Advances in Breast Cancer Research
Vol.2 No.3(2013), Article ID:33520,5 pages DOI:10.4236/abcr.2013.23009
Associations of Polymorphisms of the CYP1A1 and CYP1B1 Cytochrome P450 Genes with Breast Cancer in Kazakhstan
Structural and Functional Genomics Laboratory, Institute of Molecular Biology and Biochemistry, Almaty, Kazakhstan
Email: *imbbtimur@mail.ru
Copyright © 2013 T. S. Balmukhanov et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received March 4, 2013; revised April 6, 2013; accepted April 15, 2013
Keywords: Breast Cancer; Gene Polymorphism; CYP1A1; CYP1B1; Kazakhstan
ABSTRACT
Associations of polymorphisms in rs4646903 site of CYP1A1 and rs1056836 site of CYP1B1 genes with the breast cancer (BC) were studied in two main ethnic groups of Kazakhstan Republic (Kazakhs and Russians). Total number of BC patients was 181, controls—397. The statistically significant differences were revealed in allele frequencies (χ2 = 5.93, р = 0.004) and in genotypes distribution (χ2 = 8.71, р = 0.015) in rs4646903 site of CYP1A1 gene in Kazakh but not in Russian group. The study of CYP1В1 rs1056836 site demonstrated differences in genotype distributions (χ2 = 7.48, р = 0.023) between BC patients and controls in Russian but not in Kazakh ethnic group.
1. Introduction
Cancers in hormone-respective tissues, including breast cancers occur at high incidence rates worldwide. Numerous studies investigating the relationship between breast cancer (BC) and polymorphisms in candidate genes involved in xenobiotic detoxification have been reported earlier including metaand pooled analysis of cytochrome P450 1B1 polymorphism association with breast cancer published and unpublished data from Environmental Carcinogenes Database [1]. Racial differences in breast cancer risks of hormone receptors subtypes of BC also have been previously reported [2]. Comparative analysis of SNP in estrogen-metabolizing enzymes for ovarian, endometrial and breast cancer in Novosibirsk was performed in Russia [3]. Interethnic differences between Russians, Tatars and Bashkirs in the frequency of the CYP1A1 and CYP1A2 are described in Bashkortostan, Russia [4].
Republic of Kazakhstan is a multinational state situated in the middle of Central Asia with the population about 17 million, mainly Kazakhs—Asians and Russian— Caucasians.
No studies describing associations of BC with Cytochrome P450 family genes have been earlier performed in Kazakhstan.
2. Materials and Methods
A total of 181 breast cancer patients and 397 of healthy age-matched controls (blood-donors with no family history of cancer) were analyzed for the polymorphisms of CYP1A1 at rs4646903 and CYP1B1 at rs1056836 by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) and two-stage PCR reaction, respectively. Peripheral blood samples were collected from the subjects with informed consent to participation in this study. The average age of the BC patients was 50.3 ± 11.6 years (Kazakhs), 55.7 ± 11.7 (Russians) and of controls 50.07 ± 8.47 (Kazakhs), 54.8 ± 5.9 (Russians).
Genomic DNA was isolated from blood samples using Axygen kits (USA) according to the manufacturer’s instructions. The PCR reaction was carried out in a total volume of 20 µl, 1 x buffer containing 67 mM Tris HCl, pH 8.8, 16.6 mM (NH4)2SO4, 2 mM MgCl2, 0.01% Tween 20, 0.15 mg/ml BSA, 2 pM of each primers, 0.25 mM each of four dNTPs, and 1 unit of Taq DNA polymerase (SibEnzime, Novosibirsk, Russia).
The polymorphism at rs4646903 of CYP1A1 gene was analyzed using the following primers: forward 5’-CAGTGAAGAGGTGTAGCCGC-3’ and reverse 5’-TAGGAGTCTTGTC-TCATGCC-3’. The thermocycling conditions were as follows: initial denaturation at 94˚C - 5 min, amplification at 94˚C - 1 min, 61˚C - 1 min, 72˚C - 1 min - 30 cycles, final elongation - 72˚C, 7 min [5]. Amplified DNA fragment of 340 bp, containing the region of interest, was subjected to restriction enzyme digestion with MspI. The enzyme cleaves a C/CGC sequence at CYP1A1 3’ region, generating 200 bp and 140 bp fragments.
