00015
- Suzumura, K., Okada, T., Satake, M. and Fujimito, J. (2011) Laparoscopic Splenectomy for Inflammatory Pseudotumor of the Spleen. Hepatogastroenterology, 58, 1909-1911.
- Lee, S.H., Fang, Y.C., Luo, J.P. and Chen, H.C. (2003) Inflammatory Pseudotumor Associated with Chronic Persistent Eikenella Corrodens Infection: A Case Report and Brief Review. Journal of Clinical Pathology, 56, 868-870.
- Arber, D.A., Weiss, L.M. and Chang, K.L. (1998) Detection of Epstein-Barr Virus in the Inflammatory Pseudotumor. Seminars in Diagnostic Pathology, 15, 155-160.
- Akatsu, T., Kameyama, K., Tanab, M., Endo, T. and Kitajlma, M. (2007) Epstein-Barr Virus-Positive Inflammatory Pseudotumor of the Spleen with Concomitant Rectal Cancer: A Case Report and Review of the Literature. Digestive Diseases and Sciences, 52, 2806-2812. http://dx.doi.org/10.1007/s10620-007-9773-9
- Horiguchi, H., Matsui-Horiguchi, M., Sakata, H., Ichinose, M., Yamamoto, T., Fujiwara, M. and Ohse, H. (2004) Inflammatory Pseudotumor-Like Follicular Dendritic Cell Tumor of the Spleen. Pathology International, 54, 124-131.http://dx.doi.org/10.1111/j.1440-1827.2004.01589.x
- Zhang, M.Q., Lennerz, J.K., Dehner, L.P., Brunt, L.M. and Wang, H.L. (2009) Granulomatous Inflammatory Pseudotumor of the Spleen: Association with Epstein-Barr Virus. Applied Immunohistochemistry & Molecular Morphology, 17, 259-263. http://dx.doi.org/10.1097/PAI.0b013e318189f10f
- Cheuk, W., Chan, J.K., Shek, T.W., Chang, J.H., Tsou, M.H., Yuen, N.W., Ng, W.F., Chan, A.C. and Prat, J. (2001) Inflammatory Pseudotumor-Like Follicular Dendritic Cell Tumor: A Distinctive Low-Grade Malignant Intra-Abdominal Neoplasm with Consistent Epstein-Barr Virus Association. American Journal of Surgical Pathology, 25, 721-731.http://dx.doi.org/10.1097/00000478-200106000-00003
- Thomas, R.M., Jaffe, E.S., Zarate-Osorno, A. and Medeiros, L.J. (1993) Inflammatory Pseudotumor of the Spleen. A Clinicopathologic and Immunophenotypic Study of Eight Cases. Archives of Pathology Laboratory Medicine, 117, 921-926.
NOTES

*Corresponding author.

