Open Journal of Synthesis Theory and Applications
Vol.04 No.03(2015), Article ID:58109,16 pages
10.4236/ojsta.2015.43005
Mechanical, Thermal and Morphology Properties of Thermoplastic Polyurethane Copolymers Incorporating α,ω-Dihydroxy-[poly(propyleneoxide)-poly (dimethylsiloxane)-poly(propyleneoxide)] of Varying Poly(propyleneoxide) Molecular Weight
Zeliang Dai, Kai Yang, Qingzhi Dong
Shanghai Key Laboratory of Advanced Polymeric Materials, Key Laboratory for Ultrafine Materials of Ministry of Education, School of Materials Science and Engineering, East China University of Science and Technology, Shanghai, China
Email: qzhdong@ecust.edu.cn
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 16 January 2015; accepted 17 July 2015; published 20 July 2015
ABSTRACT
Novel segmented thermoplastic polyurethane (TPU) copolymers were synthesized using two-step solventless bulk polymerization. 4,4’-methylenediphenyl diisocyanate (MDI) and 1,4-Butanediol (BDO) were used to form hard segment of TPU and α,ω-dihydroxy-[poly(propyleneoxide)-poly (dimethylsiloxane)-poly(propyleneoxide)] (α,ω-dihydroxy-(PPO-PDMS-PPO)) was used to form soft segment of TPU, where the molar ratio of the ?N=C=O/OH was 1.02 and the hard segment weight percentage was 30%. A series of TPUs were characterized by fourier transform infrared spectroscopy (FT-IR). The investigation of triblock oligomer’s PPO molecular weight impact on the derived TPU’s mechanical properties, thermal performance, surface water repellency and morphology performance was carried by Instron material tester, thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), water contact angles (WCA), scanning electron microscope and energy dispersive X-ray spectroscopy (SEM-EDX) and wide angle X-ray diffraction (WAXD), respectively. FT-IR confirmed α,ω-dihydroxy-(PPO-PDMS-PPO) well cooperating into urethane struc- ture and analyzed hydrogen bonding between N-H group with hard segment C=O group and N-H group with soft segment C-O-C group. DSC and WAXD results showed α,ω-dihydroxy-(PPO-PDMS- PPO) segments crystallization. SEM-EDX results showed that the presence of a spherulitic morphology, which arose from the crystallization of the PPO segments. The thermal properties measured by TGA and DSC were slightly affected by molecular weight of PPO and microphase separation. The weight loss of TPUs started between 294˚C and 300˚C, and Tg was in the range of −70˚C to −107˚C. TPU copolymers’ surface hydrophobicity property was excellent with WCA range of 95˚ to 112˚. TPU-3 with 1000 molecular weight PPO has the optimized mechanical properties with tensile strength 16.4 MPa and the modulus at 100% elongation 6.2 MPa and elongation 398%.
Keywords:
Thermoplastic Polyurethane Copolymers, Mechanical Properties, Thermal Performance, Hydrophobicity Property, Morphology, α,ω-Dihydroxy-[poly(propyleneoxide)-poly(dimethylsiloxane)-poly(propyleneoxide)]

1. Introduction
PUs have been found in wide applications such as insulation rigid foams, comfort flexible foams, coatings and adhesives, due to their unique properties, and great efforts have been made in the variegation of their chemical and physical properties [1] . To improve the mechanical properties of PUs, chemical cross-linkers were introduced into the hard segment through hyperbranched polyesters (HBPs) as cross-linking agents. HBPs has specific three-dimensional structure, large number of end functional groups, and remarkable properties such as high solubility in different solvents, low viscosity in solution and in melt used in PU coatings with increased high chemical and abrasion resistance and good mechanical properties [2] [3] . Different Boltorn HBPs were used together with polyester or polyether macrodiols for the synthesis of PUs. However, for these PUs to be useful at high temperatures, their architectures should be modified by introducing thermally stable siloxane linkages [4] -[7] .
PDMS is well known for its excellent biostability and biocompatibility as many copolymers based PDMS have been synthesized and studied for various biomedical applications in recent times. The introduction of PDMS into the polymer chain has the advantage of imparting some of the attractive properties of PDMS into PUs, such as high flexibility, excellent thermal, oxidative, and hydrolytic stabilities, high UV stability, biocompatibility, hydrophobicity, and low surface energy [8] -[10] . Similar to conventional polyether- and polyester- based TPUs, PDMS prepolymers with Mn values in the range 1000 to 5000 g∙mol−1 are widely utilized in siloxane-urethane copolymers. However, the compatibility between PU and PDMS prepolymer is poor due to the great difference between their solubility parameters (the solubility parameters of PDMS and urethane group are 15.6 and 37.2 J1/2/cm3/2, respectively), which makes it difficult to prepare these modified PUs [11] .
The attempts to synthesize PDMS-based TPUs resulted in copolymer with poor mechanical properties, which was attributed to the low molecular weight and also to the absence of the intersegment hydrogen bonding between PDMS and the hard segments. To overcome this, a second soft segment macrodiol can be added to interject chemical species of intermediate polarity between the hard and PDMS soft segments. It has been established that terminal units attached to the ends of PDMS prepolymer can act as “compatibilzer” between the polar hard and the nonpolar PDMS segments. In numerous articles, the preparation of thermoplastic polyurethane and polyuria copolymers based on end-functionalized PDMS preopolymers, such as hydroxylhexyl, aminopropyl, methylaminopropyl PDMS, with high molecular weight were reported. High molecular weight siloxane-based urethane and urea copolymers with good thermal and mechanical properties can be prepared by stoichiometric reaction of diisocynates and PDMS prepolymers, without any chain extender or by the conventional prepolymer method through the employment of chain extenders.
