CellBio
Vol.2 No.3(2013), Article ID:36988,8 pages DOI:10.4236/cellbio.2013.23011
Therapeutic Efficacy of Genistein-Topotecan Combination Compared to Vitamin D3-Topotecan Combination in LNCaP Prostate Cancer Cells
Department of Biological Sciences, College of Sciences, Florida Atlantic University at Davie, Davie, USA
Email: bsandov1@fau.edu
Copyright © 2013 Shreyasee Chakraborty et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received November 17, 2012; revised December 16, 2012; accepted December 30, 2012
Keywords: Topotecan; Genistein; Vitamin D; Prostate Cancer
ABSTRACT
Background: Prostate cancer is the most common cancer in men over the age of 60 in Western countries. An estimated 241,740 new cases of prostate cancer have been diagnosed in the United States in 2012 with a death toll of 28,170. Varieties of natural phytochemicals such as genistein and topotecan have shown potential chemotherapeutic capacities and are being used to inhibit the growth and proliferation of cell in prostate cancer. Purpose of Study: In this study, we aim to determine the efficacy of Vitamin D3-Topotecan combination compared to Genistein-Topotecan in apoptosis induction in LNCaP prostate cancer cells. Methods: LNCaP cells were grown in complete RPMI medium and cultured at 37˚C, 5% CO2 for 23 - 48 hrs to achieve 70% - 80% confluence. The cells were then treated with Genistein-Topotecan, Vitamin D3-Topotecan combination and TPT alone for 24 - 48 hours. In addition, post-treatment assayed using: Trypan Blue exclusion and MTT for cell viability, Ethidium bromide/Acridine orange to determine apoptosis induction, Rhodamine 123/Ethidium bromide to differentiate between viable, apoptotic, and necrotic cells, as well as to assess possible apoptotic mechanism, and DNA fragmentation to discriminate between apoptotic and necrotic cell death. Results: The overall data indicated the dose-and time-dependent cell death in the LNCaP cells and apoptosis as the major mechanism of treatment-induced cell growth arrest. Conclusion: The Genistein-Topotecan combination treatment was significantly more efficacious in growth inhibition of LNCaP cells compared to Vitamin D3-Topotecan or Topotecan alone.
1. Introduction
In 2012 about 241,740 new cases of prostate cancer were diagnosed making this type of cancer the second most common type of male cancer in America, preceded only by skin cancer. Furthermore, nearly 28,170 died this year from this condition [1]. American men have a 1 in 6 chance of developing prostate cancer during their lifetime. Prostate cancer occurs in older men usually over the age of 60. The risk factors for prostate cancer include: age, ethnicity, family history, diet, and obesity. A variety of natural dietary phytochemicals such as Genistein in soybean, have been proven to have chemotherapeutic capabilities. Genistein isoflavone (4’5’7-trihydroxyisoflavone) is the major dietary flavonoid found in soy (derived from soybean). Soy naturally contains genistin: beta-glucoside, which is broken down in the gastrointestinal tract, into genistein through fermentation by microbes [2,3]. Exposure of malignant cells to genistein has shown genistein’s ability to inhibit cell growth and proliferation. Potential mechanisms of action have been identified in various studies and include: alteration of signal transduction pathways, caspase protease activation and regulation of the cell cycle [4-7]. In addition genistein inhibits topoisomerase II enzyme, and angiogenesis through the blockage of VEGF signaling. Genistein has structural similarity to estrogen [7-9]. Genistein-induced apoptosis in carcinoma cells has been shown to be due to genistein’s ability to control expression of apoptosisrelated genes, such as up-regulation of Bax, and utilization of an independent p53 pathway. Genistein also induces apoptosis via other signal pathways including: increase in caspase-3 protease activity, initiation of DNA damage and halting of the cell cycle at the G2/M phase [5-10]. Studies have proven its low cytotoxicity against other chemo and radiation therapy.
Another micronutrient of potential prophylactic/therapeutic significance is Vitamin D3. Vitamin D3 also known as the “Sunshine Vitamin” is a group of fat-soluble secosteroids. It can be ingested as cholecalciferol (Vitamin D3) or ergocalciferol (Vitamin D2) and the human body can also synthesize it when sun exposure is adequate. The active form of vitamin D in the body is 1,25-dihydroxyvitamin D, or calcitriol, which can be made from either vitamin D2 or vitamin D3. To make the active form, vitamin D2 and vitamin D3 are modified in the liver to produce 25-hydroxyvitamin D, which travels through the blood to the kidneys, where it is modified further to make 1,25-dihydroxyvitamin D.
Previous research indicates that the human prostate cancer cell line possesses Vitamin D receptors (VDR) [11-13]. Calcitriol [1alpha,25-dihydroxyvitamin D3] (VD3) is the natural ligand of the vitamin D receptor (VDR). Studies show that VD3 could down regulate the antiapoptotic proteins Bcl-2, Bcl-X(L), and Mcl-1, BAG1L, XIAP, cIAP1, and cIAP2 (without altering the proapoptotic Bax and Bak) in association with increase in apoptosis [14]. In VDR sensitive LNCaP cell lines, VD3 activates downstream effector protease, caspase-3, and upstream initiator protease caspase-9, the apical protease in the mitochondrial (“intrinsic”) pathway for apoptosis. VD3 induced declines in antiapoptotic proteins and also stimulated cytochrome c release from mitochondria by a caspase-independent mechanism [15]. Moreover, apoptosis induction by VD3 was suppressed by overexpressing Bcl-2, a known blocker of cytochrome c release, whereas the caspase-8 suppressor CrmA afforded little protection [15]. Thus, VD3 is capable of inhibiting expression of multiple antiapoptotic proteins in VDRexpressing prostate cancer cells, leading to activation of the mitochondrial pathway for apoptosis. Synthetic analogs of vitamin D3 like 25-hydroxyvitamin D3, is known to exhibit reduced calcemic activity and can elicit antiproliferative effects and other biological actions in LNCaP cells [16]. Taken together, epidemiological and dietary data suggest that both genistein and vitamin D3 play important role in protecting against prostate cancer [17,18].
Like Genistein, another recently known anti tumor phytochemical is Topotecan, which also induces apoptosis in cancer cells. Topotecan is an FDA approved chemotherapy agent under the commercial name of Hycamtin, also referred to as topotecan hydrochloride (topotecan HCl). It was approved for treatment of multiple types of cancer, but it is most widely used for cervical and small cell lung cancer [19,20]. It is a phytochemical and a semisynthetic derivate of camptothecin. It was isolated from the bark and stem of Camptotheca acuminata (Camptotheca, Happy tree), a tree native to China [21,22]. Camptothecin, an alkaloid phytochemical is able to arrest cell growth and proliferation in several carcinoma cell lines. However, it has low solubility and early clinical trials reported adverse side effects, including bone marrow suppression [22-24].
Topotecan has anti-neoplastic properties and is a cytotoxic quinoline-based alkaloid which, during the S phase of the cell cycle, inhibits the standard function of topoisomerase I, an enzyme that deals with replication, repair, and recombination of the double stranded DNA by relieving torsional stress with reversible single strands cuts to the molecule. Topotecan interferes with the usual DNA-Topoisomerase I binding, thus preventing the reannealing and repair of the single strands back to the stable double bond conformation. As a result the continuity of the replication fork is disrupted, replication is halted, and lethal double strand fragments are made which ultimately initiate apoptosis signaling. Intercalation displaces the downstream DNA, thus preventing religation of the cleaved strand. By specifically binding to the enzyme-substrate complex, Topotecan acts as an uncompetitive inhibitor [25-27].
