Food and Nutrition Sciences
Vol.06 No.05(2015), Article ID:55289,7 pages
10.4236/fns.2015.65046
Processing Effects on Phytochemical Content and Antioxidative Potential of Ginger Zingiber officale
R. Offei-Okyne 1, J. Patterson1, L. T. Walker2, Martha Verghese1,2*
1Nutrition and Carcinogenesis Laboratory, Department of Food & Animal Sciences, Alabama A&M University, Normal, AL, USA
2Food Chemistry Laboratory, Department of Food & Animal Sciences, Alabama A&M University, Normal, AL, USA
Email: *Martha.verghese@aamu.edu
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 November 2014; accepted 27 March 2015; published 1 April 2015
ABSTRACT
Ginger (Zingiber officinale) is rich in antioxidants and phytochemicals with anti-inflammatory, antimicrobial and anticancer properties. Ginger has been used as a condiment and for the treatment of ailments for many years. However, there are limited studies on the antioxidant and scavenging power of processed ginger. The objectives of this study were to determine the effects that process- ing has on the total phenolic and flavanoid content of ginger and its antioxidant potential using 1,1-Diphenyl-2-picrylhydrazyl (DPPH), Radical-Scavenging Activity and Ferric Reducing Antioxidant Power (FRAP). Fresh ginger (FG) was used as a control with oven drying (OD) sun drying (SD) and freeze drying (FD) as processing methods. Freeze-dried ginger (77.87%) showed a significantly higher (p < 0.05) free radical scavenging ability as compared to other treatment groups (18.40% - 72.90%). However, sundried ginger had the highest ferric-reducing antioxidant power at 35.28 ± 0.69 mmol Fe (II)/mg ginger. Total phenolic content content (mg GAE/100g) for fresh, oven died, sun-dried, and freeze-dried were 514.02 ± 3.92, 796.46 ± 8.16, 878.76 ± 13.17, and 1021.15 ± 12.95, respectively. Flavanoid content of processed ginger was highest in freeze-dried samples (458.82 CE/100g ginger).
Keywords:
Ginger, Phenolic Compound, Flavonoid Compound, DPPH

1. Introduction
Ginger, Zingiber officinale Roscoe, (Zingiberaceae) is widely cultivated in tropical and subtropical countries where it is mainly used as food spice and for medicinal purposes. In the foods and beverages industry, ginger is used as an additive for its aroma and pungency [1] . The chemical composition of fresh ginger varies with varieties or cultivar [2] , the location for which the ginger is cultivated [3] , extraction methods [4] and the processing methods [5] . In the USA, ginger ranks among 20 top-selling herbal supplements with retail sale of about United States (US) $1.38 million in the 2010, a 17.4% increase over previous year [6] . Ali, Blunden, Tanira, and Nemmar [7] reviewed the pharmacological, phytochemical and toxicological properties of ginger including anti-in- flammatory, antioxidant, anti-lipidemic, immuno-modulatory, anti-tumorigenic, and anti-hyperglycemic properties and its impact on several metabolic and age-related degenerative diseases.
Ginger is mainly traded on the world market in processed forms as powders and extracts. Attributes of exportable ginger are mainly based on aroma or pungency which is derived from key chemical components such as volatile essential oils and non-volatile oleoresin [8] . These volatile essential oils include zingeberene, curcumene and farnesene [9] , while the non-volatile pungent compounds contain the bioactive compounds such as gingerols, shogaols, zingerone and paradols. These compounds give ginger its characteristic hot sensation [10] . The most abundant non-volatile compound in ginger is gingerol [7] , but it is also rich in many bioactive compounds such diarylheptanoids, 3-dihydroshogaols and other methyl ether derivatives [10] [11] . During thermal processing, gingerol is dehydrated to form shagaol which gives ginger its characteristic pungency. pH may also effect the conversion rate of gingerol to shogoal [12] . Processing effects the chemical profile of ginger through loss of especially cyclic compounds and the appearance of others may be due to degradation, oxidation and rearrangement of constituent molecules [11] [13] . Different dehydration methods for instance microwave drying, freeze drying, air drying and vacuum drying effect the availability of some compounds [13] . In a study done by Chan, Lim, Wong,, Lim, Tan, Lianto, & Yong, [14] on the effects of thermal and non-thermal drying methods on the antioxidant properties, total phenolic content and ferric-reducing power of leaves and tea of ginger resulted in the decline of these properties and attributes in some of the drying methods.
