American Journal of Climate Change
Vol.04 No.05(2015), Article ID:61693,11 pages
10.4236/ajcc.2015.45032
Effects of Mowing Heights on N2O Emission from Temperate Grasslands in Inner Mongolia, Northern China
Lihua Zhang1*, Longyu Hou2*, Hendrikus J. Laanbroek3,4, Dufa Guo5, Qibing Wang1#
1State Key Laboratory of Vegetation and Environmental Change, Institute of Botany, Chinese Academy of Sciences, Beijing, China
2Department of Grassland Science, China Agricultural University, Beijing, China
3Department of Microbial Ecology, Netherlands Institute of Ecology (NIOO-KNAW), Wageningen, The Netherlands
4Institute of Environmental Biology, Utrecht University, Utrecht, The Netherlands
5Shandong Normal University, Jinan, China

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 10 September 2015; accepted 1 December 2015; published 4 December 2015

ABSTRACT
Grazing and mowing are two common practices for grassland management. Mowing is now recommended as an alternative to the traditional grazing for grassland conservation in Inner Mongolia, northern China. Many studies have revealed that mowing may alter ecosystem properties in various ways. However, little attention has been paid to the effect of mowing on trace gas emissions, especially on N2O flux. We conducted an experiment to investigate the effects of mowing on N2O fluxes from the semiarid grassland in Inner Mongolia. The mowing experiment, which started in 2003, comprised four mowing intensity treatments, i.e. mowing heights at 2 cm, 5 cm, 10 cm and 15 cm above the soil surface, respectively, and a control (non-mowing), with five replicates. Gas fluxes were measured through a closed static chamber technique during the growing seasons (usually from May to September, depending on local climate at the time) of 2008 and 2009, respectively. Our results showed that mowing decreased N2O emissions, above-ground biomass and total litter production. N2O emissions were greater in May and June than in other sampling periods, regardless of treatments (P < 0.05). A co-relationship analysis suggested that variations in seasonal N2O fluxes were mainly driven by variations in soil moisture, except in July and August. In July and August, above-ground plant biomass and soil total nitrogen became the major drivers of N2O fluxes under the soil temperatures between 16˚C and 18˚C. Though there were some uncertainties due to the low frequency of N2O flux measurement, our study mainly indicated that 5 cm mowing height might decrease N2O emissions in grasslands during the growing season, and soil properties affected the magnitude of the reduction.
Keywords:
N2O Fluxes, Mowing, Soil Moisture, Soil Microbial Carbon Biomass, Soil Organic Carbon

1. Introduction
The temperate steppe in northern China is a typical vegetation type on the Eurasian continent and is sensitive to anthropogenic disturbances and climate change [1] [2] . Mowing is one of the prevailing management practices in these areas [3] . Recently, mowing is highly recommended for sustainable grassland management in a national project called Grain for Green, which aims to restore the degraded ecosystems in western China [4] . Mowing, the removal of a part of plant shoot tissue, has negative effects on overall plant growth and carbon allocation [5] , which can influence root carbon exudation and rhizosphere organisms that rely on the carbon and nitrogen released from the plant roots [6] . The removal of some plants by mowing inevitably leads to the adjustment of the size of the root system and thus causes the death and decay of the roots and nodules, followed by decomposition, mineralization of nitrogen, nitrification and denitrification. Mowing also reduces the input of above-ground litter to the soil [7] , and consequently decreases the amount of coarse organic matter in the soil [8] and related gas emissions from soil, including nitrous oxide (N2O) [9] .
N2O is a powerful greenhouse gas, and it contributes approximately 6% to the anticipated global warming [10] . The global atmospheric N2O concentration increased from a preindustrial value of 270 to 322 ppb in 2008 [10] . N2O emitted from soils is considered as one of the major contributors to this rise [11] . Nitrification and denitrification are one of the key ecological processes that determine N2O production in an ecosystem. Related studies suggest that N2O emissions are determined by soil properties and processes including soil temperature, soil moisture, substrate availability [12] , soil diffusive characteristics, air-filled porosity [13] , the activity of nitrifying/denitrifying microbial communities [14] and concurrent N2O consumption processes in the soil [15] . The complexity of these factors, which regulate N2O production, consumption and emission, results in considerable uncertainties in estimating actual N2O exchange rates for given management scenarios.
Fore-studies showed that mowing decreased N mineralization in soil and had a significant negative impact on the growth and N uptake of plants growing in the soil of the mowed area [9] [16] . But due to the high spatial variability of N2O fluxes [17] and complexity of the drivers of N2O emission, estimates are still uncertain. Little is known about the effects of mowing on N2O emissions at present [18] . Therefore, it is necessary to quantify the changes in N2O emission caused by mowing to fully understand the regional budget of trace gases.