Topotecan has low solubility and it has shown to have a high toxicity level for the optimum working concentration and dosage. Therapeutically, topotecan is mainly used as second line treatment against prostate cancer or recurring prostate tumors [28]. Cytotoxic side effects include: myelosuppression, low blood counts and suppression of the immune system leading to an increase in susceptibility to infection [25,26]. However, topotecan has been associated with inducing oxidative stress and increase in ROS (Reactive oxygen species) and nitrite. Increase in ROS causes irreversible damage to protein by forming protein carbonyl derivative and also elevates DNA stress and damage, ultimately leading to the induction of apoptotic cell death [29-31].
Genistein-Topotecan (Gn-TPT) combination has been shown to induce significant levels of apoptosis in prostate cancer [32]. Synthetic analogs of Vitamin D3 like cholecalciferol (25-hydroxyvitamin D3) has low hypercalcemic activity and low toxicity [16]. Here we investigated the potential additive effect of 25-hydroxyvitamin D3 in 25-hydroxyvitamin D3-Topotecan (Vit D-TPT) combination compared to the Gn-TPT combination in apoptosis induction in LNCaP prostate cancer cells. The potential mechanism of apoptosis induction was also determined.
2. Materials and Methods
2.1. Cell Line
LNCaP (complements of Rumbaugh-Goodwin Institute for Cancer Research [RGI], Plantation, Fl) cells were cultured and maintained in complete RPMI 1640 (SigmaAldrich Chemical Co., St Louis, Mo, USA) media with 10% Fetal Bovine Serum and 1% penicillin/streptomycin.
2.2. Test Agents
Genistein isoflavone (Gn) (Sigma-Aldrich, St. Louis, MO) and 25-hydroxyvitamin D3 [Cholecalciferol] (Vit D) (Sigma-Aldrich, St. Louis, MO) were dissolved with dimethylsulfoxide (DMSO) and diluted with RPMI-media to produce aliquots of 20 μM concentration.
Topotecan Hydrochloride (TPT) (Drummond Scientific Co., Broomall, PA) was diluted into a stock solution with dimethylsulfoxide (DMSO). Stock solution of TPT was further diluted with RPMI-media to produce aliquots ranging in concentration from 2 - 10 μM (TPT 2 - 10 μM).
Final concentrations of DMSO for genistein, topotecan, and 25(OH)D3 did not exceed 0.05%.
2.3. Treatment
The LNCaP cells were cultured in 75 cm3 flask at 37˚C, 5% CO2 and 89% - 90% humidity to achieve 80% - 90% confluence. The cells were harvested, centrifuged and reconstituted into suspension with fresh RPMI 1640 media. 5 × 103 cells in 100 μl of media were dispenses into each well of 96 well microtiter plates and cultured for 48 hours to allow adherence and obtained > 80% confluence. The supernatants were aspirated and the adhered cells were treated with: 1) TPT; 2) Gn-TPT combination; 3) Vit D-TPT combination.
Cell cultures were distributed into three treatment groups, with Gn-TPT combination (Gn 20μM + TPT 2-10μM), Vit D-TPT combination (Vit D 20μM + TPT 2-10μM) and single TPT (TPT 2-10μM) dosages. Experiments had control groups consisting of LNCaP cells cultured in RPMI 1640 media (with 10% FBS, 1% penicillin/streptomycin and L-glutamine) without treatment. All treatments were done in triplicates, and cultured for 24 - 48 hours in a humidified incubator at 37˚C and 5% CO2. At 24 and 48 hours post-treatment/culture, cells were subjected to assay analysis as follows:
2.4. Trypan Blue Assay
Trypan blue exclusion-hemocytometer counting was used to assess the percent viability and initial concentration of the LNCaP cells prior to treatment, according to standard procedures [33,34].
2.5. MTT Assay
MTT [3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide], a tetrazolium dye, is used to determine metabolic status and cell viability. Since the resulting color intensity correlates directly with the amount of metabolically active cells in each well, cell viability can be quantitatively determined by measuring the optical density (OD) in individual wells. After briefly treating the cells as described previously, a volume of 10 μL of MTT reagent was added to each well at 24 hours and 48 hours post-treatment. The plates were further incubated at 37°C and 5% CO2 for 4 hours in the dark, after which a volume of 100 μL of DMSO was added to the cells to solubilize the formazan. Absorbance (OD) of the resultant solution was read at 490 nm using a Multiskan microplate reader (Molecular Devices, Inc., Sunnyvale, CA). The OD obtained was graphed against the concentrations of the drugs. The IC50 was extrapolated from the graphs as a measure to determine the concentration required to obtain expected results and compare the data.
2.6. Acridine Orange/Ethidium Bromide Fluorescence Assay
The acridine orange (AcrO)/ethidium bromide (EtBr) fluorescence assay was used to differentiate between viable, apoptotic, and necrotic cells based on fluorescence emission characteristics. Acridine orange permeates both viable and non-viable cells, causing the nuclei to emit green fluorescence. Since absorption of ethidium bromide is based on the disruption of cell membrane integrity, ethidium bromide selectively stains the nuclei of dead (non-viable) cells to produce red fluorescence. Cells that emit orange/brown colored fluorescence are indicative of apoptosis, while necrotic cells emit red fluorescence. Briefly, Ethidium bromide (25 μL) and acridine orange (75 μL) were mixed to make a cocktail: 3 μL were added to 25 μL of the cell suspensions. Wet-mounts were prepared using 10 μL of each cell suspension and analyzed under a fluorescent microscope with a bandpass filter. Apoptotic cell death was quantified by counting a total of 150 cells per 2 to 3 fields of view.
2.7. Rhodamine 123/Ethidium Bromide Fluorescence Assay
The rhodamine 123 (Rh 123)/ethidium bromide (EtBr) fluorescence assay was also used to differentiate between viable, apoptotic, and necrotic cells, as well as to determine the possible mechanism of apoptosis, based on mitochondrial transmembrane potential. Rh 123 is a cationic fluorochrome which utilizes the transmembrane potential of active mitochondria to diffuse into cells. The intact mitochondrial membranes of living cells therefore allow absorption of the dye, resulting in the emission of bright green fluorescence. Cells, in which the integrity of the mitochondrial membrane has been impaired (nonviable cells), stain lightly with Rh123. However, ethidium bromide selectively enters the disrupted membranes of dead cells and stains the nuclei to produce red fluorescence. Cell samples were briefly washed three times in PBS and resuspended in a final volume of 25 μL. Each cell suspension received 2 μL of the Rh 123 stock solution and was incubated at 37˚C for 5 minutes. Each cell suspension then received 20 μL of EtBr stock solution and was incubated at room temperature for 5 minutes. Finally, 10 μL of each cell mixture was transferred onto a microscope slide covered with a cover slip and examined/analyzed under a fluorescent microscope with a band-pass filter. Green fluorescence is indicative of viable cells; orange/brown cells are apoptotic while necrotic cells emit red fluorescence. The percentage of apoptotic cell death was quantified from an average of 150 cells spread across two to three regions/views on each slide.