Pergal et al. initially synthesized PU networks based on α,ω-dihydroxy (ethylene oxide-poly(dimethylsiloxane)- ethylene oxide) (EO-PDMS-EO) and Boltornhyperbranched polyester of the second pseudogeneration (BH-20) in the formation of a heterogeneous network, due to the appearance of macroscopic phase separation in the polymers. However, PU networks based on EO-PDMS-EO had low cross-linking density, a lower degree of microphase separation, relatively good thermal stability and poor mechanical physical properties [12] -[17] .
After that, Pergal et al. continued to investigate the effect of the soft segment structure on the properties of PU networks based on BH-20 and α,ω-dihydroxypropyl PDMS. Hydroxypropyl terminated PDMS was used as the soft segment and 4,4’-methylenediphenyl diisocyanate/hyperbranched polyester (MDI-HBP) was used as the hard segment. The results revealed that the synthesized polyurethanes have good thermal, thermomechanical and surface properties compared to EO-PDMS-EO derived PUs. Polyurethane networks prepared from this specific combination of reactants enable exploitation of their good features and simultaneously introduce good thermal and surface properties, swelling ability, rubber elasticity, and other properties, which are important for different applications, especially for elastomers or coatings. The type of PDMS macrodiols could have significant influence of the properties and final morphology of PU networks based on HBPs [18] .
Additionally, the use of a biocompatible triblockprepolymer (Mn = 6100 g∙mol−1) consisting of terminal crystabillizablepoly(e-caprolactone) blocks and a central PDMS block in the synthesis of polyurethanes was reported. The combination of the properties of PCL and PDMS, such as biocompatbibility and slow enzymatic degradation rates, makes these block copolymers excellent candidates for surface modifying additives, drug encapsulation, and biomaterial applications. Incorporation of a high content of siloxane segments into the backbone of TPUs significantly improves their biostability. The synthesis and characterization of these thermoplastic polyurethanes based on soft PCL-PDMS-PCL and hard MDI/BD segments and the influence of reaction conditions on the structure, molecular weight, thermal, and some physical properties of the segmented TPUs with high contents of PCL-PDMS-PCL segments were investigated. The results revealed that PCL segments with high soft segment contents crystallized with showing spherulitic morphology.
To overcome this problem, methyl allylpolypropyleneoxide polyether was used to hydrosilate with silanicpolydimethlydsiloxane to formulate α,ω-dihydroxyl poly(propyleneoxide)-PDMS-poly(propyleneoxide) ABA triblock oligomers (PPO-PDMS-PPO). PPO-PDMS-PPOs were used as a “compatibilizer” between nonpolar PDMS blocks and the polar urethane blocks.
We believe that the presence of PPO-PDMS-PPO macrodiol would enhance the hard segment/soft segment micorphasesepration and improve thermomechanical and surface properties of the synthesized PUs in comparison with those previously synthesized PUs based on EO-PDMS-EO or pure polyether polyol as the soft segment.
In this work, we have synthesized a series of siloxane-urethane TPU copolymers based on α,ω-dihydroxy- (PPO-PDMS-PPO) oligomers with varied PPO molecular weights using two-step solventless bulk polymerization. The study on PPO molecular weight impacting on TPU copolymers’ mechanical, thermal, hydrophobic, morphology properties is studied in this paper.
2. Experiment
2.1. Materials
α,ω-dihydroxy-polypropyleneoxide-polydimethylsiloxane-polypropyleneoxide (α,ω-dihydroxy-(PPO-PDMS-PPO)) ABA triblock oligomers (supplied by Momentive) were dried at room temperature under vacuum for 2 hours. 1H-NMR, 13C-NMR and 29Si-NMR spectroscopy methods were used to analyze the molecular weights (Mn) of PPO-PDMS-PPO, in the range of 2836 - 4636 g∙mol−1. The molecular weights of the central PDMS chain was Mn (PDMS) = 1636, while the molecular weights of terminal polypropyleneoxide polyether were varied from 600 g∙mol−1 to 1500 g∙mol−1, its structure diagram was listed as Scheme 1:
4,4’-Methylenediphenyl diisocyanate (MDI) (Bayer), with an isocyanate content of 33.6 wt.%, determined by the standard dibutylaminebacktitration method, was used as received. The structure of MDI was listed as Scheme 2:
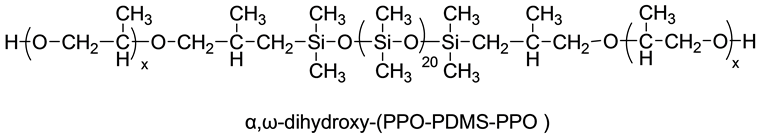
Scheme 1. α,ω-Dihydroxy-(PPO-PDMS-PPO) structure.