To quantify N2O fluxes in response to different mowing intensities, we established an experiment in 2003 with different mowing heights in a steppe ecosystem in Inner Mongolia, China. Here, we present results of N2O fluxes over the two growing seasons (from May to September) of 2008 and 2009 analyzing the relation between N2O emission and abiotic and biotic factors to identify the controls of the emission. We hypothesized that: 1) mowing would decrease N2O emission due to the removal of aboveground plant biomass, which can result in continuous decrease in the availability of substrate for N2O production [19] and the related soil microbes; 2) both soil biotic and abiotic factors play important roles in underlying mowing effects on N2O flux.
2. Results
2.1. Climate
The study area received 370 mm and 185 mm of rain for 2008 and 2009, respectively, and showed great variations in temporal distribution (Figure 1). Most of the rain fell over the summer months (from June to August), which accounted for 90% of the annual total precipitation in the two years. Soil temperature and moisture varied seasonally at this site (see Ref. [20] ). Briefly, soil temperature at 5 cm depth ranged from 13.1˚C to 31.1˚C with an average of 22.3˚C in 2008, 8.5˚C to 27.9˚C with an average of 18.1˚C in 2009. And the temperature generally negatively correlated with mowing height. Soil moisture peaked in July and had similar temporal fluctuation patterns over the two growing seasons, although the amount of rainfall was substantially different during the same period.
Figure 1. Temporal variations in N2O fluxes under different mowing treatments in 2008 (left) and 2009 (right). Panels (a) and (b) represent the no-mowing control (Mck). Panels (c) and (d), panels (e) and (f), panels (g) and (h), panels (i) and (j) represent the mowing heights of 15 cm (M15), 10 cm (M10), 5 cm (M5) and 2 cm (M2) above the soil surface, respectively. Data are mean ± SE (n = 5).
2.2. Effect of Mowing on Above-Ground Biomass and Litter
Above-ground biomass and litter measured are presented in Table 1. The above-ground biomass and total litter decreased progressively as cutting height decreased from 15 cm to 2 cm, and significant lower litter mass was found for the M2 treatments (P < 0.01). A similar pattern was also observed for ANPP, which indicates that a cutting height greater than 10 cm has minimal effects on net primary productivity. These results indicate that the
Table 1. Means of total plant litter (Litter: g∙m−2), above-ground plant biomass (AB: g∙m−2), species richness (SR), soil organic carbon (SOC: %), soil total nitrogen (TN: %), soil total phosphorus (TP: %), soil C/N ratio and cumulative N2O emission/uptake (kg∙N∙ha−1) under different mowing treatments. Values represent the mean of five replicates. Different letters in a row indicate significant difference among treatments at P < 0.05. Mck represent the no-mowing control. M15, M10, M5 and M2 represent the treatments of plant mowed at heights of 15 cm, 10 cm, 5 cm and 2 cm above soil surface, respectively. Same letter in the right upper corner are not significantly different at P = 0.05, different letters mean significant difference among treatments at P < 0.05.
effects of greater cutting heights (M15 and M10) on net primary production were limited in this grassland ecosystem. In addition, the biomass of Artemisia frigida, a species with low height, contributed to most of the total biomass of M2 and M5 plots relative to that of the control, M15 and M10 plots (data not shown).
2.3. Temporal Dynamics in N2O Fluxes
Mean N2O emission rates from the different mowing treatments varied in a range of −31.7 to 67.2 μg N2O m−2∙h−1 with means of 12.7, −4.2, 4.8, −5.2 and 4.1 µg N2O m−2∙h−1, for treatments of Mck, M15, M10, M5, and M2, respectively, over the growing season of 2008, and means of 13.6, −9.0, 3.7, −2.9 and 5.5 µg N2O m−2∙h−1 for Mck, M15, M10, M5, and M2, respectively, in 2009. The variation of N2O fluxes was relatively narrow in the dry year (2009) compared to the wet year (2008) (Figure 1).
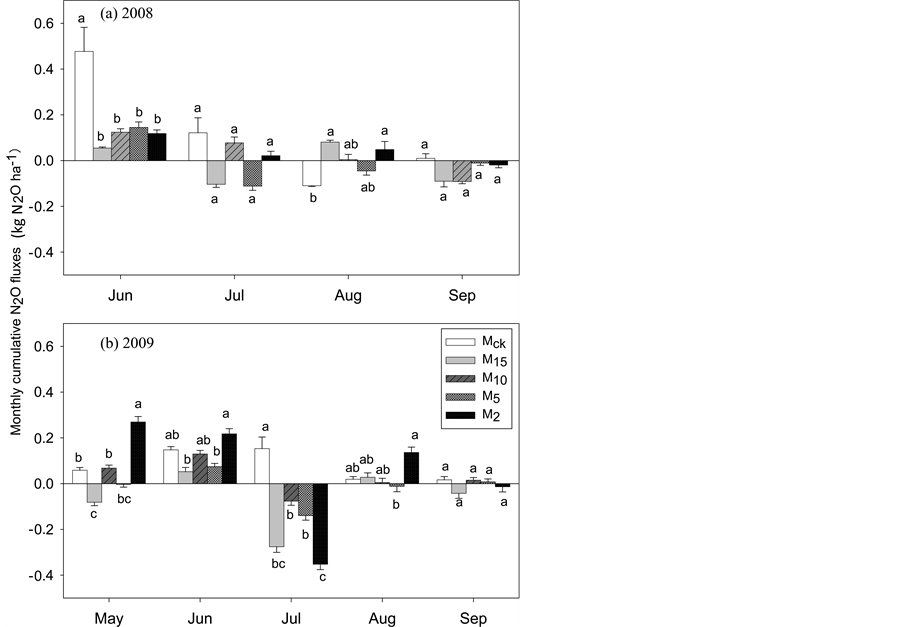
The higher variation in N2O emission rate resulted in high variation of the monthly cumulative N2O flux with a range of 0.48 to −0.35 kg N2O ha−1 over the two growing seasons (Figure 2). Measurements showed that grassland could be a N2O source (positive value)/sink (negative value) (Figure 2). Regardless of the mowing heights, the monthly cumulative N2O flux was negative in July for most of the treatments, except for the controls both in 2008 and 2009, and the M10 and M2 in 2008 (Figure 2).