2.8. DNA Fragmentation Assay
DNA fragmentation assay distinguishes between necrotic and apoptotic cell death. Cells undergoing apoptosis will cleave their nuclear DNA into 180 - 200 bp DNA breaks, which can be detected through electrophoresis. Necrotic-induced cell death results in irregular DNA breaks, which will appear as smears in the gel. Briefly, after treating the LNCaP cells as previously described, cells were collected and washed in PBS. Following the protocol from the DNeasy Blood and Tissue Kit (Qiagen, Valencia, CA), DNA was extracted from the pellet and quantified using the NanoDrop ND-2000 spectrophotometer (Nano-Drop Technologies, Wilmington, DE). A 2% agarose gel was prepared with 2 µL EtBr and loaded with 10 ng of DNA extract per well. A 1kb DNA ladder was also loaded into one well of the gel as a marker to aid quantification and sizing of the DNA fragments. Gel electrophoresis was performed at 90 - 100 mV for 30 - 45 minutes, and bands in each channel were examined.
2.9. Statistical Analyses
Experiments were performed in triplicate and repeated thrice to confirm results. Significance of the differences in mean values was determined using Graphpad Prism and the Student’s t-test. Statistical significance was defined as p ≤ 0.05.
3. Results
3.1. Growth Inhibition
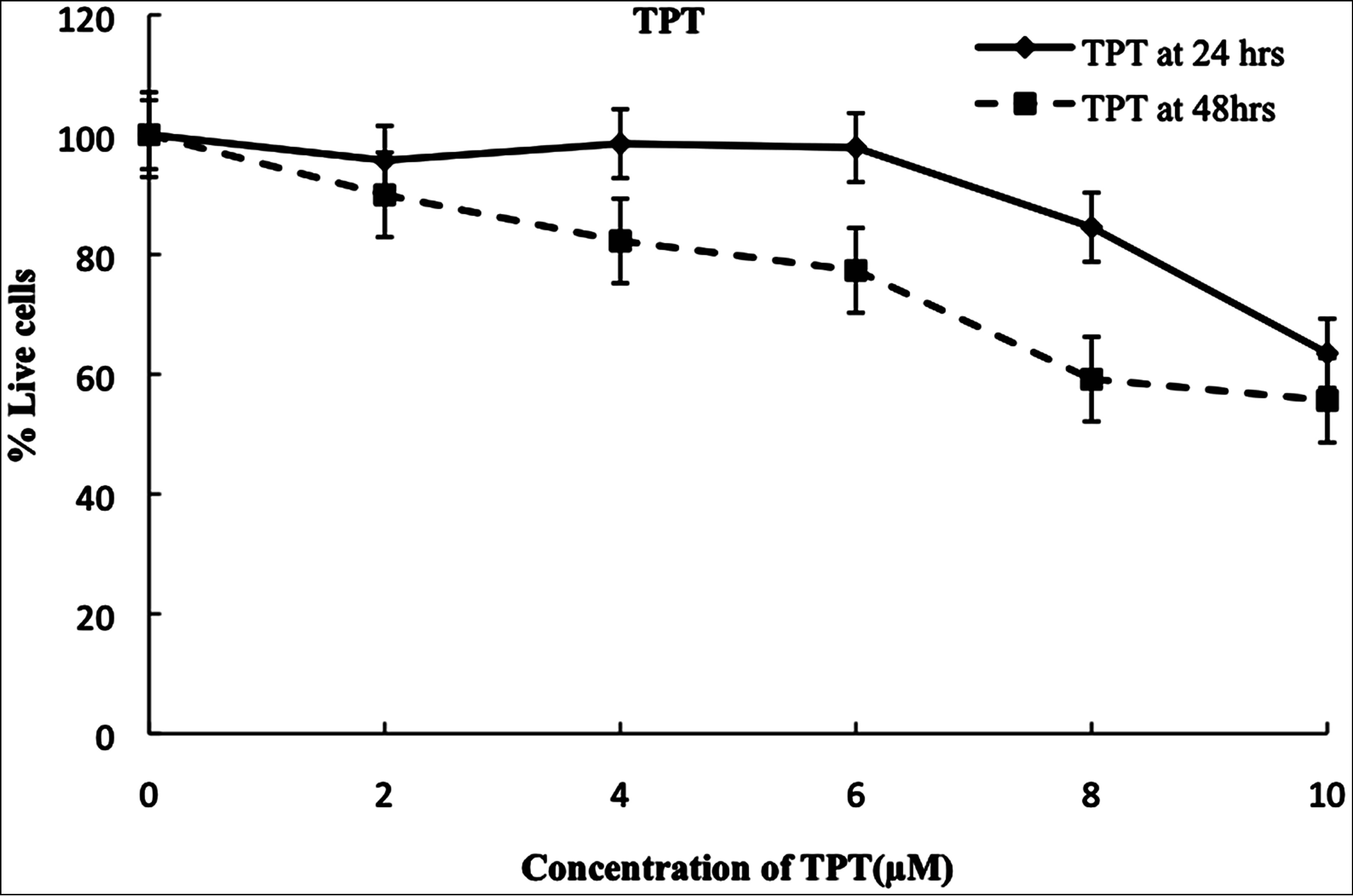
The MTT assay was utilized to test the effects of genistein, 25(OH)D3, and topotecan on the viability/chemosensitivity of LNCaP prostate carcinoma cell lines. Data collected was analyzed based on the knowledge that the absorbance is directly correlated with the amount of viable cells. Results showed that in general the number of viable cells decreased as the concentration of the drugs increased with time of exposure, demonstrating a dose dependent as well as time dependent relationship (Figures 1(a)-(c)). However, for the combination treatment of Vit D-Topotecan (VitD20µM-TPT2-10µM) there was no significant difference in comparison to the single treatment of Topotecan (P > 0.05) at 48 hours (Figure 1(d)). The combination treatment of Genistein-Topo-tecan (Gn20µM-TPT2-10µM) for upto 48 hours showed a more significant dose and time dependent decrease (P < 0.001) in cell viability than the former two treatments (Figures 2(a) and (b), Figure 1(d)). Based on MTT results from previous experiment [31], the EC50 dose of genistein was calculated as 30 µM. In the present study, TPT and Vit D did not show a significant decrease in cell viability at this concentration.
3.2. Acridine Orange/EtBr Assay and Apoptosis Induction
The AcrO/EtBr assay determines whether any of the treatment regimens induced some of the morphological landmark features of apoptosis in the treated cells. The result indicated that both combination and single treatments induced apoptosis. At 24 hours, the combination of Gn20µM-TPT10µM induced the greatest apoptosis cell death compared to the Vit D-TPT combination at the same concentrations (p < 0.01). (Figure 3).