Polymorphism at rs1056836 site of CYP1B1 gene was analyzed by means of two-stage PCR reaction [6]. At the first stage the following primers were used: F 5’-ATGCGCTTCTCCAGCTTTGT-3’; R 5’-TATGGAGCACACCTCACCTG-3’. The DNA fragment of 623 bp amplified in the first stage serves as a template for the second stage of PCR. The forward primer at the second stage was the same as in the first stage, however, the reverse primers used were specific at 3’end: R 5’-TCCGGGTTAGGCCACTTCAC-3’; R 5’-CGGGTTAGGCCACTTCAG-3’. The thermocycling conditions of both PCR reactions were as following: initial denaturation at 94˚C - 5 min, amplification at 94˚C - 1 min; 55˚C - 1 min; 72˚C - 1 min - 30 cycles; final elongation - 72˚C, 8 min.
The differences were estimated using Pearson’s χ2 test (р < 0.05) by checking the correspondence of genotypes distribution to the Hardy-Weinberg equilibrium. The OR (odds ratio) and 95% CI (confidence interval) were used to evaluate the association between genotypes and breast cancer susceptibility.
Fisher’s exact test was used when the values of genotypes were nonrandomly distributed in table cells (one of the meanings less than 6). Statistical calculations were performed using Microsoft Excel and Statistica 2007 software.
3. Results and Discussion
The CYP1A1 gene is located at 15 chromosome (15q22 - q24.1), contains 7 exons and spans about 6000 base pairs. We are testing the association of BC with the singlenucleotide polymorphism T/C at rs4646903 (ancestral allele is T) located in 3’-nontranslated sequence in 242 position at the end of 7th exon. Although the functional role of this polymorphism is not yet fully determined, its associations with different types of cancers were reported [7-9]. Results of investigations performed in different populations showed controversial results. For example, it was shown that the genotype of clinical significance for Chinese women is CC [10], whereas for Korean women T allele was shown to be a risk associated [11] and for the women of North America [12] the correlations of polymorphism in this site with BC were not evaluated.
At the first stage of our investigation we have determined the genotypes distribution and allele frequency among Kazakh and Russian ethic groups and compared the data with that of other ethnic groups obtained from National Center for Biotechnology Information (NCBI) database. As it can be seen from Table 1 allele frequency in Kazakhs (Asian type) and combined Asian group (CHB + JPT) are similar. In Russian group (Caucasian type) genotypes distribution and allele frequency are more close to those indices in Caucasian groups. Also, the significant differences in genotype distribution with Р = 1.74 * 10 - 6 and in allele frequency with Р = 5.70 * 10 - 7 were registered between Kazakh and Russian control groups.
The results of genotypes distribution and allele frequency analysis at rs4646903 site of gene CYP1A1 among BC patients of the Kazakh and Russian ethnic groups and controls are summarized in Table 2.
As it can be seen from the results, in the Russian group no significant differences were found in the genotypes distribution and allele frequency between cancer cases and controls. In contrast, the statistically significant differences in genotypes distribution and allele frequency between BC patients and controls were found at rs4646903 polymorphism in the Kazakh group. The odds
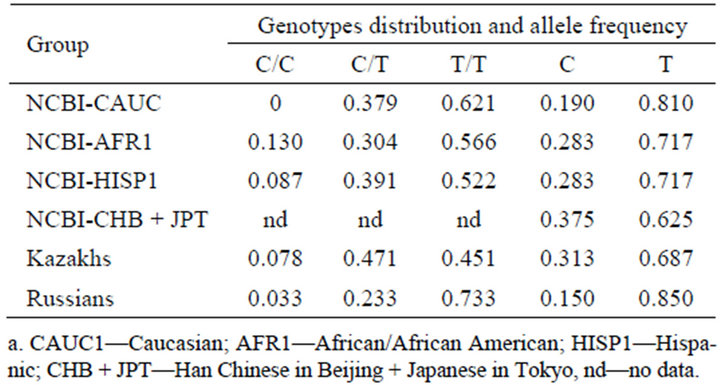
Table 1. Genotypes distribution and allele frequency at rs4646903 site of CYP1A1 gene in several ethnic groups according NCBI database and to the author’s data.

Table 2. Genotypes distribution and allele frequency at rs4646903 site of gene CYP1A1 in the Kazakh and Russian ethnic groups.
ratio (OR) for homozygotes CC in this group was equal to 2.69 (CI 95%: 1.35 - 5.36) and this value can be regarded as an indication to the association of C/T polymorphism (allele C) with breast cancer in Kazakh ethnic group.