The purpose of this research was to investigate the total phenolic, and flavonoid content, the Radical-Sca- venging Activity (DPPH) and Ferric Reducing Antioxidant Power (FRAP) from extracts of fresh and processed ginger.
2. Materials and Methods
2.1. Chemicals
All solvents and chemicals were obtained from Fisher Scientific (New Jersey, USA).
2.2. Sample Preparation
Healthy and matured ginger rhizomes were purchased from a local health store in Huntsville, Alabama, USA in April 2012. Ginger samples were washed and manually peeled, weighed and sliced into pieces of about 4 mm in thickness and further chopped in tiny bits before processing. Sundried ginger was prepared as per standard protocol (8 hours of sunlight exposed) until desired level of dryness was obtained.
2.2.1. Freeze Drying
Freeze drying of ginger was done as follows: An initial weight of 25 g of sliced ginger used was freeze dried at a temperature of −80˚C over a period of 18 hours at vacuum pressure of 3 kPa in a freeze dryer (Virtis Genesis-25 Super XL, USA)
2.2.2. Oven Drying
Twenty-six grams of chopped ginger were spread evenly in a pre-weighed aluminum pan and placed in an experimental oven (Isotemp Oven, Fisher Marietta OH, model 6925) at a temperature of 75˚C over a period of 12 hours (AOAC 1990).
2.3. Extraction Methods
In the extraction, 5 g of treatment samples were weighed into a container fully covered with aluminum foil and then 50 ml of 80% methanol was added and placed on stirrer (VWR Standard Multiposition Stirrers, model 986912, USA) for 2 hours at room temperature. The samples were centrifuged (Thermo Fischer Scientific, Lengend XTR Centrifuge, Germany) at a temperature of 4˚C at 4000 ×g for 20 minutes. The supernatant was decanted and filtered with Whatman No. 2 filter paper into round bottom flask and then attached to a rotary evaporator (BUCHI, Rotavapor R-215, Switzerland), the solvent was evaporated at a temperature of 40˚C and at a rotational speed and pressure of 270 rpm and 204 mbar respectively for a period of 5 hours. The residue was then made up to a final volume of 10 ml with 80% methanol and stored at −80˚C until further analyzed for antioxidant and free-radical-scavenging activity, total phenolic and flavonoid content. Triplicate were done for each of the samples; fresh ginger (FG), oven dried (OD), sun dried (SD) and freeze dried (FD) ginger.
2.4. Chemical Analysis of Fresh and Processed Ginger
2.4.1. Determination of Total Phenolic in Fresh and Processed Ginger
Total phenolic content was measured using a modified Folin Ciocalteu method as described by Singleton, Orthofer, and Lamuela-Raventos [15] and Dewanto, Wu, Adom, and Liu [16] . Five milligram of gallic acid was weighed and diluted in 10 mL of ddH2O from which the standard curve was generated from dilution ranging from 0.02 - 0.4 mg/mL. Twelve and a half microliters of briefly prepared Folin Ciocalteau reagent was added to each of 12.5 µL of samples and standards and then incubated for 5 minutes. Fifty microliters of ddH2O and 125 µL of 7% NaHCO3 were then added to the mixture and again incubated at room temperature for 90 minutes after which the absorbance was read at 750 nm using a microplate reader (Synergy HT, BioTek Instruments, USA). Total phenolic concentration of the fresh and processed ginger was expressed in mg Gallic Acid Equivalent per 100 g of ginger as the average of 3 replications.
2.4.2. Total Flavonoids
Total flavonoid concentration was determined by a colorimetric assay as described by Kim, Jeong, and Lee [17] . Standards were prepared from different concentrations of catechin (0.02 - 0.4 mg/ml). To 25 µL each of diluted sample, blank and standards, 7.5 μL of a 5% NaNO2 solution were added and incubated for 5 minutes after which 15 µL 10% AlCl3 was added. Fifty microliters of 1M NaOH and 40 μL of ddH2O were added to the mixture, absorbance read at 510 nm and the quantification was done using the calibration curve. Total flavonoid concentration of the fresh and processed ginger was expressed in mg catechin per 100 g of ginger as the average of 3 replications.
