Food and Nutrition Sciences
Vol.05 No.18(2014), Article ID:49903,6 pages
10.4236/fns.2014.518187
Influence of the Multispecies Probiotic Ecologic® BARRIER on Parameters of Intestinal Barrier Function
Saskia Van Hemert, Geline Ormel
Research and Development Winclove Probiotics, Amsterdam, The Netherlands
Email: Ormel@winclove.nl
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 24 June 2014; revised 16 August 2014; accepted 25 August 2014
ABSTRACT
The intestinal epithelium is a single cell layer that forms the largest and most important barrier against the external environment. A proper functioning of the intestinal barrier is essential for maintaining optimal health. A disrupted intestinal barrier function has been associated with the development of autoimmune and inflammatory diseases. Probiotics are live microorganisms that confer a health benefit to the host and have been associated with prevention of diseases such as antibiotic-associated diarrhea, irritable bowel syndrome and inflammatory bowel disease. Probiotics can improve and restore the intestinal barrier function in diverse ways and effects are thought to be species- and even strain-specific. With this research the influence of probiotic bacteria in Ecologic® BARRIER on parameters of intestinal barrier function was investigated. The results show that each bacterial strain in Ecologic® BARRIER is in vitro capable to improve the epithelial barrier in diverse ways.
Keywords:
Probiotics, Ecologic® BARRIER, Intestinal Barrier Function

1. Introduction
The intestinal mucosa is the largest interface between the outside world and the human internal milieu. Across a surface area that approximates the size of a soccer field, it is here where we prevent the highest concentration of bacteria from invading our internal environment while allowing nutrient and water absorption by a single cell layer of epithelium. The ability to control the invasion of harmful content from the lumen is called intestinal mucosal barrier function. While the epithelial layer forms the most obvious physical boundary between inner and outer environment, the full complexity of factors that control intestinal barrier function reaches beyond the epithelium and is not fully understood. Throughout the intestine a single layer of epithelial cells covers the inner surface and is responsible for this barrier function.
Tight junctions are protein structures that allow selective passage of ions and small molecules. They form, in healthy subjects, a tight barrier to protein seized molecules and bacteria. The task of the epithelium is not only to keep bacteria and antigens out while absorbing nutrients, but also to allow contact between luminal contents and immune cells. This occurs through limited and highly controlled uptake of antigens and bacteria. This seemingly paradoxical task is, however, crucial in the induction of targeted and protective mucosal immune responses to pathogens as well as to the development of oral tolerance to commensals and food antigens.
The barrier function of the intestine can be influenced by different factors, like heredity, bacterial flora, diet, psychological stress, oxidative stress, exercise, and drugs [1] . Increased permeability of the epithelial barrier has been associated with many gastrointestinal inflammatory disorders, like inflammatory bowel diseases (Crohn’s disease, ulcerative colitis and pouchitis) and celiac disease [2] . An increased permeability can also lead to increased levels of endotoxins in the blood, which are linked to systemic inflammatory diseases, like metabolic syndrome, diabetes, atherosclerosis, chronic fatigue syndrome, autism, migraine and rheumatoid arthritis. Probiotics have proven capabilities to enhance the epithelial barrier, due to different working mechanisms [3] .
Probiotics are living microorganisms that have beneficial effects on the health of the host [4] . More and more is known that probiotic effects are species- and even strain-specific. Probiotics have proven strain-dependent capabilities in vitro as well as in vivo to improve the epithelial barrier function via different mechanisms [3] [5] [6] . They affect the epithelial barrier in numerous, diverse ways. In this study, the strain specific capacities of the bacterial strains in the multispecies probiotic Ecologic® BARRIER, a probiotic product especially designed to improve the epithelial barrier and to increase the resistance to disturbance of the intestinal barrier, were investigated in different screenings.
2. Development of Ecologic® BARRIER
The product Ecologic® BARRIER contains 8 different probiotic strains (B. bifidum W23, B. lactis W52, L. acidophilus W37, L. brevis W63, L. casei W56, L. salivarius W24, Lc lactis W19, Lc. Lactis W58) in a carrier matrix of maize starch and maltodextrins with a viable cell count of 2.5 × 109 CFU/gram. The advised dosage is 2 grams , twice daily. The product has been developed by Winclove Probiotics, Amsterdam the Netherlands.