The results presented for the Kazakh group are in agreement with studies describing the presence of an association with BC in the population of China [10] and of Mexico: OR = 1.95, CI = 1.13 - 3.36 [13].
In Russian group no differences were found either in genotypes distribution or in the allele frequency of polymorphism at rs4646903 site. Interestingly, the absence of association of this polymorphism with BC in Russian ethnic group shown in the current study does not coincide with the results of two similar investigations performed in Russians inhabiting Novosibirsk, Russia, where significant differences in allele and genotype frequencies at rs4646903 site of gene CYP1A1 between patients and controls were described.
We had compared the allele and genotype T-C-TTTC-CC frequency for the investigated groups of Russians in Kazakhstan and Russia: 0.850 - 0.150 - 0.733 - 0.233 - 0.033—presented results; 0.896 - 0.104 - 0.796 - 0.201 - 0.003—Novosibirsk [14]; 0.888 - 0.112 - 0.775 - 0.225 - 0.000—Novosibirsk, [3].
These data show the presence of significant differences in genotype distribution between ethnic Russians, inhabiting Novosibirsk, Russia and Kazakhstan (χ2 = 3.874, Р = 0.049).
Earlier we have received the similar data describing the specificities of genotypes of ethnical Russians inhabiting Kazakhstan [15].
The CYP1B1 gene is located at 2 chromosome (2р22 - р21), contains 3 exons and 2 introns, and spans about 7 800 base pairs.
We have analyzed the association of BC with the single-nucleotide polymorphism C/G (rs1056836), located at 1666 position in 3rd exon, leading to leucine/valine aminoacid substitution at 432 codon (Leu/Val). This substitution (Val432 allele, G) results in three fold increase of 4-hydroxylase activity compared with Leu432 allele (C). The similar increase of catalytical activity was also shown for the other polymorphic variants: Gly48, Ser119, Ser453 [6].
In Table 3, the values of genotypes distribution and allele frequency in rs1056836 site of gene CYP1B1 in the main world ethnic groups according HapMap, NCBI and author’s data are presented.
Genotypes distribution and allele frequency at rs1056836 site of gene CYP1B1 in the Russian ethnic group are similar to those indices in the group Utah residents with Northern and Western European ancestry (CEU).
In the Kazakh group the values of genotypes distribution and allele frequency at rs1056836 site were intermediate between those of CEU and the combined Asian group of Han Chinese in Beijing, and Japanese in Tokyo (CHB & JPT).
The analysis of this polymorphism performed in China showed the elevated risk of BC in carriers of CC genotype with OR = 2.3, 95% CI = 1.2 - 4.5 in Ningxia Hui region [16] and OR = 2.8, 95% CI = 1.04 - 7.51 in Shanghai [17]. The positive associations of this polymorphism with BC were also found in Turkey [18] and Nigeria [19].
The genotypes distribution and allele frequency at rs1056836 site of CYP1B1 gene in the Kazakh and Russian ethnic groups of BC patients and controls are summarized in Table 4.
In the Russian group the statistically significant difference (χ2 = 7.48, P = 0.02) in genotype distribution was differenct in genotypes distribution and allele frequency type. This result differs from the results of other Cauca sian populations [2,20].
Positive association of polymorphism at rs1056836 of
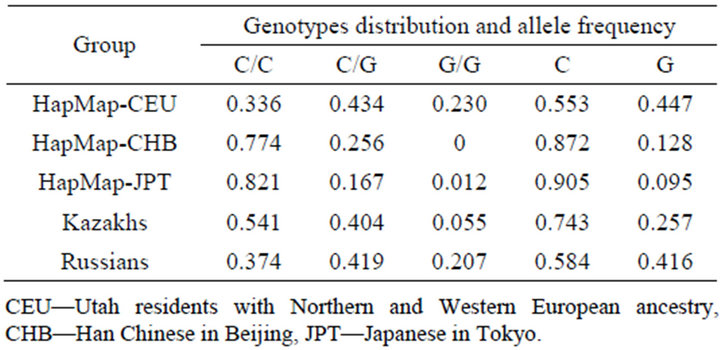
Table 3. Genotypes distribution and allele frequency in rs1056836 site of gene CYP1B1 in some world ethnic groups according to NCBI and author’s data.
