Journal of Agricultural Chemistry and Environment
Vol.03 No.01(2014), Article ID:43361,7 pages
10.4236/jacen.2014.31001
Development of controlled release formulations of thiabendazole
Yasser Z. El-Nahhal
Department of Earth and Environmental Science, Faculty of Science, The Islamic University-Gaza, Gaza, Palestine; y_el_nahhal@hotmail.com
Copyright © 2014 Yasser Z. El-Nahhal. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accor- dance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Yasser Z. El-Nahhal. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
ABSTRACT
This study aimed to develop controlled release formulation of Thiabendazole for reduced conta- minations to soil water and agricultural pro- duces. The idea behind this work was based on changing the ionization status of Thiabendazole molecule throughout pH changes to become a cation then adsorbing it to bentonite clay sur- faces. Equilibrium concentrations of Thiaben- dazole in the adsorption solutions were deter- mined by HPLC. Results showed that Thiaben- dazole was better adsorbed in clay at the lowest pH value (pH 3). Furthermore, raising the tem- perature of the adsorption reaction increased the adsorbed amount of Thiabendazole. Release experiments showed that Thiabendazole is ex- tremely slower at pH 3 than at pH 5.5 or pH 9. These promising results suggest that applica- tion of Thiabendazole clay based formulation may reduce the applied rate and that the relea- sed fraction consequently may reduce the envi- ronmental contamination. It is also advantage- ous to use these formulations in the acidic soil for plant disease control.
Keywords
Thiabendazole; adsorption; Controlled Release; pH; Bentonite
1. INTRODUCTION
Fungicides are essential agricultural materials needed to protect the agricultural products and to improve its quality. However, the frequent applications of fungicides to control plant diseases may have resulted in accumu- lation of their residues in plant tissues. Previous review
[1] showed that residues of pesticides have been detected in many agricultural products in the Meddle East coun- tries. For instance fenitrothion residues were detected in potato sample from Egypt [2], dicofol residues were de- tected in cucumber in Jordan [3], captan, thiabendazole , vinclozolin fenuron residues were detected in may agri- cultural products in Kuwait [4], fenvalerate residues were detected in apple samples in Pakistan [5], attabron triadiminol residues were detected in tomato and potato samples in Palestine [6], captafol residues were detected in a wheat monoculture after prolonged application in Germany [7], fludioxonil residues were found in citrus fruits in Italy [8]. However, fungicide residues have been detected in runoff from golf courses, under Scandinavian conditions [9], tricyclazole residues were found in rice paddy watersheds in Italy [10]. Carbendazim residues were detected in surface water samples in Chile [11]. Thiabendazole residues have been detected in citrus sam- ples [12], in fruit-based soft drinks [13], in apple juice concentrate [14], in lemons [15], in water samples [16], in sea port water and in sediment samples [17], in food- stuffs [18], in the UK mushroom industry [19]. Further- more, Thiabendazole application creates eco-toxicity, for instance, Castillo et al. [20] found a macroinvertebrate community changes following thiabendazole applica- tions in a banana plantation. Kim et al. [21] studied the Sorption of benzimidazole anthelmintics to dissolved organic matter and sewage sludge. They found that the correlations between logKoc and logKow were weak and the magnitude of the sorption coefficients to the hydro- philic organic matters (hydroxypropyl-beta-cyclodextrin and sewage sludge) were similar to or slightly smaller than those for the hydrophobic organic matters (humic acids and liposome).
García Calzón et al. [22] studied the adsorption of Thiabendazole at a Mercury/Solution Interface. They found a state of normal adsorption and a condensed two- dimensional layer at moderately charged and rather ne- gatively charged surfaces. Spanoghe et al. [23] studied the fate of Thiabendazole during storage, handling and forcing of chicory. They concluded that Thiabendazole residues applied before storage are still present on the roots at the start of the forcing cycle. Furthermore, pre- vious study [24] indicated that Thiabendazole appears to have some mobility in sand, silt loam, and sandy loam, but shows no mobility in clay. As obvious, residues of Thiabendazole have been found in many agricultural products and environmental samples. These residues may have adverse human health effects. Consequently, ini- tiative steps must be taken to reduce the accumulation of these residues in fruit, vegetable and environmental sam- ples and consequently reduce the health hazards. One approach to achieve this is to adsorb Thiabendazole mo- lecules to clay minerals under different pH values that change the ionization status of Thiabendazole from hy- drophobic and/or anionic form to cationic one conse- quently it can strongly be adsorbed to clay surface via cation exchange reaction.