3.3. Rhodamine 123/Ethidium Bromide Assay
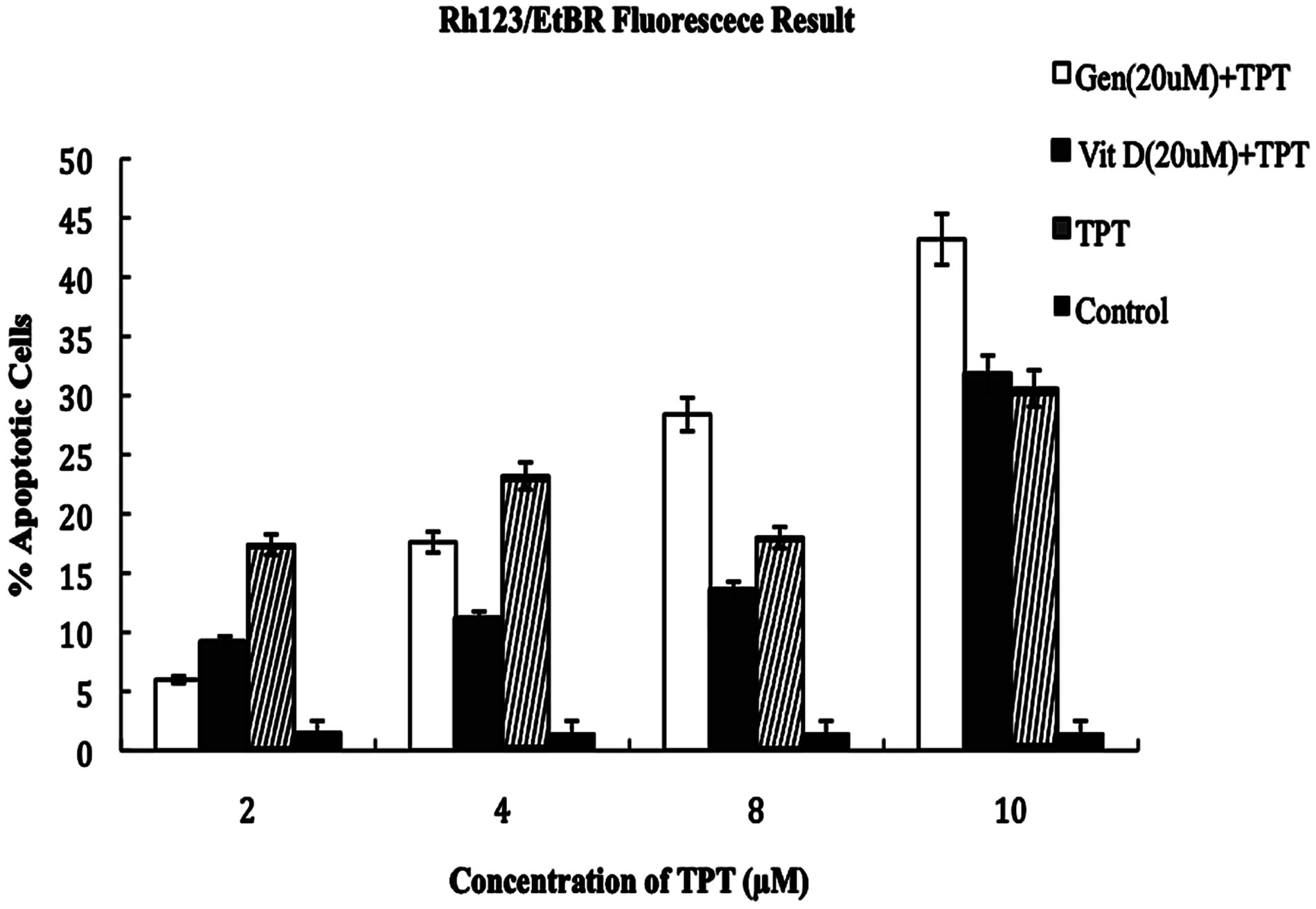
The Rh 123/EtBr assay utilized differential fluorescent staining of viable, apoptotic and necrotic cells to confirm the type of treatment-induced LNCaP cell death, as well as to determine the possible mechanism of action for apoptosis (Figure 4(b)). A greater number of orange/ brown stained cells were observed in all the treatment groups in a dose and time dependent manner (Figure 4(c)). Apoptotic cells with compromised mitochondria showed a reduced transmembrane potential and were visibly stained as described earlier. This suggested the use of the intrinsic-mitochondrial apoptotic pathway and the activation of caspase proteases involved in apoptosis. Results showed that initially 2 µM dose of single treatment with Topotecan was more effective compared to the combination treatments. However, with increased concentration, Genistein-TPT proved to be more effective in cell growth inhibition at a concentration of Gn20µM- TPT10µM (Figure 4(a)) compared to single Topotecan treatment, and VitD-TPT combination at the same concentration (p < 0.01).
3.4. DNA Fragmentation Assay