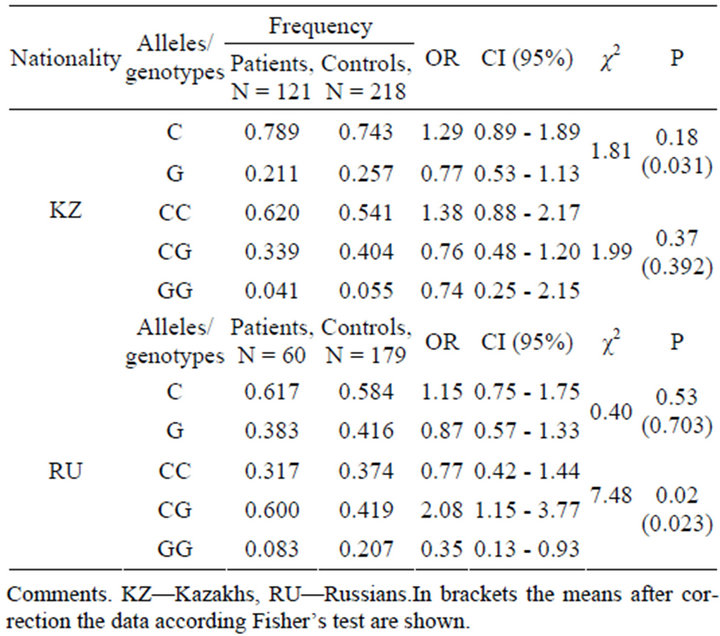
Table 4. Genotypes distribution and allele frequency at 432 codon of CYP1В1 (rs1056836) in the Kazakh and Russian ethnic groups.
CYP1В1 gene with BC was found in the population of Slovenia but only in combination with polymorphism in COMT gene (OR, 2.0; 95% CI, 1.1 - 3.5) [21], and of USA (OR = 1.2; 95% CI 0.9 - 1.6) [22] for individuals homozygous by minor allele.
In the Kazakh ethnic group no associations of genotypes distribution and allele frequency at rs1056836 site of CYP1B1 gene with BC were found. These findings are in agreement with the results of similar studies performed in Asian cohorts [23,24] and with metaand pooled analysis of associations of cytochrome P450 1B1 genes polymorphism and breast cancer [1].
REFERENCES
- V. Paracchini, S. Raimondi, I. T. Gram, D. Kang, N. A. Kocabas, V. N. Kristensen, D. Li, F. F. Parl, T. Rylander-Rudqvist, P. Soucek, W. Zheng, S. Wedren and E. Taioli, “Metaand Pooled Analyses of the Cytochrome P-450 1B1 Val432Leu Polymorphism and Breast Cancer: A HuGE-GSEC Review,” American Journal of Epidemiology, Vol. 165, No. 2, 2007, pp. 115-125. doi:10.1093/aje/kwj365
- K. W. Reding, C. Chen, K. Lowe, D. R. Doody, C. S. Carlson, C. T. Chen, J. Houck, L. K. Weiss, P. A. Marchbanks, L. Bernstein, R. Spirtas, J. A. McDonald, B. L. Strom, R. T. Burkman, M. S. Simon, J. M. Liff, J. R. Daling and K. E. Malone, “Estrogen-Related Genes and Their Contribution to Racial Differences in Breast Cancer Risk,” Cancer Causes Control, Vol. 23, No. 5, 2012, pp. 671-681. doi:10.1007/s10552-012-9925-x
- L. F. Gulyaeva, O. N. Mikhailova, V. O. Pustylnyak, I. V. Kim, A. V. Gerasimov, S. E. Krasilnikov, M. L. Filipenko and E. V. Pechkovsky, “Comparative Analysis of SNP in Estrogen-Metabolizing Enzymes for Ovarian, Endometrial, and Breast Cancers in Novosibirsk, Russia,” Advanced Experimental and Medical Biology, Vol. 617, 2008, pp. 359-366. doi:10.1007/978-0-387-69080-3_34
- G. F. Korytina, L. Z. Akhmadishina, O. V. Kochetova, Sh. Z. Zagidullin and T. V. Viktorova, “Association of Cytochrome P450 Genes Polymorphisms (CYP1A1 and CYP1A2) with the Development of Chronic Obstructive Pulmonary Disease in Bashkortostan,” Molecular Biology, Vol. 42, No. 1, 2008, pp. 32-41.