2.4.3. Determination of Ferric Reducing Antioxidant Power (FRAP)
The FRAP assay was conducted as described by Benzie and Strain [18] with modifications. Fourteen milligram of FeSO4∙7H2O was dissolved in 5 ml of distilled from which standard curve (0.1 mM - 1.0 mM) was prepared. FRAP reagents were prepared from a combination of three reagents. Reagent A, 300 mM acetate buffer of pH 3.6; Reagent B, TPTZ solution which was made up of 31 mg of 2, 4, 6 tri pyridyl-s-triazine (TPTZ) in 10 mL 40 mM HCl, and Reagent C had 54 mg of ferric chloride (FeCl3∙6H2O) dissolved in 10mL of distilled water. Ten microliters of known dilutions of ginger extracts were combined with 200 µL of FRAP reagent in a 96 well plate and the mixture was incubated for 10 minutes at 37˚C. After incubation, the samples were analyzed at an absorbance of 593 nm with a microplate reader (Synergy HT, BioTek Instrument Incorporated, Vermont, USA). The change in absorbance was compared with the standards at a minute interval for 4 minutes. The samples were analyzed in triplicates and the results are expressed as mmol Fe (II)/g dry weight.
2.4.4. Measurement of DPPH Scavenging Activity
Ginger, Zingiber officinale, has shown to exhibit highest antioxidant activity among the ginger spices [19] . The free-radical scavenging activity of 80% methanol ginger extracts of fresh and processed ginger was determined as described by Brand-Williams, Cuvelier, & Berset, [20] with some modification. Forty microliters of appropriately diluted concentrations of ginger extracts were added to 200 µL of 0.4 mg 1,1-Diphenyl-2-picrylhydrazyl (DPPH) dissolved in 10 mL of 100% methanol. The mixture was agitated and incubated in the dark at room temperature and absorbance was measured with microplate reader (Synergy HT, BioTek Instrument Incorporated, Vermont, USA) at 517 nm at time intervals of 0, 30, 60, and 90 minutes. Free radical scavenging activity was measured as the amount of extracts required to reduce the initial absorbance at 517 nm of DPPH radical concentration by 50% (IC50) as compared to the control. This was calculated as the Percentage inhibition of DPPH as shown below.
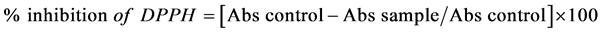
2.5. Statistical Analysis
Data was analyzed by (Analysis Of Variance) ANOVA using the SAS 9.0 (2011) statistical program using data from three independent experiments. Each experiment was repeated three times. Values were given as means ± SEM (Standard Error of the Mean) and means were separated using Tukey’s studentized range test. Level of significance were considered at P ≤ 0.05
3. Results
This study observed that selected processing methods resulted in significant (P < 0.05) differences in total phenol and flavonoid contents, as well as the free radical scavenging activity and ferric reducing antioxidant power.
3.1. Phenolic and Flavonoids
The highest total phenolic content was observed in freeze dried ginger (FD) (1021.15 mg GAE/100g) and the lowest was observed in fresh ginger (FG) extracts (514.02 mg GAE/100g). In this study, significant differences (P < 0.05) were observed among all processed ginger extracts and the FG which was used as a control. FG had a significantly (P < 0.05) lower total flavonoid content (238.96 ± 5.12 mg CE/100g) compared to processed ginger; oven dried (OD), sun dried (SD) and freeze (FD). The total flavonoid content among all the ginger extracts were significantly (P < 0.05) different with the highest flavonoid content observed in FD extracts (458.82 ± 8.37) as shown in Table 1. The total phenol and flavonoid contents for FG, OD, SD, and FD are given on a dry weight basis.
The total phenols were 35.46%, 41.50% and 49.66%, respectively higher in OD, SD and FD compared to FG (control). However, total flavonoid in FG was 26% and 42% and 92% lower compared to OD, SD and FD. Among the processed ginger, OD had significantly (P < 0.05) lower total phenolic (796.46 ± 8.16 mg GAE/100g) and flavonoid contents (302.18 ± 6.12 mg CE/100g) compared to SD and FD.
3.2. DPPH Radical Inhibition
The DPPH radical inhibition (%) by fresh and processed ginger extracts is shown in Figure 1. At the lowest concentration (12.5 mg/ml), inhibition of DPPH by FG was 9.25 which was the lowest of all ginger extracts. FD and SD ginger exhibited the greatest effect in DPPH inhibition at the lowest concentration (12.5 mg/ml).