This study is initiated to develop clay based formula- tions that controlled the release of Thiabendazole to soil environment consequently reduce the possible accumula- tion in plant tissues and to investigate the effect of dif- ferent parameters on the fate of Thiabendazole.
2. Materials and methods
Materials. We used a Ca2+-bentonite from Greece (Milos, Prassa Carys, mined 1992, M48). The reason behind using a Ca2+-bentonite clay because it contains about 95% Ca2+-montmorillonite which have high cation exchange capacity (CEC) ranged 80 - 120 mmol/g clay [25]. Thiabendazole (HPLC grade, 2-(thiazole-4-yl)ben- zimidazole; 2-(1,3-thiazol-4-yl) benzimidazole, molecu- lar weight 201.3, (Figure 1) was purchased from Fluka (Germany). It is hydrophobic with an octanol/water par- titioning coefficient KowlogP = 2.39 (pH 7), and solubili- ty in water 160 mg/L (pH 4), and 30 mg/L (pH 7 - 10). It has a pKa value 4.73 [26].
2.1. Adsorption Experiments
Influence of time on the adsorption of Thiabendazole: The stock solution of Thiabendazole was prepared by dissolving 31.9 mg in 2 - 3 mL methanol and diluting to 1 L with deionized water. The low concentration of me- thanol in the adsorption experiments had no influence on linuron adsorption.
Appropriate amounts of Thiabendazole equivalent to 30 mg/g clay were transferred to a 30 ml centrifugation tubes and diluted with water up to 25 ml. The tubes were kept under rotary shaking for 4, 6, 24 and 30 hr. The tubes were centrifuged at 20,000 g to separate the clay par-
Figure 1.Chemical structure of Thia- bendazole.
ticles. The equilibrium concentrations of Thiabendazole in supernatants were determined by HPLC as described below.
2.2. Adsorption Isotherm
The adsorption of Thiabendazole on the bentonite was measured at 22˚C ± 2˚C. Appropriate aliquots of the aqueous stock solution of Thiabendazole were diluted with water to 25 mL and added to 5 mg bentonite in 30-mL centrifuge tubes. The concentration of Thiaben- dazole ranged between 1.2 and 31.9 mg/L. The final concentration of bentonite was 0.2 g/L. The dispersions were kept under continuous rotary agitation during 48 hours. The supernatant was separated by centrifugation at 20,000 g for 0.5 h.
The concentration of Thiabendazole in the superna- tants was determined by Waters 717 HPLC with UV de- tector (detection wavelength 299 nm). Column: Nova- Pak C18 (inner diameter 3.9 mm, length 150 mm), flow rate: 1 mL/min. The mobile phase was methanol/water 50/50 (v/v). The water phase contained 0.5% ammonium hydroxide (25%). The run time was 6 min and retention time is ~3.825 ± 0.124. The injection rate was 25 µl.
The relationship between concentrations of Thiaben- dazole and peak areas of the HPLC chromatogram was linear for 0.388 - 6.38 mg/L Thiabendazole. Linear re- gression was used to determine the equilibrium concen- tration of Thiabendazole in the solutions. The regression showed R2 value close to unity (0.9997). The amount of Thiabendazole adsorbed (correctly the specific reduced surface excess nσ(n)/m) was calculated from the depletion of the Thiabendazole concentration by adsorption [27].
For each isotherm a reference solution with an inter- mediate concentration was stirred without bentonite to evaluate adsorption on the glass or other losses. Analysis showed that Thiabendazole was not adsorbed on the glass of the centrifuge tubes. All adsorption experiments were made in duplicate.
2.3. Influence of ph Values on the Adsorption of Thiabendazole
Adsorption of Thiabendazole on bentonite was meas- ured at various pH values (3, 5.5 and 9). In this proce- dure appropriate aliquots of an aqueous solution of Thiabendazole (31 mg/l) was dissolved in 1 L distilled water as mentioned above. Solutions of pH 3 and pH 5.5 were obtained by addition of few drops of acetic acid to distilled water in two separate jars whereas solution of pH 9 was obtained by addition of few drops of ammo- nium hydroxide to distilled water in a separate jar. These solutions were used for adsorption and release experi- ments.