- M. T. Wu, J. M. Lee, D. C. Wu, C. K. Ho, Y. T. Wang, Y. C. Lee, H. K. Hsu and E. L. Kao, “Genetic Polymorphisms of Cytochrome P4501A1 and Oesophageal SquamousCell Carcinoma in Taiwan,” British Journal of Cancer, Vol. 87, No. 5, 2002, pp. 529-532. doi:10.1038/sj.bjc.6600499
- M. Sasaki, Y. Tanaka, M. Kaneuchi, N. Sakuragi and R. Dahiya, “CYP1B1 Gene Polymorphisms Have Higher Risk for Endometrial Cancer, and Positive Correlations with Estrogen Receptor Alpha and Estrogen Receptor Beta Expressions,” Cancer Research, Vol. 63, No. 14, 2003, pp. 3913-3918.
- C. Kiyohara, T. Horiuchi, K. Takayama and Y. Nakanishi, “Genetic Polymorphisms Involved in Carcinogen Metabolism and DNA Repair and Lung Cancer Risk in a Japanese Population,” Journal of Thoracic Oncology, Vol. 7, No. 6, 2012, pp. 954-962. doi:10.1097/JTO.0b013e31824de30f
- D. Wang, M. Su, D. Tian and S. Liang, “Associations between CYP1A1 and CYP2E1 Polymorphisms and Susceptibility to Esophageal Cancer in Chaoshan and Taihang Areas of China,” Cancer Epidemiology, Vol. 36, No. 3, 2012, pp. 276-282. doi:10.1016/j.canep.2011.10.008
- T. N. Sergentanis, K. P. Economopoulos, S. Choussein and N. F. Vlahos, “Cytochrome P450 1A1 (CYP1A1) Gene Polymorphisms and Cervical Cancer Risk: A Meta-Analysis,” Molecular Biology Reports, Vol. 39, No. 6, 2012, pp. 6647-5654. doi:10.1007/s11033-012-1470-x
- J. R. Long, Q. Cai, X. O. Shu, H. Cai, Y. T. Gao and W. Zheng, “Genetic Polymorphisms in Estrogen-Metabolizing Genes and Breast Cancer Survival,” Pharmacogenetical Genomics, Vol. 17, No. 5, 2007, pp. 331-338. doi:10.1097/FPC.0b013e32801a3bfe
- A. Shin, D. Kang, J. Y. Choi, K. M. Lee, S. K. Park, D. Y. Noh, S. H. Ahn and K. Y. Yoo, “Cytochrome P450 1A1 (CYP1A1) Polymorphisms and Breast Cancer Risk in Korean Women,” Experimental & Molecular Medicine, Vol. 39, No. 3, 2007, pp. 361-366. doi:10.1038/emm.2007.40
- B. Diergaarde, J. D. Potter, E. R. Jupe, S. Manjeshwar, C. D. Shimasaki, T. W. Pugh, D. C. Defreese, B. A. Gramling, I. Evans and E. White, “Polymorphisms in Genes Involved in Sex Hormone Metabolism, Estrogen plus Progestin Hormone Therapy Use, and Risk of Postmenopausal Breast Cancer,” Cancer Epidemiology, Biomarkers and Prevention, Vol. 17, No. 7, 2008, pp. 1751-1759. doi:10.1158/1055-9965.EPI-08-0168
- M. Moreno-Galván, N. E. Herrera-González, V. RoblesPérez, J. C. Velasco-Rodríguez, R. Tapia-Conyer and E. Sarti, “Impact of CYP1A1 and COMT Genotypes on Breast Cancer Risk in Mexican Women: A Pilot Study,” International Journal of Biological Markers, Vol. 25, No. 3, 2010, pp. 157-163.
- E. P. Khvostova, V. O. Pustylnyak and L. F. Gulyaeva, “Genetic Polymorphism of Estrogen Metabolizing Enzymes in Siberian Women with Breast Cancer,” Genetic Testing and Molecular Biomarkers, Vol. 16, No. 3, 2012, pp. 167-173. doi:10.1089/gtmb.2011.0131
- N. A. Ajtkhozhina, V. G. Nigmatova, A. K. Khanseitova, M. A. Mendesh, E. E. Ashirbekov and T. S. Balmukhanov, “Polymorphic Markers of Some Genes Associated with Multiple Sclerosis in the Population of Kazakhstan,” Genetika, Vol. 47, No. 6, 2011, pp. 847-852.