The probiotic strains were selected based on the following criteria:
In vitro strengthening of the epithelial barrier
Inhibition of mast-cell activation
Inhibition of pro-inflammatory cytokines
Decreasing lipopolysaccharide load
2.1. In Vitro Strengthening of the Epithelial Barrier
The ability to control the invasion of harmful content from the lumen is called “intestinal mucosal barrier function”. The integrity of this barrier function can be measured with transepithelial electrical resistance (TEER). TEER is an in vitro measurement of the movement of ions across the paracellular pathway. Maintenance of the intestinal integrity is critical for essential physiological processes. Therefore, a reduction in TEER may represent an early expression of cell damage and indicates that the barrier function of the intestine is decreased.
A monolayer of villus-like Caco-2 cells was grown on a filter. Caco-2 cell lines have been extensively used over the last twenty years as a model of the intestinal barrier. The experiment was performed with an inflammatory stressor (a combination of TNF-α and IL1-ß), which is known to reduce the relative TEER of the Caco-2 cells. The monolayer of Caco-2 cells was first exposed during 1 hour to the probiotic bacteria followed by exposure to the inflammatory stressor in the presence of the same probiotic bacteria , also for 1 hour. After a recovery time of 4 and 24 hours the TEER of the monolayer was measured. The results were compared to the TEER of a monolayer that was exposed to stressor alone and an unexposed. The results are shown in Figure 1.
Three strains (B. bifidum W23, L. acidophilus W37 and L. brevis W63) in Ecologic® BARRIER could for more than 90% protect the epithelial cell against the cytokine induced dysfunction of the barrier, whereas two others (L. casei W56 and Lc. lactis W19) had a partial effect, three strains (B. lactis W52, L. salivarius W24 and Lc. lactis W58) did not show an effect in this assay.
Figure 1. Effect of strains present in Ecologic® BARRIER on cytokine induced barrier dysfunction after 24 hours incubation with TNF-a and IL-1b. The value of the control, without the addition of any stressor, was set at 100%.
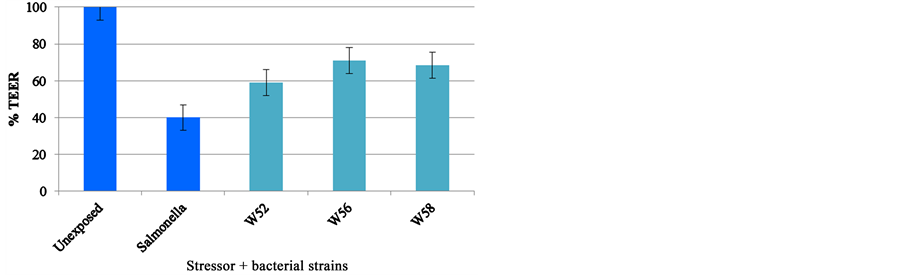
A smaller TEER screening was performed with the pathogenic bacteria Salmonella enteritis 857, which has shown to decrease the relative TEER in Caco-2 cells. In a small experiment with six bacterial strains, an epithelial cell-line (CaCo-2) was damaged by a pathogenic bacterium, Salmonella enteritidis. Three of the 6 strains were able to diminish the decrease in transepithelial resistance (and thus strengthen the barrier function) due to the Salmonella significantly (see Figure 2). These three strains are present in Ecologic® BARRIER.
From these two graphs it can be concluded that all strains in Ecologic® BARRIER, with the exception of L. salivarius W24, can strengthen the intestinal barrier function after being damaged by either Salmonella or inflammatory cytokines.
2.2. Inhibition of Mast-Cell Activation
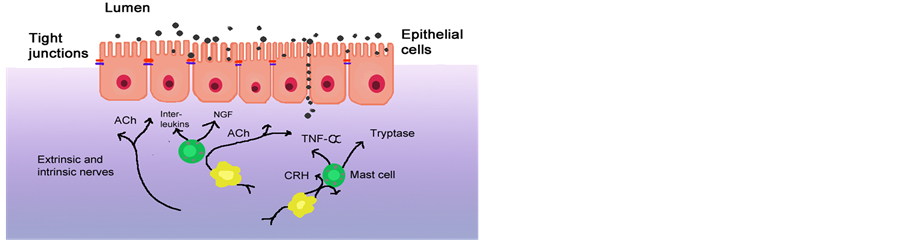
Tight junctions are important proteins in the epithelial cells to maintain the barrier. They can open by the activation of mast cells. These mast cells can be triggered by the hormone Corticotropin-Releasing Hormone (CRH), which is released during stress. Triggering of the mast-cells by CRH results in granulation of mediators like nerve growth factor, different cytokines or β-hexosaminidase. These effects are presented in Figure 3. Upon the release of these mediators, the tight junction will open resulting in increased permeability of the small intestine [7] . Research has shown that certain probiotic strains can decrease the CRH-induced degranulation of mast-cells and thereby the opening of the epithelial barrier due to stress.