In the adsorption experiments, appropriate amounts of Thiabendazole were diluted in 25 ml with distilled water having the pH values mentioned above. The concentra- tions range of Thiabendazole in all adsorption experi- ments under the mentioned pH values were 0.0 - 31.9 mg/L. Each adsorption tube in all experiments contained 5 mg bentonite. The adsorption solutions were main- tained at pH 3, 5.5 and 9. The final concentration of the clay was 0.2 g/L. The supernatants were separated by centrifugation at 20,000 g for 1 h, and Thiabendazole in the equilibrium solutions were determined by HPLC.
2.4. Influence of Temperature on the Adsorption of Thiabendazole
To measure the effect of temperature on the adsorption of Thiabendazole, the adsorption experiments mentioned above were maintained at 278, 291 and 313 K at pH 3. The remaining concentrations of Thiabendazole were de- termined by HPLC as mentioned below, in each tube was determined by HPLC as described above.
2.5. Preparation of Clay Based Thiabendazole Formulations
Appropriate amounts of Thiabendazole (31 mg) were dissolved in 1 liter of distilled water to form a stock so- lution. Appropriate amounts of Thiabendazole 13 mg/g clay complex was added to a 1 L volumetric flask con- taining 1 g of clay and various pH solution (3 - 9). The samples were kept for 48 h under continuous magnetic stirring. The clay-Thiabendazole complexes were sepa- rated by centrifugation at 6000 g, air dried and kept in plastic bottles at room temperature for use. The adsorbed amounts of Thiabendazole on the clay were determined by HPLC as mentioned above.
2.6. Release of Thiabendazole form Clay Based Formulation
Release of Thiabendazole from different formulations was conducted using Buchnner funnel. In this procedure, 50 g of air dried sandy soil was mixed through a glass cylinder with 20 mg bentonite-Thiabendazole formula- tion containing 1 mg Thiabendazole (Figure 1). The soil mixture was transferred to a Buchnner funnel (10 cm internal diameter) has a filter paper in the bottom. The soil layer was homogenized to a 0.5 - 1 cm height. The
soil layer in each funnel was carefully irrigated with 500 ml water having different pH values as mentioned above. Each volume of water (500 ml) was applied in ten por- tions during 2.5 hours. Each washing (50 ml) was col- lected after 30 min of application in a separate flask and analyzed by HPLC to determine the released fraction of Thiabendazole. The experiment was conducted with two replications to each pH value.
2.7. Data Analysis
Adsorption data were collected as an average of 3 rep- licate and the standard deviation was calculated and used as error bars to discriminate differences among isotherms. Presenting the standard deviation as an error bar is the best method to determine significant differences among adsorption isotherms. Variances among treatments were analyzed by calculating p-values associated with the t-test. Values < 0.05 indicated significant difference. It is well known that overlapping error bars indicate no dif- ference, whereas small or extremely small error bars in- dicate a significant difference. Adsorption data of each isotherm were fitted by the Freundlich equation to calcu- late the adsorption parameters (K, and n).
3. Results and Discussion
3.1. Adsorption Kinetics
The adsorbed amounts of Thiabendazole (Figure 1) by raw bentonite at different agitation time are shown in Figure 2. It can be seen that the adsorbed amounts are 18.33 ± 0.63, 17.82 ± 1.17, 19.09 ± 0.43, and 19.94 ± 0.2 mg/g, after 4 hr, 6 hr, 24 hr and 30 hr respectively. How- ever, the average adsorbed amount of all cases is 18.8 ± 0.92 mg/g. Statistical analysis showed no significant dif- ferences among the adsorbed amount at different time.
Furthermore, it is obvious that the standard deviation is very low indicating homogeneous values of average adsorbed amounts. Accordingly, it can be suggested that 4 hr to 6 hr is enough agitation time for the adsorption experiments. Similar results were previously obtained for other cases [28].
3.2. Influence of ph on Thiabendazole Adsorption
Adsorption of Thiabendazole on raw bentonite at dif- ferent pH values is shown in Figure 3. It can be seen that the highest adsorbed amount of Thiabendazole was ob- tained at the lowest pH value (pH 3) and the adsorbed amount decreased as the pH value increased. The lowest adsorbed amount was obtained at the highest pH value (pH 9). The explanation of these results is that pH value affects the ionization status of Thiabendazole. For

Figure 2. Adsorption kinetics of Thiabendazole on M48 at 40˚C. Bars represent standard deviation.