1,4-Butanediol (BDO) (Aldrich, purity is higher than 99.5%) was purified by vacuum distillation. The structure of BDO was listed as Scheme 3.
2.2. Synthesis of α,ω-Dihydroxy-(PPO-PDMS-PPO) ABA Triblock Oligomers Prepared TPU Copolymers
TPUs were synthesized using a two-step bulk polymerization method without a solvent. The reactants were MDI, BDO and α,ω-dihydroxy-(PPO-PDMS-PPO) (Mn range from 2836 to 4636 g∙mol−1). The first pre-polymer reaction was performed in a four-neck, round-bottom flask equipped with a mechanical stirrer, a dry nitrogen inlet, a reflux condenser and a dropping funnel. The flask was placed in a silicone oil bath. According to molar ratio of composition listed in Table 1 and the polymerization procedure listed in Figure 1, a series of α,ω-di- hydroxy-(PPO-PDMS-PPO) modified TPUs were synthesized. The molar ratio between the reacting ?N=C=O groups (from MDI) and the -OH groups (from BDO and α,ω-dihydroxy-(PPO-PDMS-PPO), respectively) was fixed at 1.02. The content of the hard segment was fixed at 30% by weight, and the reaction temperature was optimized at 70˚C.
A typical synthesis for the sample TPU-1 copolymer using α,ω -dihydroxy-(PPO-PDMS-PPO) with a molecular of 2836 g∙mol−1 soft segment is described as an example. The starting reaction mixture of 50 g (0.0176 mol) of α,ω-dihydroxy-(PPO-PDMS-PPO) and 34.76 g (0.139 mol) of MDI was added to a flask at room temperature and heated to 70˚C under a nitrogen atmosphere. Next, the remaining 50 g (0.0176 mol) of α,ω-dihydroxy- (PPO-PDMS-PPO) was added dropwise to the flask via the dropping funnel. The reaction mixture was stirred

Scheme 2. 4,4’-MDI.

Scheme 3. 1,4-BDO.
Figure 1. TPU copolymers preparation.
Table 1. Composition of TPU copolymers.
a: α,ω-dihydroxy-(PPO-PDMS-PPO): MDI: BDO; -N=C=O/-OH = 1.02 from TPU-1 to TPU-5. b: HS, weight % of hard segment, predetermined by the composition of the reaction mixtures.
for 150 min at 70˚C to prepare the isocyanate-terminated pre-polymer. The NCO content during the reaction was analyzed using the standard di-butylamine back-titration method. The first-step pre-polymer preparation was ended when the pre-polymer NCO content reached 6.28%. Next, 100 g of pre-polymer and a stoichiometric amount of BDO (6.02 g, 0.0668 mol) were blended together using a stirring machine. The mixture was stirred for 3 min and cast onto a Teflon® plate, which was placed in an oven at 100˚C for 1 h and subsequently dried under vacuum at 90˚C for 12 h. The cast TPU materials were maintained at room temperature for 7 days before their properties were tested.
2.3. Characterization
2.3.1. FTIR
A FT-IR (Nicolet 6700 spectrometer, America Thermo Fisher Scientific Company) was used to identify TPU copolymers’ functional groups. The scanning range spanned from 500 to 4000 cm−1 at a resolution of 4 cm−1, and 64 scans were collected for each sample.
2.3.2. SEM-EDX
The surfaces or “air exposed” areas and cross-section morphologies of TPU copolymers were analyzed via field-emission SEM. The samples were adhered to aluminum sample holders and sputter-coated with an Au layer. The TPU images were obtained using a JEOL JSM-6460LV microscope, and microphotographs were obtained at a working distance of 10 mm and an acceleration voltage of 20 kV.
2.3.3. WAXD
WAXD was performed using a diffractometric system RINT 2000, diffractometer with CuKα radiation using a copper anode (tube: 40 kV, 100 mA, λ = 0.154178 nm). The diffraction patterns were obtained in the Bragg angle range of 5˚ to 50˚. The scan speed was 0.02 s per step in all measurements.
2.3.4. Mechanical Property Tests
Mechanical property tests were carried out using an Instron 4302 universal testing machine with a 1 kN load cell and a crosshead speed of 500 mm/min. The sample length for tensile tests was 12.5 cm. At least three pieces of each sample were tested to determine the ultimate tensile strength, ultimate elongation and elastic modulus at 100% elongation. The average results for three pieces of each sample were used as the final values.
2.3.5. Hardness Tests
TPUs hardness tests were carried out using a Shore A apparatus (Hildebrand, Germany). The results of the hardness tests were recorded when the force number was stable after penetrating the needle onto the sample. The average results for five measurements of each sample were used as the final value.
2.3.6. WCA
The water contact angles of the TPU copolymer were measured using a Krüss DSA100 machine according to the sessile drop method. A single, 20 μL drop of distilled water was deposited onto the TPUs’ film surface, and the contact angle at 26˚C was recorded after 30 s using a camera connected to a computer. The final contact angle values were taken as the average of five measurements.
2.3.7. TGA
The thermal stability of the TPU copolymers was determined using Q5000 V3.5 Build 252 thermogravimetric analyzer (America TA Company); the samples were heated in the temperature range of 25˚C to 700˚C, at a heating rate of 10˚C/min. The TGA scans were recorded under a dynamic nitrogen atmosphere with a flow rate of 25 ml/min. The average weight of the samples was approximately 10 mg.