2.4. Effects of Mowing on N2O Fluxes
The seasonal cumulative N2O emissions based on a linear interpolation of spatially averaged daily or monthly mean fluxes. Over the two growing seasons, an estimated amount of 0.22 kg N2O ha−1 per season was emitted in the control treatment, of which more than 88% occurred in May and June. Occasionally, negative values were observed in the control and mowing treatments (Figure 1). The estimated seasonal cumulative emissions in the different treatments ranged from −0.27 kg N2O ha−1 to 0.33 kg N2O ha−1 (Table 1). For the total fluxes over the two growing seasons, the value for M15 was the only mowing treatment significantly different from that of the control (P < 0.05) (Table 1). Compared with the control, the M15, M10, M5 and M2 mowing treatments decreased N2O emission (Table 1).
2.5. Controls of N2O Fluxes
The results of regression analyses showed that the closest relationship existed between the N2O fluxes and soil
moisture (r2 = 0.8, P < 0.0001), followed by above-ground plant biomass (r2 = 0.30, P = 0.02) in this experiment (Figure 3(a), Figure 3(b)). Our stepwise regression analyses showed that a combination of soil moisture (partial R2 = 0.42, P = 0.0005), soil temperature (partial R2 = 0.13, P = 0.02) and total phosphorus (partial R2 = 0.08, P = 0.047) explained 62.5% of the variation in the N2O fluxes (P < 0.005) in May. Microbial biomass nitrogen explained 21.2% of the variation of the N2O fluxes (P = 0.02) in June, and soil moisture explained 21.4% of the variation (P = 0.02) in July. Other factors did not contribute significantly to the explanation of the variation in N2O fluxes in these summer months. In addition, we did not find a significant correlation between N2O fluxes and any other variables measured at P < 0.05 in August and September.
Figure 2. Monthly cumulative N2O fluxes in 2008 (a) and 2009 (b) under different mowing heights. Values are mean ± SE (n = 5). Different letters represent statistically significant differences among treatments in the same month at P < 0.05.
Figure 3. Relationships between monthly N2O fluxes and soil moisture (V/V%, (a)), above-ground plant biomass (g∙m−2, (b)), soil total nitrogen (g∙kg−1, (c)). Solid circles in (c) only plotted from the data obtained at soil temperatures between 16˚C and 18˚C.
Using the pooled data of the different treatments in the whole growing season, multi-linear regression analyses showed that N2O fluxes was correlated with soil temperature, moisture (ST, SM) and soil organic carbon (SOC) in the control treatment and correlated with ST and SM in the M5 treatment, with SOC in the M2 treatment (Table 2). Combined ST, SM and SOC can explaining 31% of the variation in the N2O fluxes in the control treatment and combined ST and SM can explaining 27% in M5 treatment. Across the 25 sites all of the treatments, combined ST, SM and SOC can explaining 12% of the variation in N2O in spatial (Table 2).
3. Discussion
3.1. Controls of Changes in N2O Fluxes
Our observations, showing a non-linear decrease in N2O emission with decreases in mowing height, generally supported the hypothesis that a long term mowing treatment would decrease grassland N2O emissions to the atmosphere. Our observations are in line with the results of another similar investigation also conducted in a grazed grassland in Inner Mongolia [21] , but opposite to the findings in a heavily grazed alpine grassland by Gao [22] . Mowing not only led to lower N2O emission rates, but also led to lower above-ground plant and litter biomass (Table 1), and most likely to a removal of nitrogen from the system. Nitrification is commonly repressed by nitrogen limitation, which explains the decline in N2O with mowing height. Indeed we observed a linear relation between N2O fluxes and total soil nitrogen though this relationship was not robust because of low measurement frequency and was only found for soil temperatures ranging from 16˚C to 18˚C. This was in line with findings that N mineralization and nitrification were repressed during the growing season but increased after that period [23] .