Figure 3. Adsorption of thiabendazole on M48 at different pH values. Bars represent standard deviation.
instance, it can be suggested that at pH 3 or pH 5.5 large fractions of Thiabendazole molecules became positively charged molecules consequently large adsorbed amount were obtained. At high pH value (pH 9) large fraction of Thiabendazole molecules became negatively charged or non charged molecules. In this case raw bentonite par- ticles can not adsorb a negatively charged Thiabenda- zole molecule due to repulsion between the negatively charged bentonite surfaces and Thiabendazole. Further- more, the hydrophilic bentonite surfaces can not adsorb large fraction from Thiabendazole molecules due to the hydrophobocity of Thiabendazole at high pH value. The effects of pH values on the ionization status of Thiaben- dazole molecules are shown in Figure 4. Our suggestion is supported by the previous results of El-Nahhal and Safi [29], who provided acceptable ionization status of bromoxynile adsorption on organo-clays and by Rodri- guez et al [30] who achieved Thiabendazole separation using a buffer of formic acid-ammonium formate at pH 3.5 with 2% of methanol using solid phase extraction.
In addition, further support to our discussion can also be obtained from previous reports [24,31] who provided different KowlogP values for Thiabendazole at different pH values.
3.3. Adsorption Isotherms at Different pH
Figure 5 illustrates the adsorption isotherms of Thia- bendazole on bentonite clay different pH values. As ex- pected, Thiabendazole was poorly adsorbed on raw ben- tonite (M48) high pH (pH 9), whereas low pH (pH 5.5 and pH 3) significantly enhanced the adsorbed amounts of Thiabendazole. It can be seen that at low equilibrium concentration (less than 0.5 mg/l) a sharp increase in the adsorbed amounts of Thiabendazole was observed. In- creasing Thiabendazole concentration in the equilibrium solution did not produce further increase in the adsorbed amount of Thiabendazole, and the adsorption reached to plateau. This feature of adsorption was obtained in the organic cation and clay minerals. Moreover, reaching a plateau suggests that the available adsorption site in bentonite surfaces were nearly occupied by Thiabenda- zole molecules. Similar feature was obtained with ad- sorption of cationic herbicides, paraquat and diquat, ad- sorption on clay [32] and organic cations to clays [25]. The explanation of pH effect on the adsorption of Thia- bendazole is given above.
Figure 3 shows the adsorption isotherms of Thiaben- dazole on bentonite at various pH values.
Thiabendazole has an acid dissociation constant (pKa) of 4.73 and is mostly neutral at pH < 4.73, becoming deprotonated (anionic) at pH > 7.
Accordingly, the adsorbed amount of Thiabendazole obtained at pH 3 is higher than the adsorbed amount

Figure 4. Influence of pH value on the ionization status of Thiabendazole.
Figure 5. Adsorption of Thiabendazole on clay (M48) at various values of pH at 291 K.
obtained at pH 5.5 and pH 9 (Figure 3). Statistical ana- lysis showed a significant difference, p-value with t-test = 0.004.
The high adsorbed amount at pH 3 was because, at pH 3, Thiabendazole molecules are mostly protonated, and have 2 positive charges in the N-atoms, in this case they may be adsorbed as cations on hydrophilic sites of the bentonite surfaces. At pH 5.5, a part of Thiabendazole molecules has a one positive charge in the N-atoms and the other part is in the neutral form (Figure 4). In this case, the one positive charged molecules are adsorbed on the hydrophilic sites of bentonite surfaces, and the neu- tral forms stay in solution; accordingly, low adsorbed amounts of Thiabendazole molecules were observed. At pH 9 the majority of Thiabendazole molecules are nega- tively charged or in equilibrium with the neutral form. In this case the negatively charged molecules stay in the solution due to the electronic repulsion between the anio- nic form of Thiabendazole and the negatively charged bentonite surfaces. Moreover, very little of the neutral Thiabendazole molecules are adsorbed the negatively charged bentonite surfaces through water bridge. The low adsorption in this case is due to the hydrophilic sur- face of bentonite particles and the hydrophobic proper- ties of Thiabendazole molecules. This explanation is sup- ported by previous reports of [29] that found a high ad- sorbed amount of bromoxynil in soil and wheat char at low pH. Furthermore, the shape of the isotherm tends to be L-type and adsorption occurs as the Longmuir type at various pH values, due to changes in the electronic form of Thiabendazole a result of pH changes.