2.3.8. DSC
DSC was performed on a Q2000 V23.12 Build 103 differential scanning calorimeter (America TA Company); the samples were scanned, under a nitrogen atmosphere, at a heating rate of 10˚C/min and a cooling rate of 20˚C/min over a temperature range of ?150˚C to 200˚C. The weight of the samples was approximately 5 mg. In the first scan, the samples were heated from −150˚C to 200˚C to eliminate their thermal history. The samples were subsequently cooled to −150˚C and finally heated to 200˚C.
3. Results and Discussion
3.1. FT-IR
Figure 2 displays the IR spectra of α,ω-dihydroxy-(PPO-PDMS-PPO) prepared various TPU copolymers with different PO molecular weights. The absorption of the characteristic peaks occurred at 803 cm−1 (Si-CH3 rocking- band), 1016 cm−1 (Si-O-Si stretching band), and 1258 cm−1 (symmetric CH3 bending band) showed incorporation of the PDMS segment with methyl allyl-polypropyleneoxide to formulate the siloxane-urethane copolymers. The absorption bands occurred at approximately 3325 cm−1 (N-H stretching band) and 1701 - 1728 cm−1 (the carbonyl, C=O, stretching band) showed urethane linkage formulation of TPU copolymers. Additionally, the examination of the FT-IR absorption bands of Silxoane-Urethane TPU copolymers in the carbonyl, C=O, stretching (1600 - 1800 cm−1) and N-H stretching (3200 - 3500 cm−1) indicated the hydrogen bonds presence [18] . According to the literature, part of C=O (1701 cm−1) groups were hydrogen bonded, the other part of C=O (1728 cm−1) groups were free non-bonded urethane C=O stretching [19] [20] .
FT-IR spectroscopy has been used to study the degree of phase separation for Siloxane-Urethane copolymers [21] . The degree of phase separation could be measured through the proportion of C=O hydrogen bonding. Urethane hydrogen bonding was formulated by N-H group with hard segment C=O group and N-H group with soft
Figure 2. FT-IR Spectra of α,ω-dihydroxy-(PPO-PDMS-PPO) prepared TPU copolymers.
segment C-O-C group. Figure 3 displays IR spectra in the range of 1600 cm−1 to 1800 cm−1 of Siloxane-Ure- thane TPU copolymers. 5 TPUs with various molecular weights of PPO exhibited two absorptions (1728 cm−1 and 1701 cm−1) bands in the 1600 - 1800 cm−1 domain.
According to the Gaussian deconvolution methodology, C=O absorption areas were analyzed by origin software to determine 1701 cm−1 peak intensity and 1728 cm−1 peak intensity. Equitation method is the following:
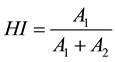 (1)
(1)
where HI represents hydrogen bonding index of urethane group, A1 represents hydrogen-bonded urethane carbonyl group in 1701 cm−1 and A2 represents free (non-bonded) urethane carbonyl group at 1728 cm−1. Calculated HI is summarized in Table 2.
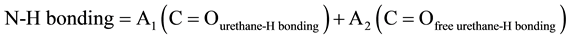 (2)
(2)
C=Ourethane-H bonding represents Hard segment concentration, and C=Ofree urethane-H bonding represents soft segment concentration.
Additionally, Figure 4 has shown the trend that the C=O 1701 cm−1 peak area decreased with the increase of PPO molecular weight. Table 2 confirmed that hydrogen bonding of C=O absorption intensity declined from
Figure 3. FT-IR spectra of TPUs in the 1600 - 1800 cm−1 domain.
Figure 4. FT-IR spectra of TPUs in 3100 - 3500 cm−1 domain.
Table 2. Curve-fitting results (area, %) for the C=O stretching region of the FT-IR spectra of TPU copolymers.
87.4% to 80.0% when methyl allyl-polypropyleneoxide molecular weights increased from 600 to 1500. It was explained that higher propylene oxide in the Siloxane-Urethane copolymers extended the soft segment concentration which resulted in hydrogen bonding of C-O-C and N-H group. The higher hydrogen bonding of C-O-C and N-H group provided less phase separation, which would be doubly confirmed by later SEM analysis.