Figure 1. DPPH radical scavenging activity of ginger extracts at different concentration. Abbreviations: DPPH-1,1-Diphenyl-2-picrylhydrazyl.
Table 1. Total phenolic and flavonoid content in fresh and processed ginger.
FG: Fresh ginger; OD: Oven dried ginger; SD: Sun dried ginger; FD: Freeze dried ginger; GAE: Gallic Acid Equivalents; CE: Catechin Equivalents. Values are Mean ± SEM; n = 4. abcdValues not sharing common superscript are significantly different (P < 0.05) using Tukey’s studentized range test.
The DPPH inhibition at 25 - 100 mg/ml was similar for the processed ginger (OD, SD and FD). However, FG had a similar inhibition compared to OD, SD and FD only at 50 mg/ml. The percentage DPPH inhibition increased from 12.5 mg to 100 mg/ml with the highest inhibition seen at 100 mg/ml. FG showed lower potential in scavenging free radicals at lower concentrations, but had the highest inhibition (100%) at the highest concentration (100 mg/ml).
At a concentration of 12.5 mg/ml, processed ginger (OD, SD and FD) had a 5-, 6- and 7-fold greater inhibition of the DPPH radical compared to FG. However, at 25 mg/ml, OD, SD and FD had a 38%, 42% and 48% higher effect in inhibition of the DPPH radical compared to FG.
3.3. Ferric Reducing Antioxidant Potential (FRAP)
The Ferric Reducing Antioxidant Potential (FRAP) of ginger extracts is shown in Table 2. The highest FRAP was observed in SD (35.28 ± 0.69 mmol Fe [II]/mg) and the lowest observed in OD extracts (27.78 ± 0.63 mmol Fe [II]/mg). In this study, no significant differences (P < 0.05) were observed among the control (FG), OD and FD. However, there were significant differences (P < 0.05) between SD and all the processed ginger (OD) and (FD) and the control (FG). The FRAP was 3% higher in FG compared to OD. However, FRAP in FG was 4.12% and 23.14% higher compared to FD and SD. Among the processed ginger extracts, OD (27.78 ± 0.63 mmol Fe (II)/mg) had a significantly (P < 0.05) lower FRAP compared to FD and SD.
4. Discussion
In this study, processing impacted the chemical profile of ginger, as it was observed in Table 1 and Table 2 and Figure 1. Studies by Noor, Azian, Mustafa Kamal, & Nurul Azlina [21] , indicated that processing changed the structure of the cell wall, impacting on the availability of phytochemicals. They further stated that cell walls upon rapture, releases essential oils and oleoresin (which is rich in these phytochemicals). Ali et al. [7] studied the extraction of different parts of Halia spp of ginger with different solvents: methanol, acetone, and chloroform. Their studies revealed increase in extract with increase polarity of solvent.
A comparison of organic solvent extracts of processed and unprocessed ginger as antibacterial agent was studied by Gao and Zhang [22] . Although the study showed antibacterial activity against gram-positive bacteria and gram-negative, there was no significant differences among the different processed ginger groups.
Thermal methods are known to cause a decline in the total phenolic content compared to non thermal methods [14] [23] . This was evident in this study where the total phenolic and flavonoid content of the processed ginger increased with reducing temperature used in processing, that is, from oven drying, sun drying, and freeze drying.
The low levels of antioxidant activity seen in the control (fresh ginger) were as a result of lower yield in essential oils. The DPPH activity is comparable to the FRAP of the processed and fresh ginger [24] . A similar result was seen in a study by Denre (2014), where the DPPH inhibition of several herbs and spices was negatively correlated with the total phenolic content [25] .
5. Conclusion
According to the results in this experiment, total phenols in the processed ginger were higher compared to the unprocessed ginger. Freeze dried ginger had the highest phenolic and flavonoids compared to other processed
Table 2. Free-radicial scavenging (DPPH) activity and ferric reducing antioxidant power (FRAP) of fresh and processed ginger.
FG: Fresh ginger; OD: Oven dried ginger; SD: Sun dried ginger; FD: Freeze dried ginger; DPPH: Diphenynl-1-picrylhydrazyl; FRAP: Ferric Reducing Antioxidant Power. Values are Mean ± SEM; n = 4. abcdValues not sharing common superscript are significantly different (P < 0.05) using Tukey’s studentized range test.
ginger (OD and SD). Similar results was seen where FD had showed higher potency in ferric reducing antioxidant power and percentage DPPH inhibition. However, at concentration of 50 mg/ml processed and fresh ginger had the same DPPH inhibition percentage. At higher concentration (100 mg/ml), FG was a better free radical scavenger compared to the processed ginger.