Furthermore, the shape of the isotherms was similar in all cases. According to Gile et al. [33] classification, iso- therms of Thiabendazole could be classified as L type based on the initial slope of the curve in the concentra- tion range studied. Adsorption isotherms were regular, positive and concave to the concentration axis (Figures 5 and 6), which indicated no strong competition for the adsorption sites between the solvent (water) and Thia- bendazole molecules on bentonite surfaces. This suggests that Thiabendazole adsorption becomes easier as its con- centration in the aqueous phase rises. These isotherms indicate the tendency of large adsorbed molecules to as- sociate rather than to remain as isolated units. Fitting the data in Figures 5 and 6 to the Freundlich equation gives the adsorption parameter, (Q = KC1/n) where Q is the adsorbed amount in mg/g, C is the equilibrium concen- tration of Thiabendazole, K reflects the binding coeffi- cient, and n is the physical adsorption parameter.
It can be seen in Table 1 that the K value of adsorption on raw bentonite at pH 3 is several times higher than at pH 5.5 and pH 9, indication the high affinity of Thia- bendazole to bentonite surfaces at low pH. Furthermore, the binding coefficient (K) is the highest at 291 K. This
Figure 6. Influence of temperature on the adsorption of Thia- bendazole on M48. Bars represent standard deviation.
Table 1. Physical parameters of Thiabendazole adsorption at different conditions.
suggests that the optimum adsorption occurred at 291 K. Furthermore, the regression values (R2) in all cases are close to 1 indication of a strong positive association.
3.4. Adsorption Isotherms at Different Temperature
Adsorption of Thiabendazole at different temperature degrees are shown in Figure 6. It can be seen that ad- sorption of Thiabendazole is more pronounced at 291 than at 278 K and at 313 K. The explanation of these results is that at a low temperature (278 K), the chemical potential of Thiabendazole molecules is reduced and the molecules tend to from crystals due to their low solubili- ty in water, 0.16 g/l, 20˚C [26]. At 291 K, nearly the op- timum temperature of solubility in water, Thiabenda- zole molecules are very active and interact with the ad- sorption site at the bentonite surfaces accordingly high adsorbed amount of Thiabendazole was observed. In contrast, at high temperature (323 K), the system absorbs heat energy and an increase in the chemical potential of Thiabendazole molecules may occur. This step makes more Thiabendazole molecules in a dynamic mobility in the aqueous solution; accordingly, low adsorption was observed, indicating low adsorption affinity at high tem- perature. Statistical analysis of the data in Figure 6 indi- cates significant difference among the adsorbed amount at 278, 291 and 323 K, as the error bars which represent the standard deviation do not overlap at any point of the equilibrium concentration (Figure 6). Furthermore cal- culation of p-values among the three isotherms showed value ranges between 0.034 - 0.029, indicating signifi- cant differences among all cases.
This supports the suggestion that optimal adsorption occurred at temperature 291 K.
Changes in molar free energy (DGo) of the adsorption reaction of Thiabendazole at different temperatures were calculated using the following equation [21]:
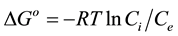
where, DGo is the molar free energy change (Kcal mol−1), R is the gas constant (1.986 cal K−1 mol), T is the abso- lute temperature of the adsorption reaction, and Ci/Ce are the initial and equilibrium concentrations, respectively.
The values of DGo for adsorption at 278 K, 291 K and 313 K from distilled water were −0.802 ± (0.313), −2.147 ± (0.504) and −0.369 ± (0.18), respectively. These results are in accordance with the data in Figure 6. These values are within the expected range for physical adsorption [34]. Analysis of variance between the calcu- lated free energy at 278 K and 291, and between 291 and 313 gave p-values of 0.0005 and 8.86E−06 respectively.
3.5. Release of Thiabendazole
The release of Thiabendazole from bentonite-Thiaben- dazole complexes at different pH values are shown in Figure 7. It is evident that % released fraction of Thia- bendazole at pH 3, pH 5.5 and pH 9 are: 1.48 ± 0.05, 25.71 ± 4.46 and 79.88 ± 9.8 respectively. The explana- tion of these results is that adsorption of Thiabendazole at low pH values (3, 5.5) tends to be attraction between the positive charge of Thiabendazole (Figure 4) and the negative charge of the Ca-bentonite clay. This interac- tions is very strong, accordingly the release of Thiaben- dazole molecules from the clay surfaces depends on the pH of added water and the possibility of Thiabendazole molecules to form hydrogen bonding with water mole- cules.