3.2. Morphology and Element Analysis of the TPU Copolymers by SEM-EDX
SEM micrographs of five TPU copolymer films (TPU-1, TPU-2, TPU-3, TPU-4 and TPU-5) are displayed in Figures 5-9. The brighter regions in the micrographs represent hard domains, and the darker regions represent soft-segment-rich matrix. TPU-1 produced by α,ω-dihydroxy-(PPO-PDMS-PPO) with 600 molecular weights PPO clearly exhibits two separate morphologies, and the hard segments are not well distributed. With the PPO molecular weights of α,ω-dihydroxy-(PPO-PDMS-PPO) gradually increasing from 600 to 1200, the hard segment’ distribution around the soft segment became more uniform and better. However, TPU-5 made by α,ω-di- hydroxy-(PPO-PDMS-PPO) with 1500 molecular weights PPO exhibits poor hard segment distribution. This indicates that high PPO molecular weights in the α,ω-dihydroxy-(PPO-PDMS-PPO) structure provides less siloxane-urethane copolymers phase separation since the higher PPO oligomer has the longer hydrophobic and hydrophilic groups to well cooperate MDI and hard segments. The SEM microphotographs of TPU copolymers clearly showed a spherulitic structure typical for semincrystalline polymers. Additionally, the SEM microphotographs showed that TPU copolymers had distinct microphase separation. The micro-phase separation was present because of the large differences in solubility parameters between the PDMS soft segment and the urethane groups. In addition, a distinct ball-like structure of the SEM graphs of all TPU films were displayed, which arose from accumulation of the hard segments. From the obtained graphs, it could be concluded that the cross-sectioned morphology of TPU was affected by the molecular weight of PPO block of the soft segment. With increasing the PPO molecular weight of soft segment, hard segment soluble in soft segment phase increased, the micro-phase separation became worse. The tendency of phase separation was consistent with FTIR analysis results. The accumulation of hard segments became more and more obvious with the increase of PPO molecular weight, which served as physical cross-linking point roles. The accumulation of hard segments was well-distributed and the number was larger in the TPU-2 and TPU-3. But the accumulation of hard segments became larger and the uniformity of the distribution of dropped in the TPU-4 and TPU-5.
The EDX analysis results of TPU samples surface are displayed in Figure 10 and element % is summarized into Table 3. It could be noticed that silicone weight percentage on the surface of samples decreased from 20.48% to 12.24% with the increase of PPO molecular weights. This suggested that there was declined content of PDMS on the surface of TPU samples, which confirmed the tendency of shorter molecular PPO-PDMS-PPO easily to migrate to TPU surface. Additional confirmation of this conclusion was obtained from theoretically calculated Si-percentage in the synthesized samples (from 15.75% for TPU-1 to 9.63% for TPU-5), which were smaller than the values experimentally determined by EDX surface analysis. It also indicated that Carbon weight percentage on the surface of sample increased from 60.63% (TPU-1) to 65.55% (TPU-5) with the increase of PPO molecular weights of α,ω-dihydroxy-(PPO-PDMS-PPO) oligomer. This suggested that the α,ω-dihydroxy-(PPO- PDMS-PPO) triblock oligomer migrated to the surface of siloxane-urethane copolymers, which confirmed the further discussion on water repellency performance improvement based on using α,ω-dihydroxy-(PPO-PDMS- PPO) triblockolilgmers modified TPU copolymers.
Figure 5. TPU-1 SEM image.
Figure 6. TPU-2 SEM image.
Figure 7. TPU-3 SEM image.
Figure 8. TPU-4 SEM image.
Figure 9. TPU-5 SEM image.
3.3. WAXD Analysis of TPU Copolymers
The WAXD profile is displayed in Figure 11. Based on X-ray diffraction data, the unit cell of poly (MDI-BDO) is triclinic with dimensions a = 5.33 Å, b = 5.26 Å, c = 38.68 Å, α = 113.6, β = 116.0, γ = 94.4 degrees [22] . The amorphous halo of TPU copolymers was occurred at 2θ values of 12 degrees due to the presence of the PDMS block was observed, which indicated that PDMS was well cooperating into the TPU copolymers’ structure and well separated. With the PPO molecular weight increasing from 600 to 1500, the amorphous halo arbitrary intensity declined which exhibited that higher PPO in the α,ω-dihydroxy-(PPO-PDMS-PPO) prepolymer increased the polyurethane crystallization. TPU-1 copolymer had a peak at 2θ values of 20, which originated from the terminated PPO block of PDMS. The diffactograms of TPU-1 were very similar to the diffracograms of other four TPUs (such as TPU-2, TPU-3, TPU-4 and TPU-5). From the X-ray data, it was concluded that the soft PPO-PDMS-PPO segments in the TPUs formed a crystalline structure. This is due to crystallization resulting from PPO sequences [23] .
3.4. Mechanical Properties
Siloxane-Urethane elastomers made by different molecular weight of α,ω-dihydroxy-(PPO-PDMS-PPO) with varied PPO molecular weight mechanical properties were measured and shown in Figure 12. The results in
Figure 10. TPU copolymer surface element analysis.
Table 3. The EDX analysis results from TPU-1 to TPU-5.
a: weight percentage, b: molar percentage.
Table 4 shown that TPU-3 had the best tensile strength 16.4 MPa and the modulus at 100% elongation 6.2 MPa with acceptable good elongation 398%. Compared the tensile strength and 100% elongation modulus of TPU-1, TPU-2 and TPU-3, we could tell that the physical properties of TPUs increased with the PPO molecular weight increase from 600 to 1000, which could be explained that certain molecular weight PPO in the ABA triblock
Figure 11. TPU copolymers’ WAXD profiles.
Figure 12. Correlation between TPU mechanical property and PPO molecular weight.
Table 4. Mechanical property of TPU copolymers.
helped to improve the compatibility of silicone chain and urethane chain. The better compatibility of siloxane- urethane elastomers had the better mechanical properties. Compared the tensile strength and 100% elongation modulus of TPU-3, TPU-4 and TPU-5, we could tell that physical properties declined with the PPO molecular weight increase from 1000 to 1500, which could be explained that super higher molecular weight of PPO increased the soft segment concentration of the final elastomers. The higher soft segment concentration had the less N-H hydrogen bonding effect resulting in poor mechanical properties. The confirmed the FT-IR and SEM analysis results.