Acknowledgements
This work was supported by the USDA National Institute of Food and Agriculture, [Evans-Allen] project [ALAX- 012-0412].
Cite this paper
R.Offei-Oknye,J.Patterson,L. T.Walker,MarthaVerghese, (2015) Processing Effects on Phytochemical Content and Antioxidative Potential of Ginger Zingiber officale. Food and Nutrition Sciences,06,445-451. doi: 10.4236/fns.2015.65046
References
- 1. Bartley, J.P. and Jacobs, A.L. (2000) Effects of Drying on Flavour Compounds in Australian-Grown Ginger (Zingiber officinale). Journal of the Science of Food and Agriculture, 2, 209-215.
http://dx.doi.org/10.1002/(SICI)1097-0010(20000115)80:2<209::AID-JSFA516>3.0.CO;2-8 - 2. Wohlmuth, H., Smith, M.K., Brooks, L.O., Myers, S.P. and Leach, D.N. (2006) Essential Oil Composition of Diploid and Tetraploid Clones of Ginger (Zingiber officinale Roscoe) Grown in Australia. Journal of Agricultural and Food Chemistry, 4, 1414-1419.
http://dx.doi.org/10.1021/jf0521799 - 3. Bailey-Shaw, Y.A., Williams, L., Junor, G., Green, C.E., Hibbert, S.L., Salmon, C. and Smith, A.M. (2008) Changes in the Contents of Oleoresin and Pungent Bioactive Principles of Jamaican ginger (Zingiber officinale Roscoe.) during Maturation. Journal of Agricultural and Food Chemistry, 14, 5564-5571.
http://dx.doi.org/10.1021/jf072782m - 4. Gong, F., Fung, Y.S. and Liang, Y.Z. (2004) Determination of Volatile Components in Ginger Using Gas ChromatographyMass Spectrometry with Resolution Improved by Data Processing Techniques. Journal of Agricultural and Food Chemistry, 21, 6378-6383.
http://dx.doi.org/10.1021/jf040102z - 5. Variyar, P.S., Gholap, A.S. and Sharma, A. (2006) Changes in Flavor Components In-Irradiated Fresh Ginger (Zingiber officinale) Rhizomes during Storage. Journal of Herbs, Spices and Medicinal Plants, 12, 25-35.
http://dx.doi.org/10.1300/J044v12n01_03 - 6. Blumenthal, M., Lindstrom, A., Lynch, M.E. and Rea, P. (2011) Herbal Sales Continue Growth-Up to 3.3% in 2010. HerbalGram.
http://www.cms.herbalgram.org/herbalgram/issue90/MarketReport.html - 7. Ali, B.H., Blunden, G., Tanira, M.O. and Nemmar, A. (2008) Some Phytochemical, Pharmacological and Toxicological Properties of Ginger (Zingiber officinale): A Review of Recent Research. Food and chemical Toxicology, 2, 409420.
http://dx.doi.org/10.1016/j.fct.2007.09.085 - 8. Famurewa, A.V., Emuekele, P.O. and Jaiyeoba, K.F. (2011) Effect of Drying and Size Reduction on the Chemical and Volatile Oil Contents of Ginger (Zingiber officinale). Journal of Medicinal Plants Research, 14, 2941-2944.
- 9. Govindarajan, V.S. (1982) Chemistry, Technology, and Quality Evaluation: Part 1. Critical Reviews in Food Science and Nutrition, 17, 1-96.
http://dx.doi.org/10.1080/10408398209527343 - 10. Wohlmuth, H., Leach, D.N., Smith, M.K. and Myers, S.P. (2005) Gingerol Content of Diploid and Tetraploid Clones of Ginger (Zingiber officinale Roscoe). Journal of Agricultural and Food Chemistry, 53, 5772-5778.
http://dx.doi.org/10.1080/10408398209527343 - 11. Jolad, S.D., Lantz, R.C., Chen, G.J., Bates, R.B. and Timmermann, B.N. (2004) Fresh Organically Grown Ginger: Composition and Effects on LPS-Induced PGE2 Production. Phytochemistry, 65, 1937-1954.