Under low pH values (pH 3, 5.5), the electrostatic in- teractions between Thabendazole molecules and water is stronger than the hydrogen bonding with water mole- cules accordingly low fraction of Thabendazole is availa- ble for solubility and release at water has low pH value. Under high pH value (pH 9), the electronic status of Tha- bendazole changed from positively charged molecules to a neutral or negatively charged ones. Accordingly the possibility of hydrogen bonding between Thabendazole and water molecules is very high beside the fact that the electrostatic interaction between Thabendazole and Ca- bentonite clay became very week. Accordingly, high
fraction of Thabendazole is available for release as evident in Figure 7.
Kinetics of Thiabendazole release at different water volumes are shown in Figure 8. It can be seen that upon application of 0.05 L water (first washing) about 80% of the initial Thiabendazole concentration was released from raw bentonite-Thiabendazole complex at pH 9, whereas 26% and 1.5% Thiabendazole concentrations were released at pH 5.5 and pH 3. Further application of eluting water up to 500 ml resulted in the further release of Thiabendazole up to 97%, 70% and 2.6% from raw bentonite-Thiabendazole at pH 9, pH 5.5 and pH 3 re- spectively. Thus, the release of Thiabendazole from ben- tonite-Thiabendazole complex at pH 3 was significantly slower than at pH 5.5 and pH 9.
The explanation of these results is that the release of Thiabendazole from bentonite-Thiabendazole complexes is controlled by the binding coefficients, (K) values, which are very high at pH 3 and pH 5.5 and low at pH 9 (Table 1). These results are in agreement with a previous report [28], which found that herbicide release was con- trolled by binding coefficients. These results are in ac- cordance with the adsorption data (Figure 5).
Figure 7. Influence of solution pH on the release fraction of Thiabendazole from bentonite (M48). Bars represent standard deviation.
Figure 8. Kinetics of Thiabendazole release from bentonite (M48) at different solutions pH. Bars represent standard deviation.
4. Concluding remarks
This study reveals that changing the solution pH of Thiabendazole resulted in changing the ionization status of the molecules and consequently enabled them to be adsorbed on the hydrophilic bentonite surfaces. The ra- tionale for this work is that adsorption of Thiabendazole on bentonite surfaces can be enhanced by changing solu- tion pH. These findings showed that the highest adsorbed amounts were obtained at pH 3 and at 291 K (Figures 3 and 6). The release of Thiabendazole was very slow from bentonite complexes at lower pH (Figures 7 and 8). The environmental relevance of this work is that bentonite clay can be used to develop environmentally acceptable Thiabendazole formulations for safe controlled release formulation.
ACKNOWLEDGEMENTS
Dr Y. El-Nahhal acknowledges Alexander von Humboldt Stiftung Foundation Fellowship Grant no IV-PAL/1104842 STP, Germany. Special thanks go to Prof Dr G.lagaly at Kiel University.
REFERENCES
[1] El-Nahhal, Y. (2004) Contamination and safety status of plant food in Arab countries. Journal of Applied Science, 4, 411-417. http://dx.doi.org/10.3923/jas.2004.411.417
[2] Dogheim, S.M., Gad Alla, S.A., El-Syes, S.M.A., Almaz, M.M. and Salama, E.Y. (1996) Organochlorine and orga- nophosphorus pesticide residues in food from Egyptian local markets. Journal of the Association of Official Ana- lytical Chemists, 79, 949-952.
[3] Nazer, I.K. and Masoud, H.A. (1986) Residues of diclofol on cucumber grown under plastic covers in Jordan. Jour- nal of Environmental Science and Health Part B, 21, 387- 399. http://dx.doi.org/10.1080/03601238609372532
[4] Sawaya, W.N., Al-Awadhi, F.A., Saeed, T., Al-Omair, A., Ahmad, N., Husain, A., Khalafawi, S., Al-Omirah, H., Dashti, B., Al-Amiri, H. and Al-Saqer J. (1999) Kuwait’s total diet study: Dietary intake of organochlorine, carba- mate, benzimidazole and phenylurea pesticide residues. Journal of the Association of Official Analytical Chem- ists, 82, 1458-1465.