The hardness was mainly determined by the hard segment of elastomer and secondly determined by the hard segment distribution in the elastomer. In this research, hard segment weight percentage was fixed at 30%, the hardness of different structured siloxane-urethane elastomers didn’t change a lot.
3.5. Water Contact Angle of TPU Copolymer Films
The hydrophobicity of the surface of TPU copolymers’ film was investigated by water contact angle measurements (WCA). Water contact angle of the films greater than 90˚ were indicated hydrophobic [16] . Water contact angle results listed in Figure 13 shown that PPO molecular weight significantly impacted the final TPU hydrophobic performance. With the increase of the PPO molecular weight, TPU water contact angle was reduced from 112˚ to 95˚. The lower surface tension siloxane chain had the higher desperation in the surface when the PPO molecular weight was small which demonstrated the better hydrophobic performance with higher water contact angle. During SEM-EDX discussion, we saw that TPU-5 had the less Silicone weight percentage (9.63%) and higher Carbon weight percentage (65.55%) in the surface, which had the smaller water contact angle (95˚).
Thus, the introduction of PDMS chain to the TPU backbone significantly improved the wetting ability and hydrophobicity. However, the PPO block also had the negative impact on the wetting performance and hydrophobic performance. Overall five TPU copolymers had the improved and good hydrophobicity property compared to non-siloxane modified TPU.
3.6. Thermal Properties of TPU Copolymers
3.6.1. TG Analysis of TPU Copolymers
The thermal stability and degradation of TPU samples were investigated by TGA, as shown in Figure 14(a) & Figure 14(b). The characteristic temperature for weight losses of 5%, 50% and 90% were listed in Table 5. The weight loss of TPU copolymers started between 294˚C and 300˚C. From the presented results, it was concluded that the thermal stability of the TPU samples based on PPO-PDMS-PPO with different PPO molecular weight had the similar polymer degradation starting temperature which was much higher than pure urethane without siloxane modified urethane elastomers (Tg 5% = 281˚C). Table 5 indicated that PPO molecular weight didn’t impact the TPU thermal stability when the heating temperature was below 350˚C. When the copolymer degradation percentage was higher than 50%, the thermal degradation speed was affected by different PPO molecular
Figure 13. Correlation between TPU water contact angle and PPO molecular weight.

Figure 14. (a) TG curves of TPUs; (b) DTG curves of TPUs.
Table 5. Thermal analysis results of TPU copolymers.
weight and chain length. The longer PPO chain had the worse thermal stability performance and fast degradation. It was explained that the Si-C chain of copolymer started to break down during the temperature 350˚C - 400˚C, which could be observed on the DTG curves listed in Figure 14(b).
3.6.2. DSC Analysis of TPU Copolymers
The glass transition temperature (Tg) of TPU copolymers were measured by DSC at a heating rate of 10˚C/min, under a nitrogen atmosphere. The DSC curves of the Siloxane-Urethane Elastomers obtained during the second heating run were shown in Figure 15. The values of Tg and Tm tabulated in Table 5. From the results of the DSC curves and the value of Tg, we concluded that the Tg of TPU modified by PPO-PDMS-PPO increased from −107˚C to −70˚C. TPU-1 with 600 MW PPO had the lowest Tg which was closer closed to pure siloxane copolymers’ Tg (−123˚C). And TPU-5 with 1500 MW PPO had the highest Tg which was higher than pure polypropyleneoxide polyether (PPG) prepared TPU Tg (−46˚C) [24] . The data suggested that higher PPO molecular weight had the higher lower temperature Tg. The variation of melting point Tm of hard segment was very small in the range of 186˚C to 189˚C. The slight decline in Tm is the consequent of the decreasing degree of regularity of the crystallites and poorer microphase separation with the PPO molecular weight increase. This was explained by FT-IR analysis results that soft segment hydrogen bonding was increased since the PPO molecular weight increased in the PPO-PDMS-PPO structure resulting in less two phase separation and hard segment controlled the soft segment mobility. The more control came from hard segment, the Tg moved to higher temperature. And it was doubly confirmed by the WAXD results on the crystallization.
3.7. Conclusions
Novel TPU copolymers based on MDI/BDO as the hard and high content of α,ω-dihydroxy-(PPO-PDMS-PPO) as the soft segments were successfully synthesized by two-step solventless bulk polymerization, where the molar ratio of the -N=C=O/-OH was 1.02 and the hard segment weight percentage was 30%. The TPU copolymers incorporating α,ω-dihydroxy-(PPO-PDMS-PPO) of varying poly(propyleneoxide) molecular weight was analyzed
Figure 15. DSC thermograms (second heating run) of TPU copolymers.