In July of 2008 and 2009, a number of weak N2O uptake peaks occurred in some of the mowing treatments (Figure 2(a), Figure 2(b)), leading to significant uptake of N2O on monthly basis (Figure 2(b)). Though temperature was favorable in July, microbial activity may have decreased due to the low precipitation. Additionally, plants strongly compete for the mineralized N, leaving less N for nitrifying and denitrifying bacteria [8] [24] . This competition may be the reason for the N2O uptake observed in grasslands during July in our study. This result confirms that grassland soils may act as net sinks of atmospheric N2O under certain conditions due to high N2O reduction activity by denitrifying microorganisms [16] [25] . N2O reduction by denitrifying microorganism is associated with reduced levels of oxygen, which is usually related to increased moisture contents [26] . Despite the different patterns of N2O fluxes in July 2008 and 2009, soil moisture contents were not significantly different between these months [20] . Hence, unknown soil factors might have been responsible for the observed differences in the fluxes in July.
In August, however, there was a net N2O emission except for the control in 2008 and M5 in both years (Figure 2(b)). At this time of the year, the temperature is still high enough for soil microbial activity (data not shown). Therefore soil microbes will decompose plant material, resulting in increased mineralization of N. However, most of the plants are getting senescent and require fewer nutrients from soil for growth at that time, consequently the competition between plants and microorganisms for mineralized N will be reduced. The higher N availability likely promotes microbial nitrification leading to a high N2O production.
Using the pooled data of the whole growing season and all treatments, stepwise regression analyses showed
Table 2. Relationships between N2O flux and soil temperature (ST), soil moisture (SM), soil organic carbon (SOC), and the combined effects of ST/SM/SOC in the control and mowing treatments. The value in the table is the R2.
One asterisk, P < 0.1; two asterisks, P < 0.05; three asterisks, P < 0.01; ST-SM refers to soil temperature-soil moisture; ST-SM-SOC refers to soil temperature-soil moisture-soil organic carbon.
that soil moisture was the most important controlling factor for N2O fluxes, explaining 80% of the observed variations in seasonal average N2O fluxes (P < 0.0001) at our study site (Figure 3(a)). Similar results were reported for grasslands in Ireland [27] and New Zealand [28] . Therefore, our results confirm soil moisture is a major control of seasonal N2O fluxes and determines microbial N2O emissions in the semi-arid grassland of Inner Mongolia, China. In some agricultural regions, soil moisture was also found to be the controlling factor of N2O fluxes [29] . Because soil moisture is the key determinant of the microbial processes that consume or produce N2O, soil moisture shifts in arid and semiarid regions will likely affect N2O fluxes.
3.2. Effect of Mowing on N2O Fluxes
The relationship between N availability and N2O flux is commonly examined for predicting N2O fluxes on large scales, and Millar [30] reported that N availability is the only factor affecting N2O fluxes at meter-scales. However, in our study, we find the significant positive relationships between total nitrogen and N2O fluxes only in a narrow temperature range (Figure 3(c)).
Our other hypothesis was that both soil biotic and abiotic factors underlying mowing effects of decreasing plant biomass play important roles in regulating N2O fluxes. Since the cumulative seasonal N2O fluxes were only slightly different among different mowing treatments, and no consistent changes were found among all of the mowing treatments (Table 1, Table 3), we tested the effects of mowing on N2O fluxes by comparing the pooled data of all the mowing treatments including that of the control. The result suggests that mowing might decrease N2O emissions by reducing plant litter and above-ground biomass (Table 3), since the decreased plant litter and biomass might result in decreases in the substrates supplied to microbes, which are involved in nitrification and denitrification processes. The above of our finding agrees with that of some previous studies. Zou [31] established a positive linear relationship between above-ground plant biomass and N2O emissions. Kammann [32] found that increasing numbers of cuts reduced N2O emissions. However, in other cases, for instance, Beck and Christensen [33] found that N2O emissions increased when all above-ground grass was removed, and Klumpp [34] only observed some small peaks of N2O emission in response to cutting events. There was a significant effect of the differences in soil moisture on the N2O fluxes after mowing (Figure 3(a)), which explains 80% of the variation in the N2O emissions induced by mowing (P < 0.001). Similar results were found in a study performed in grazed grasslands by Wolf [21] .
4. Material and Methods
4.1. Study Site
This study was carried out at the Duolun Restoration Ecology Research Station (116˚17'E, 42˚02'N), Institute of Botany, Chinese Academy of Sciences (IBCAS), which is located at Duolun County in Inner Mongolia, China. The area is situated in a semiarid, middle temperate zone and characterized by a continental monsoon climate. The mean annual air temperature is around 2.1˚C, with monthly mean temperatures ranging from −17.5˚C in January to 18.9˚C in July. The mean annual precipitation is approximately 385 mm, with 80% precipitation occurring from mid-June to late September. The topography is featured by low foothills at elevations of 1150 - 1800 m. The soil at the study site is classified as Chestnut soil in the Chinese soil classification, which equals to Calcic Kastanozem in IUSS system. And it contains 62.75% ± 0.04% sand, 20.30% ± 0.01% silt and 16.95% ± 0.01% clay, with mean soil (0 - 10 cm) bulk density of 1.31 g∙cm−3 and pH value of 7.12. The dominant plant species in the temperate grassland are Stipa krylovii Roshev., Cleistogenes squarrosa (Trin.) Keng., Artemisia frigida Willda, Potentilla acaulis L., Allium bidentatum Fisch. Ex Prokh. and Agropyron cristatum (L.) Gaertn.