- H. Jiao, C. Liu, W. Guo, L. Peng, Y. Chen and F. L. Martin, “Association of CYP1B1 Polymorphisms with Breast Cancer: A Case-Control Study in the Han Population in Ningxia Hui Autonomous Region, P. R. China,” Biomark Insights, Vol. 12, 2010, pp. 21-27.
- W. Zheng, D. W. Xie, F. Jin, J. R. Cheng, Q. Dai, W. Q. Wen, X. O. Shu and Y. T. Gao, “Genetic Polymorphism of Cytochrome P450-1B1 and Risk of Breast Cancer,” Cancer Epidemiology, Biomarkers and Prevention, Vol. 9, No. 2, 2000, pp. 147-150.
- N. A. Kocabaş, S. Sardaş, S. Cholerton, A. K. Daly and A. E. Karakaya, “Cytochrome P450 CYP1B1 and Catechol OMethyltransferase (COMT) Genetic Polymorphisms and Breast Cancer Susceptibility in a Turkish Population,” Archives of Toxicology, Vol. 76, No. 11, 2002, pp. 643- 649. doi:10.1007/s00204-002-0387-x
- M. N. Okobia, C. H. Bunker, S. J. Garte, J. M. Zmuda, E. R. Ezeome, S. N. Anyanwu, E. E. Uche, U. Osime, J. Ojukwu, L. H. Kuller, R. E. Ferrell and E. Taioli, “Cytochrome P450 1B1 Val432Leu Polymorphism and Breast Cancer Risk in Nigerian Women: A Case Control Study,” Infection Agent Cancer, Vol. 10, No. 4, 2009, pp. S1-S12. doi:10.1186/1750-9378-4-S1-S12
- P. Sillanpää, L. Heikinheimo, V. Kataja, M. Eskelinen, V. M. Kosma, M. Uusitupa, H. Vainio, K. Metsola and A. Hirvonen, “CYP1A1 and CYP1B1 Genetic Polymorphisms, Smoking and Breast Cancer Risk in a Finnish Caucasian Population,” Breast Cancer Research and Treatment, Vol. 104, No. 3, 2007, pp. 287-297. doi:10.1007/s10549-006-9414-6
- J. Z. Cerne, M. Pohar-Perme, S. Novakovic, S. Frkovic-Grazio, V. Stegel and K. Gersak, “Combined Effect of CYP1B1, COMT, GSTP1, and MnSOD Genotypes and Risk of Postmenopausal Breast Cancer,” Journal of Gynecologic Oncology, Vol. 22, No. 2, 2011, pp. 110-119. doi:10.3802/jgo.2011.22.2.110
- A. J. Sigurdson, P. Bhatti, S. C. Chang, P. Rajaraman, M. M. Doody, L. Bowen, S. L. Simon, R. M. Weinstock, M. S. Linet, M. Rosenstein, M. Stovall, B. H. Alexander, D. L. Preston and J. P. Struewing, “Polymorphisms in Estrogen Biosynthesis and Metabolism-Related Genes, Ionizing Radiation Exposure, and Risk of Breast Cancer among US Radiologic Technologists,” Breast Cancer Research and Treatment, Vol. 118, No. 1, 2009, pp. 177-184. doi:10.1007/s10549-009-0307-3
- K. M. Lee, J. Abel, Y. Ko, V. Harth, W. Y. Park, J. S. Seo, K. Y. Yoo, J. Y. Choi, A. Shin, S. H. Ahn, D. Y. Noh, A. Hirvonen and D. Kang, “Genetic Polymorphisms of Cytochrome P450 19 and 1B1, Alcohol Use, and Breast Cancer Risk in Korean Women,” British Journal of Cancer, Vol. 88, No. 5, 2003, pp. 675-678. doi:10.1038/sj.bjc.6600761
- N. Shimada, M. Iwasaki, Y. Kasuga, S. Yokoyama, H. Onuma, H. Nishimura, R. Kusama, G. S. Hamada, I. N. Nishimoto, H. Iyeyasu, J. Motola Jr., F. M. Laginha, N. Kurahashi and S. Tsugane, “Genetic Polymorphisms in Estrogen Metabolism and Breast Cancer Risk in Case-Control Studies in Japanese, Japanese Brazilians and Non-Japanese Brazilians,” Human Genetics, Vol. 54, No. 4, 2009, pp. 209-215. doi:10.1038/jhg.2009.13
NOTES
*Corresponding author.