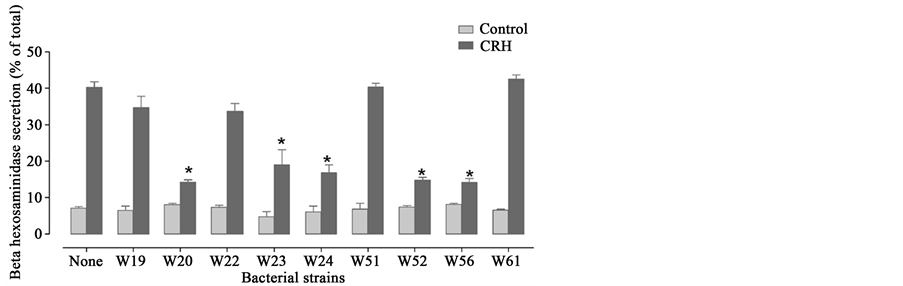
In this experiment mast-cells were pre-treated with probiotic strains and subsequently stimulated with CRH. The amount of released β-hexosaminidase was measured to get insight on the protective effects of probiotics to inhibit the increase of the permeability. The results are shown in Figure 4. The strains B. bifidum W23, L. salivarius W24, B. lactis W52 and L. casei W56 prevent the release of β-hexosaminidase upon CRH treatment, thereby inhibiting an increase in permeability. Lc. lactis W19 had no preventive effect. The other strains of Ecologic® BARRIER were not tested in this assay.
2.3. Stimulations of Regulatory Cytokines
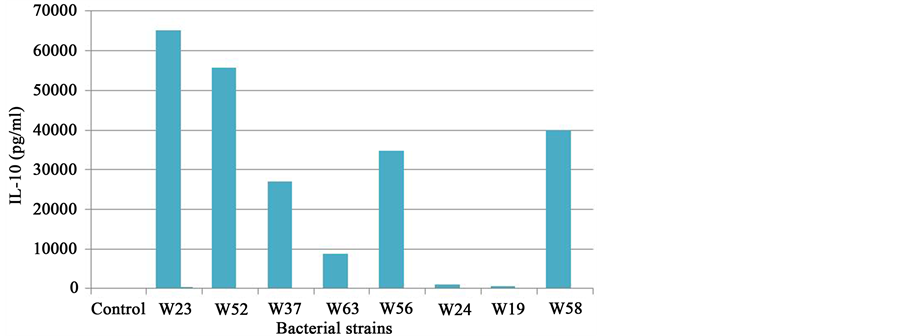
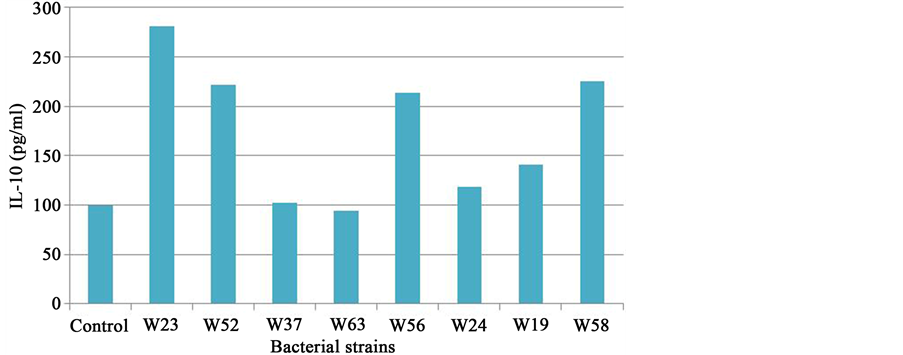
A large part of the immune system (approximately 80%) is concentrated in and around the intestinal mucosa. The intestinal microbiota is involved in maturation of the immune system as demonstrated in studies in germ- free mice [9] . In turn the microbiota in the intestine plays an important role in the regulation of functions in the immune system [10] . The immune system can be modulated by probiotic bacteria and these effects are highly species- and strain-specific [10] -[12] . Inflammatory signals cause disruption of the epithelial barrier. Interleukin-10 (IL-10) is an important cytokine to regulate immune responses and to prevent excessive pro-inflamma- tory responses. Induction of IL-10 by different probiotic strains was measured with an in vitro test with peripheral blood mononuclear cells (Figure 5 and Figure 6).