by FT-IR which showed that incorporation of the PDMS segment with methyl allyl-polypropyleneoxide. And urethane hydrogen bonding was formulated by N-H group with hard segment C=O group and N-H group with soft segment C-O-C group. Hydrogen bonding of C=O absorption intensity decreased with the increase of methyl allyl-polypropleneoxide molecular weights. DSC and WAXD results showed that α,ω-dihydroxy-(PPO- PDMS-PPO) segments crystalized, whereas the hard segments containing two MDI/BDO units could not form a crystalline structure, probably because of their short chain length. SEM-EDX results showed that the presence of a spherulitic morphology, which arose from the crystallization of the PPO segments. High PPO molecular weights in the α,ω-dihydroxy-(PPO-PDMS-PPO) structure provided less TPU copolymers morphology microphase separation since the higher PPO oligomer had longer hydrophobic and hydrophilic group to well cooperate MDI and hard segments. The thermal properties measured by TGA were slightly affected by molecular weight of PPO and microphase separation. The weight loss of the siloxane-urethane copolymers started between 294˚C and 300˚C. Tg of TPU copolymers were in the range of −70˚C to −107˚C with revealing the excellent low temperature performance. TPU copolymers’ surface hydrophobicity property was analyzed by Water contact angle test. The water contact angle was in the range of 95˚ to 112˚ indicating a good hydrophobic performance. When PPO molecular weight was 1000, TPU-3 had the optimized mechanical properties with tensile strength 16.4 MPa and the modulus at 100% elongation 6.2 MPa and elongation 398%.
The synthesized TPUs based on α,ω-dihydroxy-(PPO-PDMS-PPO) oligomers are hydrophobic thermoplastic copolymers that would be considered for use in numerous applications, for example as medical implants. The influences of different contents of soft α,ω-dihydroxy-(PPO-PDMS-PPO) oligomers on the microstructure-me- chanical property relationships and biocompatibility behavior of TPUs will be studied and discussed in further publications.
Cite this paper
ZeliangDai,KaiYang,QingzhiDong, (2015) Mechanical, Thermal and Morphology Properties of Thermoplastic Polyurethane Copolymers Incorporating α,ω-Dihydroxy-[poly(propyleneoxide)-poly (dimethylsiloxane)-poly(propyleneoxide)] of Varying Poly(propyleneoxide) Molecular Weight. Open Journal of Synthesis Theory and Applications,04,41-57. doi: 10.4236/ojsta.2015.43005
References
- 1. Chattopadhyay, D.K. and Raju, K.V.S.N. (2007) Structural Engineering of Polyurethane Coatings for High Performance Applications. Progress in Polymer Science, 32, 352-418.
http://dx.doi.org/10.1016/j.progpolymsci.2006.05.003 - 2. Zagar, E. and Zigon, M. (2011) Aliphatic Hyperbranched Polyesters Based on 2, 2-Bis(methylol)propionic Acid— Determination of Structure, Solution and Bulk Properties. Progress in Polymer Science, 36, 53-88.
http://dx.doi.org/10.1016/j.progpolymsci.2010.08.004 - 3. Gite, V.V., Mahulikar, P.P. and Hundiwale, D.G. (2010) Preparation and Properties of Polyurethane Coatings Based on Acrylic Polyols and Trimer of Isophoronediisocyanate. Progress in Organic Coatings, 68, 307-312.
http://dx.doi.org/10.1016/j.porgcoat.2010.03.008 - 4. Asif, A., Shi, W.F., Shen, X.F. and Nie, K.M. (2005) Physical and Thermal Properties of UV Curable Waterborne Polyurethane Dispersions Incorporating Hyperbranched Aliphatic Polyester of Varying Generation Number. Polymer, 46, 11066-11078.
http://dx.doi.org/10.1016/j.polymer.2005.09.046 - 5. Cao, Q. and Liu, P. (2006) Structure and Mechanical Properties of Shape Memory Polyurethane Based on Hyperbranched Polyesters. Polymer Bulletin, 57, 889-899.
http://dx.doi.org/10.1007/s00289-006-0650-z - 6. Czech, P., Okrasa, L., Ulanski, J., Boiteux, G., Mechin, F. and Cassagnau, P. (2007) Studies of the Molecular Dynamics in Polyurethane Networks with Hyperbranchedcrosslinkers of Different Coordination Numbers. Journal of Applied Polymer Science, 105, 89-98.
http://dx.doi.org/10.1002/app.26106 - 7. Maji, P.K. and Bhowmick, A.K. (2009) Influence of Number of Functional Groups of Hyperbranchedpolyol on Cure Kinetics and Physical Properties of Polyurethane. Journal of Polymer Science Part A: Polymer Chemistry, 47, 731- 745.
http://dx.doi.org/10.1002/pola.23185 - 8. Florian, P., Jena, K.K, Allauddin, S., Narayan, R. and Raju, K.V.S.N. (2010) Preparation and Characterization of Waterborne Hyperbranched Polyurethane-Urea and Their Hybrid Coatings. Industrial & Engineering Chemistry Research, 49, 4517-4527.
http://dx.doi.org/10.1021/ie900840g - 9. Adhikari, R., Gunatillake, P.A. and Bown, M. (2003) Effect of Polydimethylsiloxanemacrodiol Molecular Weight on Properties and Morphology of Polyurethane and Polyurethaneurea. Journal of Applied Polymer Science, 90, 1565-1573.
http://dx.doi.org/10.1002/app.12748 - 10. Sheth, J.P., Aneja, A., Wilkes, G.L., Yilgor, E., Atilla, G.E., Yilgor, I. and Beyer, F.L. (2004) Influence of System Variables on the Morphological and Dynamic Mechanical Behavior of Polydimethylsiloxane Based Segmented Polyurethane and Polyurea Copolymers: A Comparative Perspective. Polymer, 45, 6919-6932.