- 12. Bhattarai, S., Tran, V.H. and Duke, C.C. (2001) The Stability of Gingerol and Shogaol in Aqueous Solution. Journal of Pharmaceutical Sciences, 90, 1658-1664.
http://dx.doi.org/10.1002/jps.1116 - 13. Ding, S.H., An, K.J., Zhao, C.P., Li, Y., Guo, Y.H. and Wang, Z.F. (2012) Effect of Drying Methods on Volatile of Chinese Ginger (Zingiber officinale Roscoe). Food and Bioproducts Processing, 90, 515-524.
http://dx.doi.org/10.1016/j.fbp.2011.10.003 - 14. Chan, E.W.C., Lim, Y.Y., Wong, S.K., Lim, K.K., Tan, S.P., Lianto, F.S. and Yong, M.Y. (2009) Effects of Different Drying Methods on the Antioxidant Properties of Leaves and Tea of Ginger Species. Food Chemistry, 113, 166-172.
http://dx.doi.org/10.1016/j.foodchem.2008.07.090 - 15. Singleton, V.L., Orthofer, R. and Lamuela-Raventós, R.M. (1999) Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. Methods in Enzymology, 299, 152-178.
http://dx.doi.org/10.1016/S0076-6879(99)99017-1 - 16. Dewanto, V., Wu, X., Adom, K. and Liu, R. (2002) Thermal Processing Enhances the Nutritional Value of Tomatoes by Increasing the Total Antioxidant Activity. Journal of Agricultural and Food Chemistry, 50, 3010-3014.
http://dx.doi.org/10.1021/jf0115589 - 17. Kim, D., Jeong, S.W. and Lee, C.Y. (2003) Antioxidant Capacity of Phenolic Phytochemicals from Various Cultivars of Plums Original Research Article. Food Chemistry, 3, 321-326.
- 18. Benzie, I.F.F. and Strain, J.J. (1999) Ferric Reducing/Antioxidant Power Assay: Direct Measure of Total Antioxidant Activity of Biological Fluids and Modified Version for Simultaneous Measurement of Total Antioxidant Power and Ascorbic Acid Concentration. Methods in Enzymology, 299, 15-27.
http://dx.doi.org/10.1016/S0076-6879(99)99005-5 - 19. Nakatani, N. (1997) Antioxidants from Spices and Herbs. In: Shahidi, F., Ed., Natural Antioxidants: Chemistry, Health Effects, and Applications, AOCS Press, Champaign, 64-75.
- 20. Brand-Williams, W., Cuvelier, M.E. and Berset, C. (1995) Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT-Food Science and Technology, 28, 25-30.
http://dx.doi.org/10.1016/S0023-6438(95)80008-5 - 21. Noor Azian, M., Mustafa Kamal, A.A. and Nurul Azlina, M. (2004) Changes of Cell Structure in Ginger during Processing. Journal of Food Engineering, 62, 359-364.
http://dx.doi.org/10.1016/S0260-8774(03)00251-6 - 22. Gao, D. and Zhang, Y. (2010) Comparative Antibacterial Activities of Extracts of Dried Ginger and Processed Ginger. Pharmacognosy Journal, 2, 41-44.
http://dx.doi.org/10.1016/S0975-3575(10)80077-X - 23. Kortei, N., Odumetten, G.T., Obodai, M., Appiah, V., Akuamou, F., Adu-Bobi, A., Annan, S.N., Armah, J.N. and Acquah, S.A. (2014) Evaluating the Effect of Gamma Radiation on the Total Phenolic Content, Flavanoids, and an Antioxidant Activity of Dried Pleurotus ostreatus Stored in Packaging Materials. Advances in Pharmaceutics, 2014, Article ID: 262807.
http://dx.doi.org/10.1155/2014/262807 - 24. El-Ghorab, A.H., Nauman, M., Anjum, F.M., Hussain, S. and Nadeem, M. (2010) A Comparative Study on Chemical Composition and Antioxidant Activity of Ginger (Zingiber officinale) and Cumin (Cuminum cyminum). Journal of Agricultural and Food Chemistry, 58, 8231-8237.
http://dx.doi.org/10.1021/jf101202x - 25. Denre, M. (2014) The Determination of Vitamin C, Total Phenolics, and Antioxidant Activity of Some Commonly Cooking Spices Crops Used in West Bengal. International Journal of Plant Physiology and Biochemistry, 6, 66-70.
http://dx.doi.org/10.5897/IJPPB2014.0210
NOTES
*Corresponding author.