The presence of uniform DNA fragments (Lane 4 - 6, Figure 5) indicated the induction of apoptotic cell death in LNCaP cells when treated with the combination dosages and single dose of topotecan. DNA breaks of 180 - 200 bp were consistent with the morphological characteristics of apoptosis. Control samples did not demon-
 (a)
(a)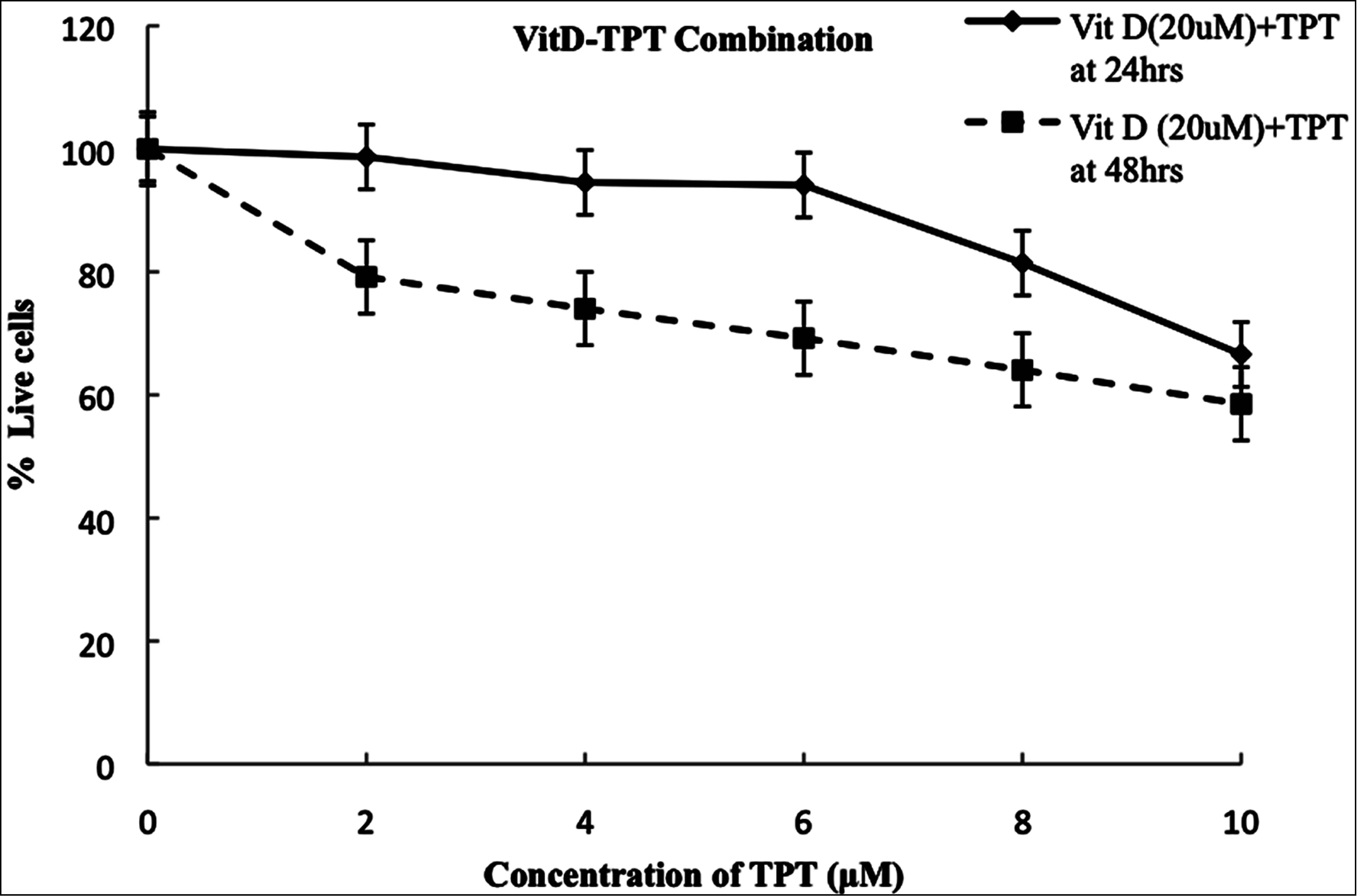 (b)
(b)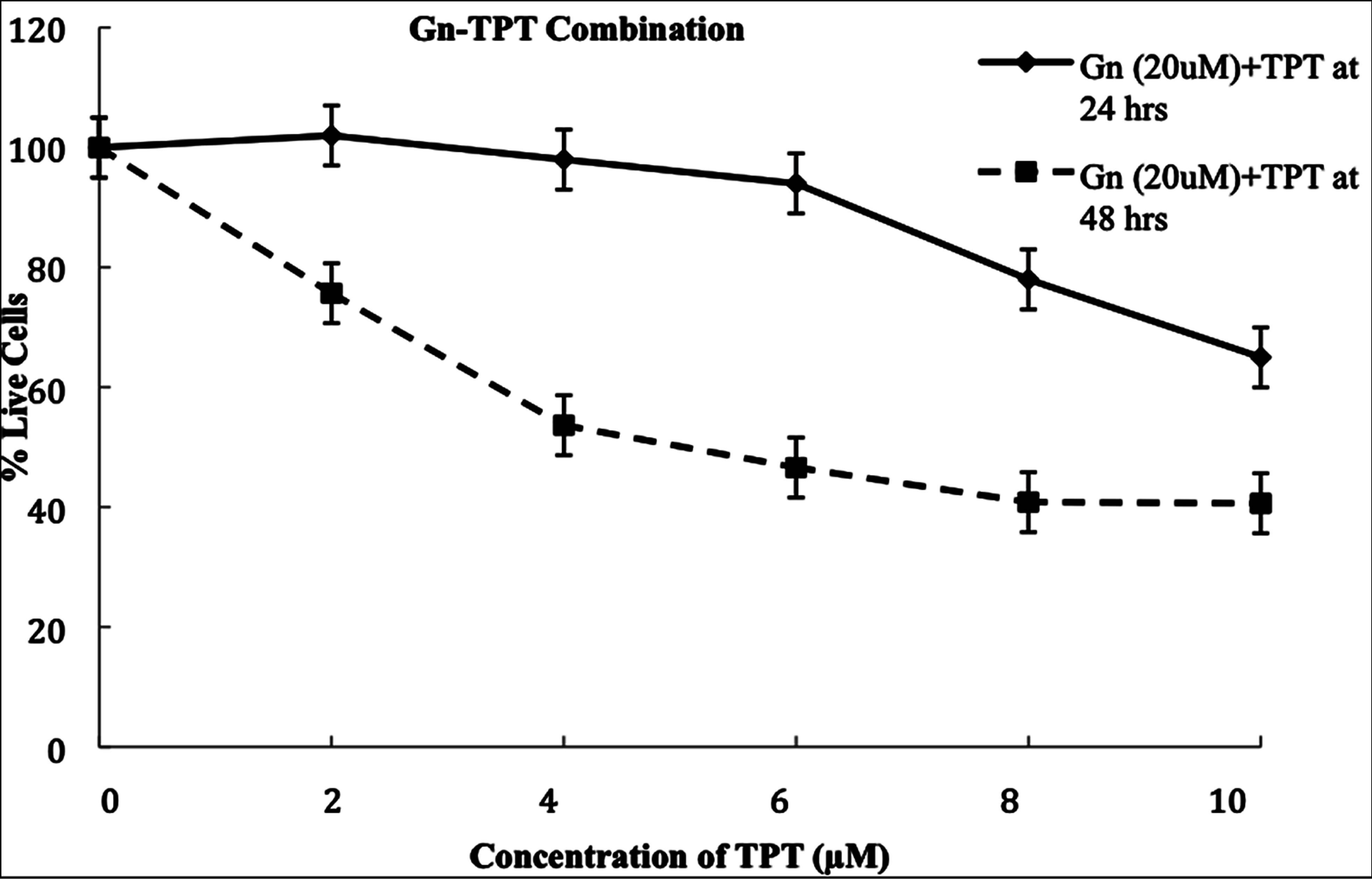 (c)
(c) (d)
(d)
Figure 1. (a)-(c) Growth and viability of LNCap cells was assessed using the MTT Assay. The cells were treated with varying concentrations of Topotecan(TPT2-10μM), Vitamin D3-Topotecan(VitD20µM-TPT2-10μM) and Genistein-Topotecan (Gn20µM-TPT2-10μM) combinations for 24 - 48 hrs; then incubated at 37˚C, 5% CO2, and 89% humidity, as previously described in the experiment. Data are the mean ± SEM(Standard Error of the Mean) of three independent experiments performed in triplicate. Bar = SEM(Standard Error of the Mean). d) Growth and viability of LNCap cells was assessed using the MTT Assay. The cells were treated with varying concentrations of Topotecan (TPT2-10μM), Vitamin D3-Topotecan(VitD20µM-TPT2-10μM) and Genistein-Topotecan(Gn20µM-TPT2-10μM) combinations for 48 hrs; then incubated at 37˚C, 5% CO2, and 89% humidity, as previously described in the experiment. Data are the mean ± SEM( standard Error of the Mean) of three independent experiments performed in triplicate. Bar = SEM ( Standard Error of the Mean).