Six strains in Ecologic® BARRIER; B. bifidum W23, B. lactis W52, L. acidophilus W37, L. brevis W63, L. casei W56 and Lc. lactis W58 stimulate IL-10 production, thereby preventing excessive pro-inflammatory responses and disruption of the epithelial barrier.
Figure 2. Probiotic effect of Ecologic® BARRIER on Salmonella-induced decrease in transepithelial electrical resistance.
Figure 3. A simplified scheme of potential mechanisms involved in neuro-immune modulation of the intestinal barrier function.
Figure 4. Probiotic effect on mast-cells stimulated with CRH. Data from F. Lutgendorff, Defending the barrier. Effects of probiotic on endogenous de- fens mechanims. 2009, University of Utrecht: Utrecht, The Netherlands [8] . Strains marked with * showed a significant decrease in permeability.
2.4. Decreasing Lipopolysaccharide (LPS) Load
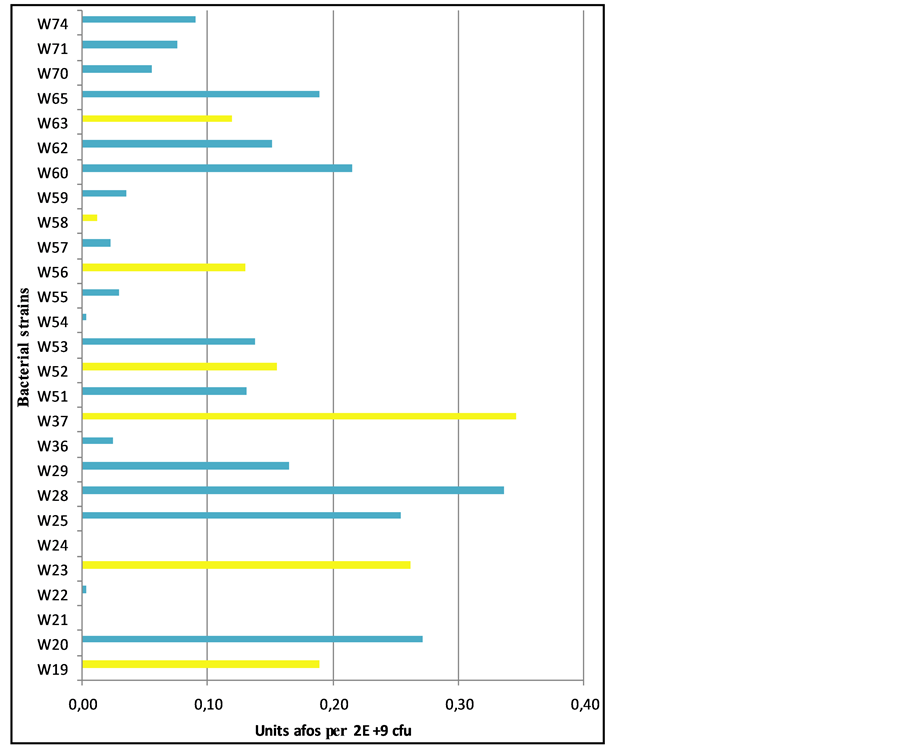
Lipopolysaccharide (LPS) is part of the cell wall of Gram-negative bacteria and is a very pro-inflammatory component. Leaky gut syndrome is associated with increased levels of LPS in blood plasma and lowering of the uptake of LPS is an important aspect of a good intestinal barrier function. Some probiotic bacteria have the capacity to break down LPS by the activity of the enzyme alkaline phosphatase. In an in vitro experiment the activity of this enzyme has been measured. In this assay the substrate p-nitrophenylphosphate is split is p-nitro- phenol, which has a yellow color. The color intensity is measured with a spectrophotometer. The results are shown in Figure 7. From the strains present in Ecologic® BARRIER, Lc. Lactis W19, B. bifidum W23 and L. acidophilus W37 are among the most active strains with regard to alkaline phosphatase activity.
Figure 5. Il-10 production of PHA stimulated peripheral blood mononuclear cells by individual strains present in Ecologic® BARRIER.
Figure 6. Il-10 production of non-stimulated peripheral blood mononuclear cells by individual strains present in Ecologic® BARRIER.