[5] Tahir, S., Anwer, T., Ahmad, I., Aziz, S., Mohammad, A. and Ahad, K. (2001) Determination of pesticide residues in fruits and vegetables in Islamabad market. Journal of Environmental Biology, 22, 71-74.
[6] Safi, J., Abou Foul, N., El-Nahhal, Y. and El-Sebae, A. (2001) Monitoring of pesticide residues on green pepper, potatoes, Vicia faba, green bean and green peas in Gaza Governarate (PNA), Palestine. Journal of Pest Control and Environmental Science, 9, 55-72.
[7] Garcia-G, J.E., Kirchhoff, J. and Grossmann F. (1990) Behavior of captafol residues after prolonged application in a wheat monoculture. Journal of Environmental Sci- ence and Health Part B, 2, 185-204. http://dx.doi.org/10.1080/03601238609372532
[8] Schirra, M., D’Aquino, S., Palma, A., Marceddu, S., An- gioni, A., Cabras, P., Scherm, B. and Migheli, Q. (2005) Residue level, persistence, and storage performance of ci- trus fruit treated with fludioxonil. Journal of Agriculture and Food Chemistry, 53, 6718-6724. http://dx.doi.org/10.1021/jf051004w
[9] Stromqvist, J. and Jarvis, N. (2005) Sorption, degradation and leaching of the fungicide iprodione in a golf green under Scandinavian conditions: measurements, modelling and risk assessment. Pest Management Science, 61, 1168- 1178. http://dx.doi.org/10.1002/ps.1101
[10] Padovani, L., Capri, E., Padovani, C., Puglisi, E. and Trevisan, M. (2006) Monitoring tricyclazole residues in rice paddy watersheds. Chemosphere, 62, 303-314. http://dx.doi.org/10.1016/j.chemosphere.2005.05.025
[11] Palma, G., Sanchez, A., Olave, Y., Encina, F., Palma, R. and Barra, R. (2004) Pesticide levels in surface waters in an agricultural-forestry basin in Southern Chile. Chemos- phere, 57, 763-770. http://dx.doi.org/10.1016/j.chemosphere.2004.08.047
[12] Turiel, E. and Martín, E. (2012) A Molecularly imprinted stir bars for selective extraction of thiabendazole in citrus samples. Journal of Separation Science, 35, 2962-2969. http://dx.doi.org/10.1002/jssc.201200554
[13] García-Reyes, J.F., Gilbert-López, B., Molina-Díaz, A. and Fernández-Alba, A.R. (2008) Determination of pesti- cide residues in fruit-based soft drinks. Analytical Chemi- stry, 80, 8966-8974. http://dx.doi.org/10.1021/ac8012708
[14] He, Q., Kong, X., Zhao, J., Li, J., Yue, A. and Zhang, Y. (2008) Determination of benomyl, carbendazim and thia- bendazole in apple juice concentrate using solid-phase extraction coupled with ion exchange chromatography. Chinese Journal of Chromatography, 26, 563-567.