 (a)
(a)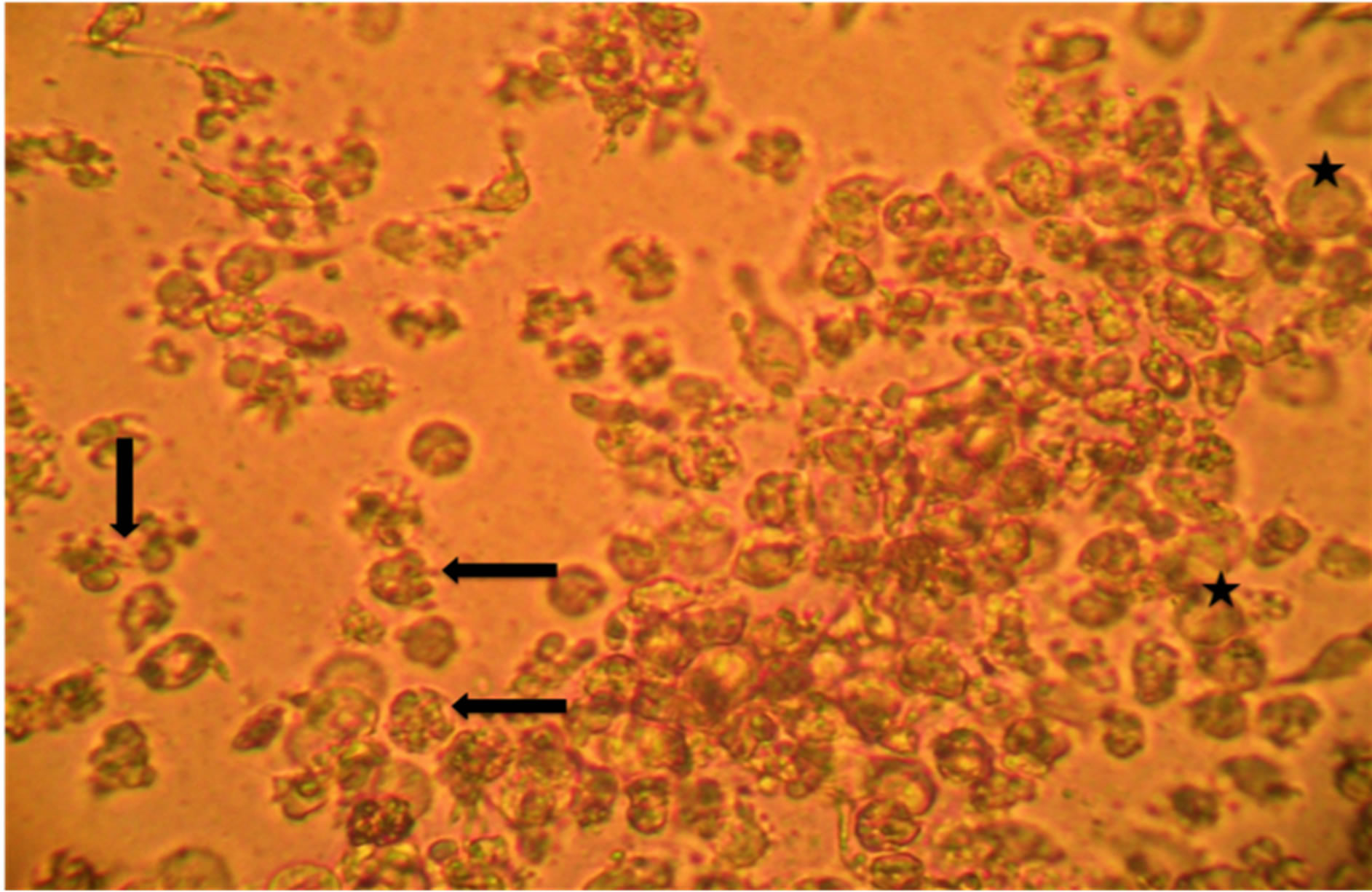 (b)
(b)
Figure 2. (a) Growth and viability of LNCap cells without treatment after 48 hours (Brightfield imaging at 200× total magnification); (b) Fragmentation, shrinkage, and death; indicated by black arrows, of LNCap cells with (TPT10µM- Genestein20µM) combination treatment after 48 hours. Normal, vaible cells indicated by black stars (Brightfield imaging at 200× total magnification).
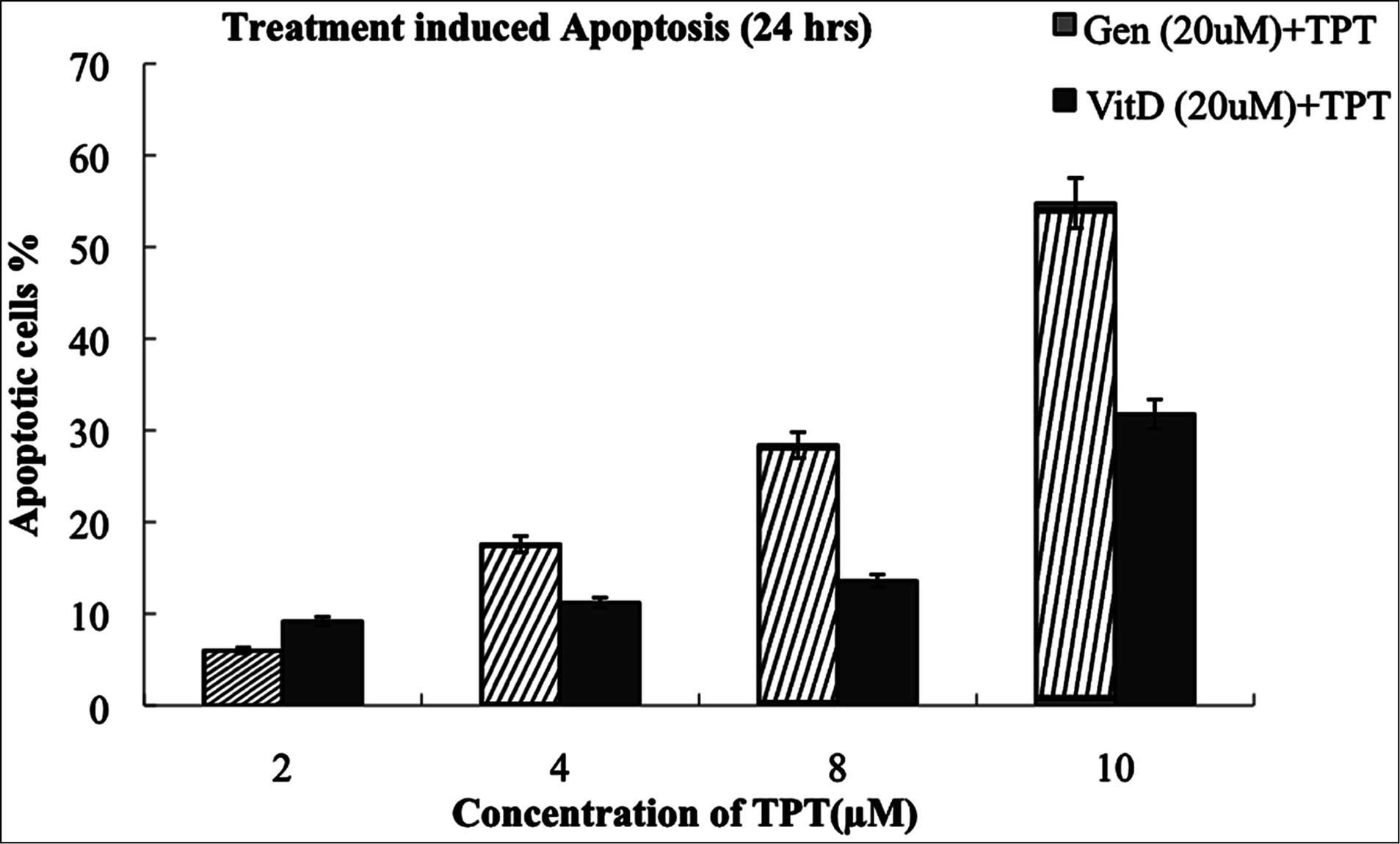
Figure 3. Treatment-induced apoptosis on LNCaP cell line was assessed using Et Br-AcrO to stain after 24 hrs. The percentage of apoptotic cells was quantified from 150 cells per 2 fields of view. At each concentration the difference between Gn + Tpt and VitD + Tpt is significant (p < 0.01).