2.5. Overview of in Vitro Results
Table 1 presents a summary of the capacities of each bacterial strain in Ecologic® BARRIER to influence intestinal barrier function. As can be seen form the table, strains in Ecologic® BARRIER all perform well on 1 or more parameters of intestinal barrier function.
3. Discussion and Conclusions
Probiotic bacteria can have an effect on the epithelial barrier via different molecular pathways and routes. While probiotic effects are species- and even strain-specific a multispecies product is a very good choice to support these different routes. All strains present in Ecologic® BARRIER were carefully selected based on their in vitro capacities to improve the epithelial barrier and have shown to positively influence one or more parameters of intestinal barrier function. Because bacteria can negatively influence each others working mechanisms, bacterial interaction has also been examined (data not shown). Bacteria in Ecologic® BARRIER do not negatively influence each other. With the results of the conducted experiments we expect to have developed a probiotic product which is able to improve the intestinal barrier function. However, this will have to be shown in vivo as well. To our knowledge Ecologic® BARRIER is the first probiotic product especially developed to improve the intestinal barrier function.
A probiotic product which improves the epithelial barrier might have positive effects on systemic inflammatory diseases and diseases associated with increased pro-inflammatory cytokines, like migraine, autism, diabetes
Figure 7. Alkaline phosphatase activity of different strains. Strains present in Ecologic® BARRIER are marked yellow.
Table 1. Overview of influence of bacterial strains in Ecologic® BARRIER on parameters of intestinal barrier function.
+: Positive effect; −: no data or no effect.
type II, arthritis and inflammatory bowel diseases. There are plans to test the effect on epithelial barrier function ex vivo, or in vivo.
References
- Farhadi, A., Banan, A., Fields, J. and Keshavarzian, A. (2003) Intestinal Barrier: An Interface between Health and Disease. Journal of Gastroenterology and Hepatology, 18, 479-497. http://dx.doi.org/10.1046/j.1440-1746.2003.03032.x
- Marchiando, A.M., Graham, W.V. and Turner, J.R. (2010) Epithelial Barriers in Homeostasis and Disease. Annual Review of Pathology-Mechanisms of Disease, 5, 119-144.
- Ohland, C.L. and Macnaughton, W.K. (2010) Probiotic Bacteria and Intestinal Epithelial Barrier Function. American Journal of Physiology. Gastrointestinal and Liver Physiology, 298, G807-G819.
- Hill. C., et al. (2014) The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nature Reviews Gastroenterology & Hepatology, 11, 506-514.
- Miyauchi, E., et al. (2012) Mechanism of Protection of Transepithelial Barrier Function by Lactobacillus Salivarius: Strain-Dependence and Attenuation by Bacteriocin Production. American Journal of Physiology and Gastrointestinal Liver Physiology, 303, G1029-G1104. http://dx.doi.org/10.1152/ajpgi.00003.2012
- van Hemert, S., Verwer, J. and Schütz, B. (2013) Clinical Studies Evaluation Effects of Probiotics on Parameters of Intestinal Barrier Function. Advances in Microbiology, 3, 212-221,
- Zheng, P.Y., Feng, B.S., Oluwole, C., Struiksma, S., Chen, X., Li, P., Tang, S.G. and Yang, P.C. (2009) Psychological Stress Induces Eosinophils to Produce Corticotrophin Releasing Hormone in the Intestine. Gut, 58, 1473-1479. http://dx.doi.org/10.1136/gut.2009.181701
- Lutgendorff, F. (2009) Defending the Barrier. Effects of Probiotic on Endogenous Defens Mechanims. Universiteit Utrecht, Utrecht.
- Hooper, L.V. and Gordon, J.I. (2001) Commensal Host-Bacterial Relationships in the Gut. Science, 292, 1115-1118. http://dx.doi.org/10.1126/science.1058709
- Delcenserie, V., Martel, D., Lamoureux, M., Amiot, J., Boutin, Y. and Roy, D. (2008) Immunomodulatory Effects of Probiotics in the Intestinal Tract. Current Issues in Molecular Biology, 10, 37-54.
- Gill, H. and Prasad, J. (2008) Probiotics, Immunomodulation, and Health Benefits. Advances in Experimental Medicine and Biology, 606, 423-454.
- Nova, E., Warnberg, J., Gomez-Martinez, S., Diaz, L.E., Romeo, J. and Marcos, A. (2007) Immunomodulatory Effects of Probiotics in Different Stages of Life. British Journal of Nutrition, 98, S90-S95.