Table 3. Results (F-values) of repeated measures ANOVAs for the effects of year (Y), mowing (M), and their interactions on the legume biomass, species number, above-ground biomass, litter, total coverage and instantaneous N2O flux (μg∙m−2∙h−1). One asterisk, P < 0.05; two asterisks, P < 0.01; three asterisks, P < 0.001.
4.2. Experimental Design
The mowing experiment was set up in 2003 and consists one control (non-mowing, Mck) and four mowing height treatments, i.e., mowing heights at 2 cm (M2), 5 cm (M5), 10 cm (M10) and 15 cm (M15) above soil surface, respectively, in a complete randomized block design, with five replicates. The five treatments were randomly assigned to the five plots (10 m by 20 m in size) in each of five blocks. Any adjacent blocks and plots within a block were 4 m apart from each other. Mowing with complete removal of the plant cuttings was carried out in late August each year starting in 2003. The precipitation data from 2008 to 2009 were provided by a meteorological station in an open field, which is approximately 2 km away from the experimental site and run by the Duolun Restoration Ecology Research Station, IBCAS. Prior to the setup of the experiment, the site had been kept free from disturbance and large animal grazing by fencing since 2001.
4.3. N2O Flux Measurements
N2O fluxes were measured by a static chamber technique following Zhang [35] -[37] . Briefly, the chamber consists of a stainless steel permanent base (50 × 50 × 12 cm) and a stainless steel top (50 × 50 × 50 cm). The base, with a 3-cm-deep groove on the upper side for water sealing, was driven into soil down to 12 cm each year approximately a month before the measurement started in each plot [38] . The chamber top was covered by heat-isolating and light-impenetrable materials outside and equipped with one rubber septum for gas sampling and two electric fans inside for mixing the air in the chamber headspace continuously and thoroughly [39] . During the measurements, the top was installed on the base, with its downside placed in the groove. The grooves were filled with water to seal the chamber. Gas samples were collected at 10-min intervals for 30 min (i.e., 0, 10, 20 and 30 min) through the septa using 60-ml syringes with airtight stopcocks. All gas samples were brought to a laboratory at the research station for N2O analyses within 12 h after sampling. N2O concentration was analyzed using a gas chromatograph (HP 5860, Agilent Technologies). N2O flux was calculated from the linear slope of the mixing ratio changes in the four samples taken at 0, 10, 20 and 30 min after the chamber was closed. Negative flux values indicate gas uptake from the atmosphere, and positive flux values indicate gas emissions to the atmosphere (A threshold value was used for selecting “good” from “bad” measurements, e.g., r2 > 0.90). The detection limit for our approach was 3 μg∙N∙m−2∙h−1 for N2O based on the chamber dimensions, the sampling time, and the reproducibility of repeated measurements of the N2O standard gases (0.6%). The N2O flux was measured weekly from June to September in 2008 and once every two weeks from May to September in 2009 on 9:00 am-12:00 am, local time. Concurrent with the N2O flux sampling, air temperature, soil temperature (ST) and soil moisture (SM, v/v%) next to the chambers were measured. Air temperature was measured at 40 cm height above the soil surface, and soil temperature was measured at 5 cm depth using a portable digital thermometer. Soil moisture was measured using a portable soil moisture measuring kit ML2x (ThetaKit, Delta-T Devices, Cambridge, UK).
4.4. Measurements of Vegetation and Soil
Prior to experiments in 2008 and 2009, vegetation variables were measured once at peak biomass (i.e., 10-20 August). A 1 × 1 m frame with a 10 cm × 10 cm grid was put above the canopy adjacent to the flux chambers in each 10 m × 20 m plot. The soil cover of each species was visually estimated following Yang [40] in all of the 100 grid cells, and then summed as the total coverage for the quadrat. After the measurement of the coverage, all plants were clipped at ground level within the quadrat of 1 m2 and separated into living vegetation as above ground biomass (ANPP; see also reference 20) and dead plant material as standing litter. The total litter was the sum of the standing litter and litter collected from the surface within the quadrat. All plant material sampled was oven-dried at 65˚C for 48 hours and weighed.
Soil samples (0 - 10 cm layer) were collected monthly during the growing season 2009 using a soil corer (5 cm diameter) at the time of the fourth gas sampling. At each soil sampling, three soil cores were taken randomly at each plot and mixed evenly. These samples were separated into two sets of sub-samples: one set was stored at 4˚C for microbial analysis, and the other was air-dried for soil organic carbon (SOC), total nitrogen (TN) and total phosphorus (TP) analyses (for details see Zhang et al., 2012).