http://dx.doi.org/10.1016/j.polymer.2004.06.057 - 11. Hernandez, R., Weksler, J., Padsalgikar, A. and Runt, J. (2007) Microstructural Organization of Three-Phase Polydimethylsiloxane-Based Segmented Polyurethanes. Macromolecules, 50, 5441-5449.
http://dx.doi.org/10.1021/ma070767c - 12. Pergal, M.V., Antic, V.V., Sanja, O., Milena, M.C. and Jasna, D. (2011) Influence of the Content of Hard Segments on the Properties of Novel Urethane-Siloxane Copolymers Based on a Poly(ε-caprolactone)-b-poly-(dimethylsiloxane)-b- poly(ε-caprolactone) Triblock Copolymer. Journal of Serbia Chemical Society, 76, 1703-1723.
http://dx.doi.org/10.2298/JSC110307146P - 13. Pergal, M.V., Antic, V.V., Govedarica, M.N., Godjevac, D., Ostojic, D. and Jonlagic, J. (2011) Synthesis and Characterization of Novel Urethane-Siloxane Copolymers with a High Content of PCL-PDMS-PCL Segment. Journal of Applied Polymer Science, 122, 2715-2730.
http://dx.doi.org/10.1002/app.33926 - 14. Pergal, M.V., Antic, V.V., Tovilovic, G., Nestorov, J., Vasiljevic-Radovic, D. and Djonlagic, J.J. (2012) In vitro Biocompatibility Evaluation of Novel Urethane-Siloxane Co-Polymers Based on Poly(ε-caprolactone)-block-poly(di-me- thylsiloxane)-block-poly(ε-caprolactone). Journal of Biomaterials Science, 23, 1629-1657.
- 15. Pergal, M.V., Dzunuzovic, J.V., Kicanovic, M., Vodnik, V., Pergal, M.M. and Jovanovic, S. (2011) Thermal Properties of Poly(urethane-ester-siloxane)s Based on Hyperbranched Polyester. Russia Journal Physical Chemistry, 85, 2251- 2256.
http://dx.doi.org/10.1134/S0036024411130243 - 16. Pergal, M.V., Dzunuzovic, J.V., Ostojic, S., Pergal, M.M., Radulovic, A. and Jovanovic, S. (2012) Poly(urethane-si- loxane)s Based on Hyperbranched Polyester as Crosslinking Agent: Synthesis and Characterization. Journal of Serbia Chemical Society, 77, 919-935.
http://dx.doi.org/10.2298/JSC111013006P - 17. Dzunuzovic, J.V., Pergal, M.V., Poreba, R., Ostojic, S., Lazic, N., Spirkova, M. and Jovanovic, S. (2012) Studies of the Thermal and Mechanical Properties of Poly(urethane-siloxane)s Cross-Linked by Hyperbranched Polyesters. Indus- trial & Engineering Chemistry Research, 51, 10824-10832.
http://dx.doi.org/10.1021/ie300927z - 18. Pergal, M.V., Dzunuozic, J.V. and Pergal, M.M. (2013) Structure-Property Correlation Study of Novel Poly(urethane- ester-siloxane) Networks. Industrial & Engineering Chemistry Research, 52, 6164-6176.
http://dx.doi.org/10.1021/ie400467j - 19. Ciolino, A.E., Pieroni, O.I., Vuano, B.M., Villar, M.A. and Valles, E.M. (2004) Synthesis of polybutadiene-graft- poly(dimethylsiloxane) and Polyethylene-graft-poly(dimethylsiloxane) Copolymers with Hydrosilylation Reactions. Journal of Polymer Science Part A: Polymer Chemistry, 42, 2920-2930.
http://dx.doi.org/10.1002/pola.20032 - 20. Zhang, C.L., Hu, J. and Chen, S.J. (2010) Theoretical Study of Hydrogen Bonding Interactions on MDI-Based Polyurethane. Journal of Molecular Modeling, 16, 1391-1399.
http://dx.doi.org/10.1007/s00894-010-0645-4 - 21. Milian, B., Vesna, A. and Marija, P. (2013) Influence of the Chemical Structure of Poly(urea-urethane-siloxane)s on Their Morphological, Surface and Thermal Properties. Polymer Bulletin, 70, 2493-2518.
http://dx.doi.org/10.1007/s00289-013-0968-2 - 22. Quay, J.R., Sun, Z., Blackwell, J., Briber, R.M. and Thomas, E.L. (1990) The Hard Segment Unit Cell for MDI-BDO- Based Polyurethane Elastomer. Polymer, 31, 1003-1008.
http://dx.doi.org/10.1016/0032-3861(90)90244-S - 23. Miller, J.A., Lin, S.B., Hwang, K.K.S., Wu, K.S., Gibson, P.E. and Cooper, S.L. (1985) Properties of Polyether-Poly- urethane Block Copolymers: Effects of Hard Segment Length Distribution. Macromolecules, 18, 32-34.
http://dx.doi.org/10.1021/ma00143a005 - 24. Yilgor, I. and McGrath, J.E. (1988) Polysiloxane Containing Copolymers: A Survey of Recent Developments. Advanced Polymer Science, 86, 1-86.
http://dx.doi.org/10.1007/BFb0025274

