 (a)
(a)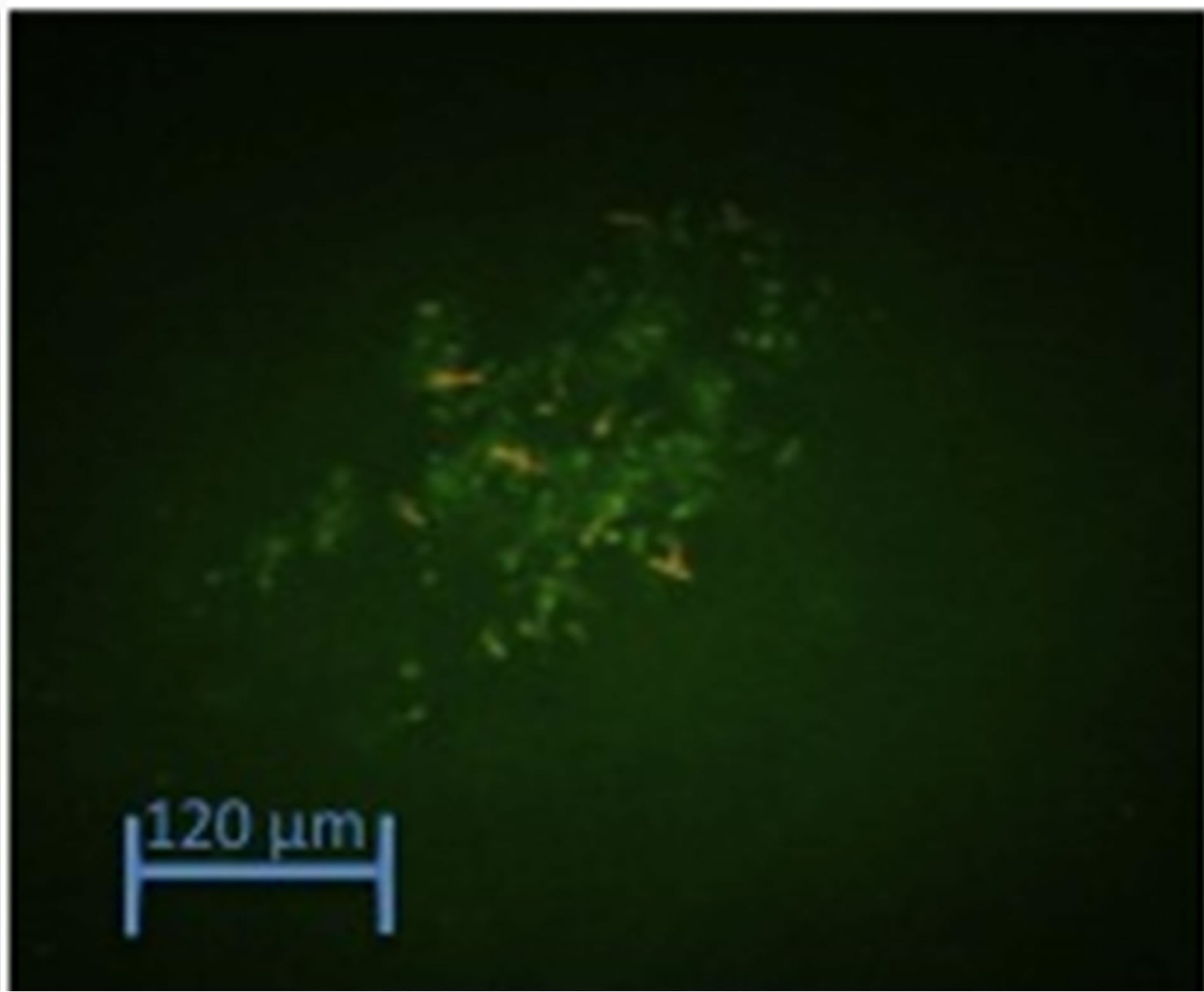 (b)
(b)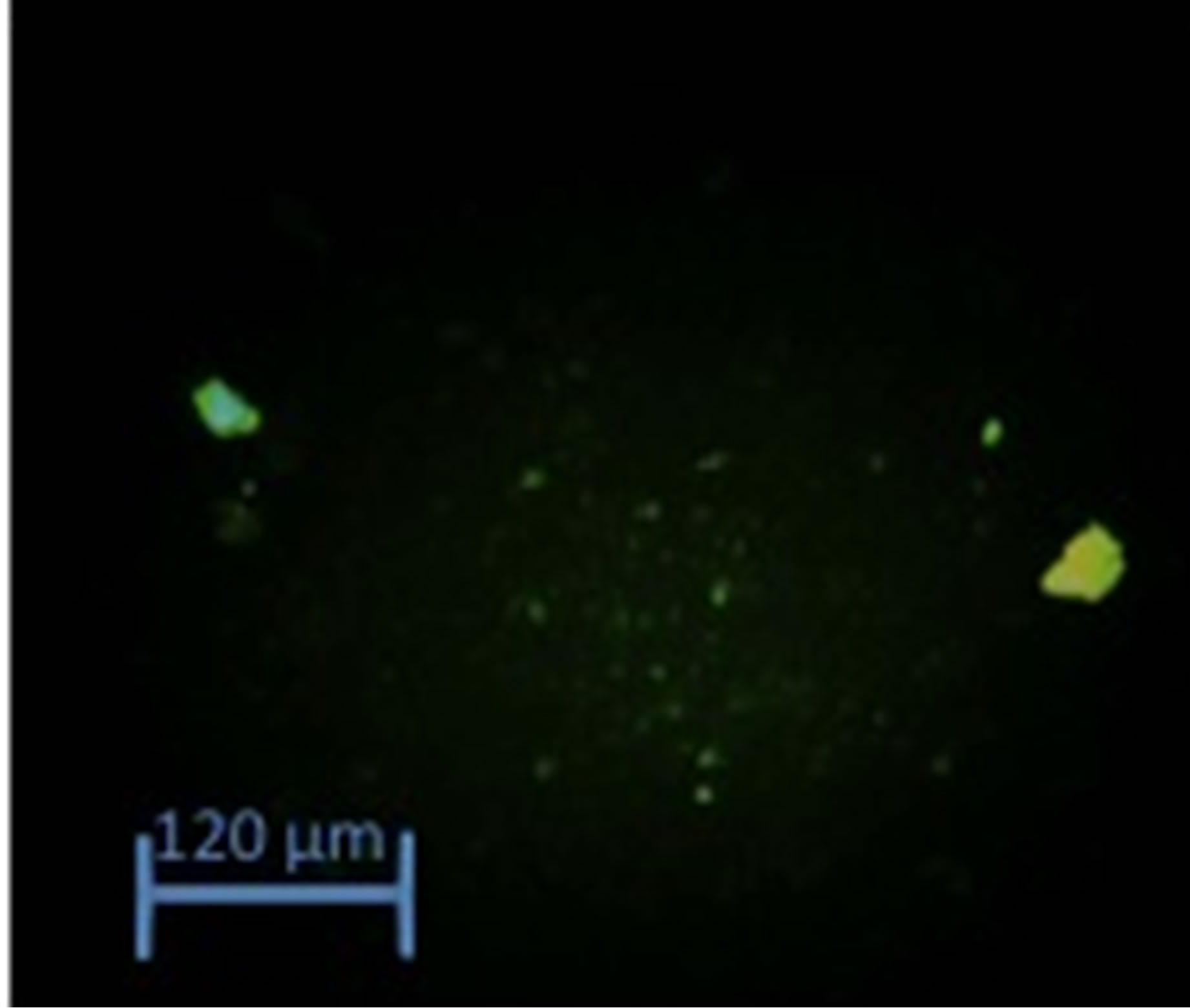 (c)
(c)
Figure 4. (a) Graph of Rh 123/EtBr fluorescence after 24- hours treatment. After 24 hours of treatment, LNCaP cells were mixed with Rh 123/EtBr and examined under the microscope for emission of fluorescent spectra indicative of apoptosis. The percentage of apoptotic cells was quantified from 150 cells per 2 fields of view. At each concentration the difference between Gn+Tpt and VitD+Tpt is significant (p < 0.01). (b) and (c) Images of Rh 123/EtBr fluorescence after 24-hours treatment. Green fluorescence depict viable cells, Orange/brown cells are apoptotic; (b) Control cells; (c) Cells treated with Gn(20µM)+TPT(10 µM).
strate cell death as shown by the single band (Lanes 2 and 7, Figure 5).