[15] Prousalis, K.P., Polygenis, D.A., Syrokou, A., Lamari, F.N. and Tsegenidis, T. (2004) Determination of carben- dazim, thiabendazole, and o-phenylphenol residues in le- mons by HPLC following sample clean-up by ion-pair- ing. Analytical and Bioanalytical Chemistry, 379, 458- 463. http://dx.doi.org/10.1007/s00216-004-2614-1
[16] Piccirilli, G.N. and Escandar, G.M. (2009) Flow injection analysis with on-line nylon powder extraction for room- temperature phosphorescence determination of thiaben- dazole. Analytica Chimica Acta, 646, 90-96. http://dx.doi.org/10.1016/j.aca.2009.05.033
[17] Harino, H., Mori, Y., Yamaguchi, Y., Shibata, K. and Senda, T. (2005) Monitoring of antifouling booster bio- cides in water and sediment from the port of Osaka, Japan. Archives of Environmental Contamination and Toxicolgy, 48, 303-310. http://dx.doi.org/10.1007/s00244-004-0084-2
[18] García-Reyes, J.F., Jackson, A.U., Molina-Díaz, A. and Cooks, R.G. (2009) Desorption electrospray ionization mass spectrometry for trace analysis of agrochemicals in food. Analtical Chemistry, 81, 820-829. http://dx.doi.org/10.1021/ac802166v
[19] Grogan, H.M. and Jukes, A.A. (2003) Persistence of the fungicides thiabendazole, carbendazim and prochloraz- Mn in mushroom casing soil. Pest Management Science, 59, 1225-1231. http://dx.doi.org/10.1002/ps.759
[20] Castillo, L.E., Martínez, E., Ruepert, C., Savage, C., Gi- lek, M., Pinnock, M. and Solis, E. (2006) Water quality and macroinvertebrate community response following pesticide applications in a banana plantation, Limon, Costa Rica. The Science of the Total Environment, 367, 418-432. http://dx.doi.org/10.1016/j.scitotenv.2006.02.052
[21] Kim, H.J., Lee, D.S. and Kwon, J.H. (2010) Sorption of benzimidazole anthelmintics to dissolved organic matter surrogates and sewage sludge. Chemosphere, 80, 256- 262. http://dx.doi.org/10.1016/j.chemosphere.2010.04.029
[22] García-Calzón, J.A., Muñíz-Alvarez, J.L. and López- Fonseca, J.M. (1999) Adsorption and two-dimensional condensation of thiabendazole at a mercury/solution in- terface. Journal of Colloid and Interface Science, 219, 144-148. http://dx.doi.org/10.1006/jcis.1999.6455
[23] Spanoghe, P., Ryckaert, B., Van-Gheluwe, C. and Van- Labeke, M.C. (2008) Fate of vinclozolin, thiabendazole and dimethomorph during storage, handling and forcing of chicory. Pest Management Science, 66, 126-131. http://dx.doi.org/10.1002/ps.1838
[24] USEPA Prevention, Pesticides and Toxic Substances (2002) Thiabendazole and salts. EPA-738-F-02-002.
[25] El-Nahhal, Y., Nir, S., Polubesova, T., Margulies, L. and Rubin, B. (1998) Leaching, phytotoxicity and weed con- trol of new formulations of alachlor. Journal of Agricul- tural Food Chemistry, 46, 3305-3313. http://dx.doi.org/10.1021/jf971062k
[26] Tomlin, C.D. (2000) The pesticides manual. 11th Edition, British Crop Protection Council.
[27] El-Nahhal, Y. and Lagaly, G. (2005) Salt effects on the adsorption of a pesticide on modified bentonite. Colloid and Polymer Science, 283, 968-974. http://dx.doi.org/10.1007/s00396-004-1244-7
[28] El-Nahhal, Y. and Safi, J. (2006) Bentonite for controlled release of linuron. Journal of Pest Control and Environ- mental Science, 14, 57-71.
[29] El-Nahhal, Y. and Safi, J. (2010) Adsorption of bromox- ynil to modified bentonite: Influence of pH, and temper- ature. Journal of Pesticide Science, 35, 333-338. http://dx.doi.org/10.1584/jpestics.G09-41
[30] Rodríguez, R., Picó, Y., Font, G. and Mañes, J. (2002) Analysis of thiabendazole and procymidone in fruits and vegetables by capillary electrophoresis-electrospray mass spectrometry. Journal of Chromatography A, 949, 359- 366. http://dx.doi.org/10.1016/S0021-9673(01)01395-4
[31] European Commission, Directorate-General Health and Comsumer Protection (2001) Thiabendazole. 7603/VI/97- Final, 22 March 2001.
[32] Rytwo, G. and Tavasi, M. (2003) Addition of a monova- lent cationic pesticide to improve efficacy of bipyridyl herbicides in Hulah Valley soils. Pest Management Sci- ence, 59, 1265-1270. http://dx.doi.org/10.1002/ps.732
[33] Giles, C.H., Mac-Ewan, T.H., Nakhwa, S.N. and Smith, D.A. (1960) System of classification of solution adsorp- tion isotherms, and its use in diagnosis of adsorption me- chanisms and in measurement of specific surface areas of solids. Chemical Society, 111, 3973-3993.
[34] Kishk, M.F., Abu-Sharar, M.T., Bakry M.N. and Abu- Donia B.M. (1979) Sorption-desorption characteristics of methyl parathion by clays. Archives of Environmental Contamination and Toxicology, 8, 637-645. http://dx.doi.org/10.1007/BF01054866