4.5. Statistical Analysis
A repeated measures analysis of variance (ANOVA) was performed to examine inter-annual variability in SOC, TN and TP with the pooled data of all treatments during the growing season. Between-subject effects were evaluated as mowing effects and within-subject effects were interpreted as time-of-season effects. Regression analyses were made between N2O fluxes and the measured variables (e.g., ST, SM, SOC, TN, TP, MBC, MBN and ANPP, total litter, total coverage). Because some parameters were measured at different times, we used the means of the whole growing season. Therefore, every variable had five treatments with five replicates. Differences in seasonal cumulative N2O flux, total litter and ANPP among treatments were determined by simple one- way ANOVA. To examine which variable had the strongest effect on N2O fluxes, a stepwise multiple regression analysis was applied between the mean N2O fluxes over the whole growing season (as independent variable) and the measured variables (as dependent variables). All statistical analyses were conducted with the SAS 8.0 software package (SAS Institute Inc., Cary, NC, USA).
Acknowledgements
We thank Shuxin Xu, Shihuan Song, Xin Li and Guangquan Wang for their help in setting up the field facilities and experimental measuring. This study was supported by the National Natural Science Foundation of China (41371111, 40801037), the National Basic Research Program of China (973 program) (2010CB951300, 2011CB403200), the Key Projects of the Knowledge Initiative Program of the Chinese Academy of Sciences (KZCX2-YW-JC404) and the grants from the State Key Laboratory of Vegetation and Environmental Change, Institute of Botany, Chinese Academy of Sciences.
Author Contributions Statement
Conceived and designed the experiments: LZ. Performed the experiments: LZ, QW, DG. Analyzed the data: LZ, QW, HJL. Contributed reagents/materials/analysis tools: LZ, QW. Wrote the paper: LZ, QW, HJL. All authors reviewed the manuscript.
Competing Financial Interests
The authors declare no competing financial interests.
Cite this paper
LihuaZhang,LongyuHou,Hendrikus J.Laanbroek,11,DufaGuo,QibingWang, (2015) Effects of Mowing Heights on N2O Emission from Temperate Grasslands in Inner Mongolia, Northern China. American Journal of Climate Change,04,397-407. doi: 10.4236/ajcc.2015.45032
References
- 1. Christensen, L., Coughenour, M.B. and Chen, Z.Z. (2004) Vulnerability of the Asian Typical Steppe to Grazing and Climate Change. Climate Change, 63, 351-368.
http://dx.doi.org/10.1023/B:CLIM.0000018513.60904.fe - 2. Niu, S., Wu, M. and Wan, S. (2009) Nitrogen Effects on Net Ecosystem Carbon Exchange in a Temperate Steppe. Global Change Biology, 16, 144-155.
http://dx.doi.org/10.1111/j.1365-2486.2009.01894.x - 3. Tix, D. and Charvat, I. (2005) Aboveground Biomass Removal by Burning and Raking Increases Productivity in Tallgrass Prairie. Restoration Ecology, 13, 20-28.
http://dx.doi.org/10.1111/j.1526-100X.2005.00004.x - 4. Liu, X.Y., Liang, T.G., Long, R.J. and Guo, Z.G. (2009) Classification Management Mechanisms for Grassland Resources and Sustainable Development Strategies in Northern China. Acta Ecologica Sinica, 29, 5851-5859.
- 5. Ferraro, D.O. and Oesterheld, M. (2002) Effect of Defoliation on Grass Growth. A Quantitative Review. Oikos, 98, 125-133.
http://dx.doi.org/10.1034/j.1600-0706.2002.980113.x - 6. Hamilton III, E.W., Frank, D.A., Hinchey, P.M. and Murray, T.R. (2008) Defoliation Induces Root Exudation and Triggers Positive Rhizospheric Feedbacks in a Temperate Grassland. Soil Biology & Biochemistry, 40, 2865-2873.
http://dx.doi.org/10.1016/j.soilbio.2008.08.007 - 7. Valko, O., Torok, P., Matus, G. and Tothmeresz, B. (2012) Is Regular Mowing the Most Appropriate and Cost-Effective Management Maintaining Diversity and Biomass of Target Forbs in Mountain Hay Meadows? Flora, 207, 303-309.
http://dx.doi.org/10.1016/j.flora.2012.02.003 - 8. Mikola, J., Setälä, H., Virkajärvi, P., Saarijärvi, K. and Ilmarinen, K. (2009) Defoliation and Patchy Nutrient Return Drive Grazing Effects on Plant and Soil Properties in a Dairy Cow Pasture. Ecological Monographs, 79, 221-224.
http://dx.doi.org/10.1890/08-1846.1 - 9. Flechard, C., Neftel, A., Jocher, M., Amman, C. and Fuhrer, J. (2005) Bi-Directional Soil/Atmosphere N2O Exchange over Two Mown Grassland Systems with Contrasting Management Practices. Global Change Biology, 11, 2114-2127.
http://dx.doi.org/10.1111/j.1365-2486.2005.01056.x - 10. IPCC (2013) Working Group I Contribution to the IPCC Fifth Assessment Report Climate Change 2013: The Physical Science Basis Summary for Policymakers.
- 11. Dorien, M.K., Dolfing, J., Wrage, N. and Van Groenigen, J.W. (2011) Nitrifier Denitrification as a Distinct and Significant Source of Nitrous Oxide from Soil. Soil Biology and Biochemistry, 43, 174-178.
http://dx.doi.org/10.1016/j.soilbio.2010.09.030 - 12. Wrage, N., Velthof, G.L., van Beusichem, M. and Oenema O. (2001) Role of Nitrifer Denitrification in the Production of Nitrous Oxide. Soil Biology and Biochemistry, 33, 1723-1732.
http://dx.doi.org/10.1016/S0038-0717(01)00096-7 - 13. Neftel, A., Blatter, A., Schmid, M., Lehmann, B. and Tarakanov, S.V. (2000) An Experimental Determination of the Scale Length of N2O in the Soil of a Grassland. Journal of Geophysical Research: Atmospheres, 105, 12095-12103.
http://dx.doi.org/10.1029/2000JD900088 - 14. Steenwerth, K. and Belina, K.M. (2008) Cover Crops and Cultivation: Impacts on Soil N Dynamics and Microbiological Function in a Mediterranean Vineyard Agroecosystem. Applied Soil Ecology, 40, 370-380.
http://dx.doi.org/10.1016/j.apsoil.2008.06.004 - 15. Cavigelli, M.A. and Robertson, G.P. (2001) Role of Denitrifier Diversity in Rates of Nitrous Oxide Consumption in a Terrestrial Ecosystem. Soil Biology and Biochemistry, 33, 297-310.
http://dx.doi.org/10.1016/S0038-0717(00)00141-3 - 16. Sørensen, L.I., Kytoviita, M.M., Olofsson, J. and Mikola, J. (2008) Soil Feedback on Plant Growth in a Sub-Arctic Grassland as a Result of Repeated Defoliation. Soil Biology and Biochemistry, 40, 2891-2897.
http://dx.doi.org/10.1016/j.soilbio.2008.08.009 - 17. Billings, S.A., Schaeffer, S.M. and Evans, R.D. (2002) Trace N Gas Losses and N Mineralization in Mojave Desert Soils Exposed to Elevated CO2. Soil Biology and Biochemistry, 34, 1777-1784.
http://dx.doi.org/10.1016/S0038-0717(02)00166-9 - 18. Blüthgena, N., Dormann, C.F., Pratic, D., Klaus, V.H., Kleinebecker, T., Hölzel, N., Alt, F., Boch, S., Gockel, S., Hemp, A., Müller, J., Nieschulze, J., Renner, S.C., Schöning, I., Schumacher, U., Socher, S.A., Wells, K., Birkhofer, K., Buscot, F., Oelmann, Y., Rothenwöhrer, C., Scherber, C., Tscharntke, T., Weiner, C.N., Fischer, M., Kalko, E.K.V. and Linsenmair, K.E. (2012) A Quantitative Index of Land-Use Intensity in Grasslands: Integrating Mowing, Grazing and Fertilization. Basic and Applied Ecology, 13, 207-220.
http://dx.doi.org/10.1016/j.baae.2012.04.001 - 19. Berliner, D. and Kioko, J. (1999) The Effect of Long-Term Mowing and Ungulate Exclusion on Grass Species Composition and Soil Nutrient Status on the Athi-Kapiti Plains, Central Kenya. African Journal of Range & Forage Science, 16, 62-70.
http://dx.doi.org/10.2989/10220119909485720 - 20. Zhang, L.H., Guo, G.F., Niu, S.L., Wang, C.H., Shao, C.L. and Li, L.L. (2012) Effects of Mowing on Methane Uptake in a Semiarid Grassland in Northern China. PLoS ONE, 7, e35952.
http://dx.doi.org/10.1371/journal.pone.0035952 - 21. Wolf, B., Zheng, X., Bruggemann, N., Chen, W. and Dannenmann, M. (2010) Grazing-Induced Reduction of Natural Nitrous Oxide Release from Continental Steppe. Nature, 464, 881-884.
http://dx.doi.org/10.1038/nature08931 - 22. Gao, Y.H., Luo, P., Wu, N., Chen, H. and Wang, G.X. (2008) Impacts of Grazing Intensity on Nitrogen Pools and Nitrogen Cycle in an Alpine Meadow on the Eastern Tibetan Plateau. Applied Ecology and Environmental Research, 6, 69-79.
http://dx.doi.org/10.15666/aeer/0603_069079 - 23. Both, G.J., Gerards, S. and Laanbroek, H.J. (1992) Temporal and Spatial Variation in the Nitrite-Oxidizing Bacterial Community of Grassland Soil. FEMS Microbiology Ecology, 101, 99-112.
http://dx.doi.org/10.1016/0168-6496(92)90051-T - 24. Verhagen, F.J.M., Hageman, P.E.J., Woldendorp, J.W. and Laanbroek, H.J. (1994) Competition for Ammonium between Nitrifying Bacteria and Plant Roots in Soil in Pots: Effects of Grazing by Flagellates and Fertilization. Soil Biology and Biochemistry, 26, 89-96.
http://dx.doi.org/10.1016/0038-0717(94)90199-6 - 25. Wrage, N., Lauf, J., del Prado, A., Pinto, M. and Pietrzak, S. (2004) Distinguishing Sources of N2O in European Grasslands by Stable Isotope Analysis. Rapid Communications in Mass Spectrometry, 18, 1201-1207.
http://dx.doi.org/10.1002/rcm.1461 - 26. Schlesinger, W.H. (2013) An Estimate of the Global Sink for Nitrous Oxide in Soils. Global Change Biology, 19, 2929-2931.
http://dx.doi.org/10.1111/gcb.12239 - 27. Leahy, P., Kiely, G. and Scanlon, T.M. (2004) Managed Grasslands: A Greenhouse Gas Sink or Source? Geophysical Research Letters, 31, L20507.
http://dx.doi.org/10.1029/2004GL021161 - 28. Müller, C. and Sherlock, R.R. (2004) Nitrous Oxide Emissions from Temperate Grassland Ecosystems in the Northern and Southern Hemispheres. Global Biogeochemical Cycles, 18, GB1045.
http://dx.doi.org/10.1029/2003gb002175 - 29. Izaurrade, R.C., Lemke, R.L., Goddard, T.W., McConkey, B. and Zhang, Z. (2004) Nitrous Oxide Emissions from Agricultural Topo Sequences in Alberta and Saskatchewan. Soil Science Society of America Journal, 68, 1285-1294.
http://dx.doi.org/10.2136/sssaj2004.1285 - 30. Millar, N., Robertson, G.P., Grace, P.R., Gehl, R.J. and Hoben, J.P. (2010) Nitrogen Fertilizer Management for Nitrous Oxide (N2O) Mitigation in Intensive Corn (Maize) Production: An Emissions Reduction Protocol for US Midwest Agriculture. Mitigation and Adaptation Strategies for Global Change, 15, 185-204.
http://dx.doi.org/10.1007/s11027-010-9212-7 - 31. Zou, J., Huang, Y., Sun, W., Zheng, X. and Wang, Y. (2005) Contribution of Plants to N2O Emissions in Soil-Winter Wheat Ecosystem: Pot and Field Experiments. Plant and Soil, 269, 205-211.
http://dx.doi.org/10.1007/s11104-004-0484-0 - 32. Kammann, C., Grunhage, L., Muller, C., Jacobi, S. and Jager, H.J. (1998) Seasonal Variability and Mitigation Options for N2O Emissions from Differently Managed Grasslands. Environmental Pollution, 102, 179-186.
http://dx.doi.org/10.1016/S0269-7491(98)80031-6 - 33. Beck, H. and Christensen, S. (1987) The Effect of Grass Maturing and Root Decay on N2O Production in Soil. Plant and Soil, 103, 269-273.
http://dx.doi.org/10.1007/BF02370399 - 34. Klumpp, K., Bloor, J.M.G., Ambus, P. and Soussana, J.F. (2011) Effects of Clover Density on N2O Emissions and Plant-Soil N Transfers in a Fertilized Upland Pasture. Plant and Soil, 343, 97-107.
http://dx.doi.org/10.1007/s11104-010-0526-8 - 35. Sullivan, B.W., Dore, S., Kolb, T.E., Hart, S.C. and Montes-Helu, M.C. (2010) Evaluation of Methods for Estimating Soil CO2 Efflux across a Gradient of Forest Disturbance. Global Change Biology, 16, 2449-2460.
- 36. Parkin, T.B. (2008) Effect of Sampling Frequency on Estimates of Cumulative Nitrous Oxide Emissions. Journal of Environmental Quality, 37, 1390-1395.
http://dx.doi.org/10.2134/jeq2007.0333 - 37. Zhang, L.H., Song, C.C. and Wang, D.X. (2007) Effects of Exogenous Nitrogen on Freshwater Marsh Plant Growth and N2O Fluxes in Sanjiang Plain, Northeast China. Atmospheric Environment, 41, 1080-1090.
http://dx.doi.org/10.1016/j.atmosenv.2006.09.029 - 38. Song, C., Xu, X., Tian, H. and Wang, Y. (2009) Ecosystem-Atmosphere Exchange of CH4 and N2O and Ecosystem Respiration in Wetlands in the Sanjiang Plain, Northeastern China. Global Change Biology, 15, 692-705.
http://dx.doi.org/10.1111/j.1365-2486.2008.01821.x - 39. Wang, Y.S. and Wang, Y.H. (2003) Quick Measurement of CH4, CO2 and N2O Emissions from Short-Plant Ecosystems. Advances in Atmospheric Sciences, 20, 842-844.
http://dx.doi.org/10.1007/BF02915410 - 40. Yang, H., Wu, M., Liu, W., Zhang, Z. and Zhang, N. (2011) Community Structure and Composition in Response to Climate Change in a Temperate Steppe. Global Change Biology, 17, 452-465.
http://dx.doi.org/10.1111/j.1365-2486.2010.02253.x
NOTES
*The first common authors.
#Corresponding author.