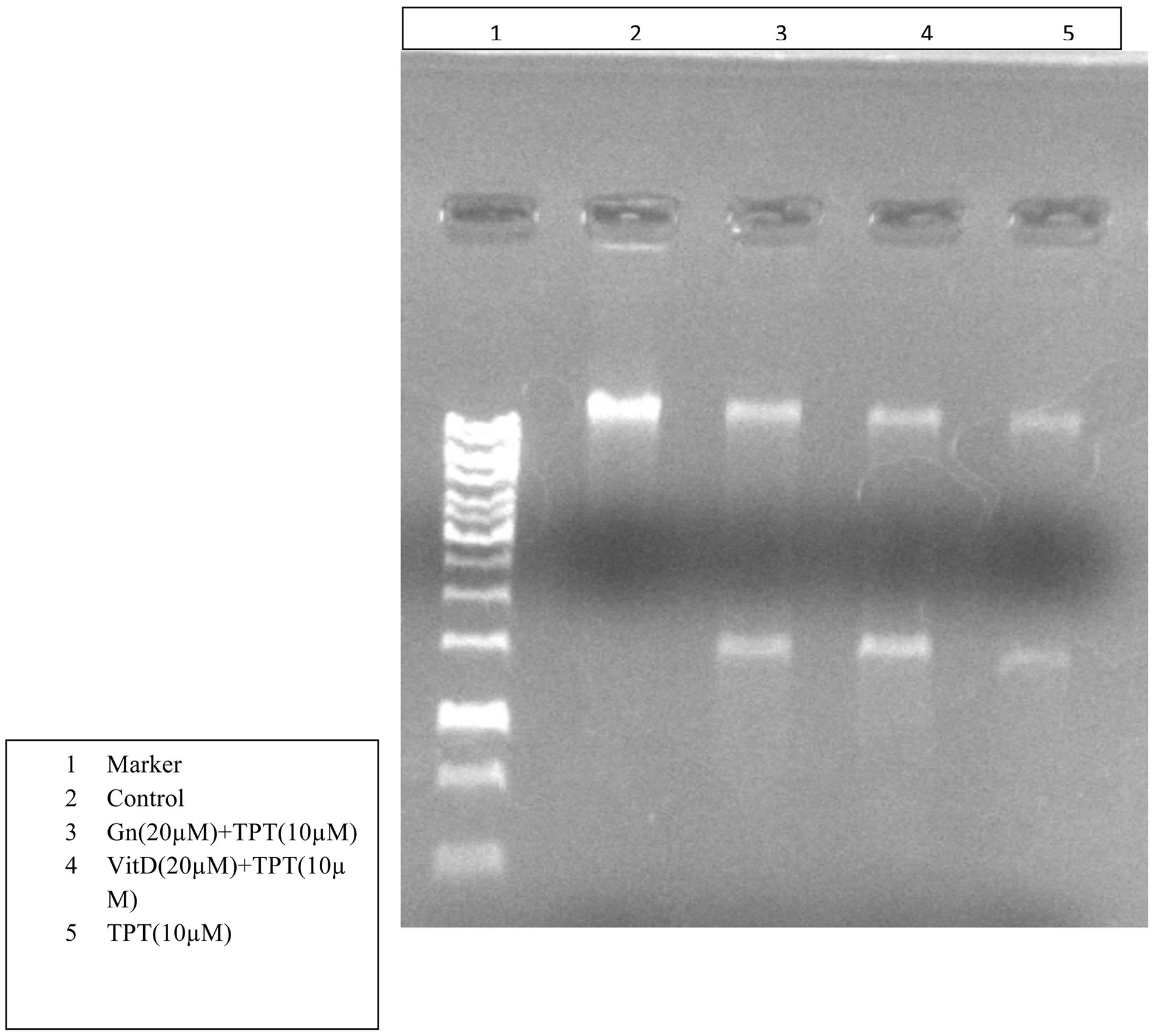
Figure 5. DNA Fragmentation and laddering in Gel Electrophoresis confirmed the apoptotic cell death in treatment groups. No fragments were detected in the control sample.
4. Discussion
Genistein Isoflavone is a well-known phytochemical with anti-cancer properties. Previous investigations have documented the therapeutic capacity of genistein in carcinoma treatment [4-10]. Anti-proliferative effect of cholecalciferol on prostate cancer cells has also been demonstrated [12-15]. The aim of this study was to determine and compare the potential efficacy of genistein-topotecan and Vitamin D3-TPT combinations on LNCaP prostate cancer cells. Topotecan as a single treatment decreased LNCaP cell viability, consistent with previous reports [32]. The present data indicates that the combination treatments with Gn-TPT were significantly more cytotoxic to the cells. The results also indicate that the combination treatment with Gn-TPT was significant at a lower EC50 relative to VitD-TPT combination, consistent with previous reports [32]. Furthermore, the combination treatment of VitD-TPT did not show significant changes compared to the single TPT treatment.
The Rh123/Et Br and AcrO/Et Br revealed treatment-induced apoptosis to be the main mode of cell growth inhibition in LNCaP cells. The Gn-TPT combination treatment yielded the highest percentage of apoptotic cells at a low EC50 comparative to VitD-TPT combination. The result obtained from the Rh123/Et Br/AcrO implicates the mitochondrial pathway in the treatmentinduced apoptosis and the involvement of the intrinsic pathway. The involvement of Caspases 9 and 3 in the signaling pathway of Gn-TPT combination has been demonstrated previously [32] indicating the utilization of the intrinsic apoptotic pathway. The DNA fragmentation assay reiterates the fact by displaying distinct bands for the fragmented DNA.
5. Conclusion
The overall data highlights the significance of Gn-TPT combination over Vit D-TPT combination and TPT in treatment-induced apoptosis. The Gn-TPT combination was significantly more efficacious at lower EC50 (lower cytotoxic level) than either compound alone, implying a greater therapeutic potential of this combination. The apoptosis induction was analyzed to be via intrinsic pathway with increased activity in a time-dependent manner; therefore, suggesting elevation of caspases 9 and 3 as a part of such pathway.
6. Acknowledgements
The authors acknowledge the equipment support of Florida Atlantic University. The collaboration and materials of Rumbaugh-Goodwin Institute for Cancer Research [RGI] at Nova Southeastern University is greatly acknowledged.
REFERENCES
- American Cancer Society, “Prostate Cancer Detailed Guide”. http://www.cancer.org/Cancer/ProstateCancer/DetailedGuide/prostate-cancer-key-statistics
- M. Fukutake, K. Takahashi, H. Ishida, et al., “Quantitation of Genistein and Genistin in Soyabeans and Soyabean Product,” Food and Chemical Toxicology, Vol. 34, No. 5, 1996, pp. 457-461. doi:10.1016/0278-6915(96)87355-8
- C. A. Lamarliniera, “Protection against Breast Cancer with Genistein: A Component of Soy,” The American Journal of Clinical Nutrition, Vol. 71, No. 6, 2000, pp. 1705S-1707S.
- J. K. Kumi-Diaka, M. Hassanhi, K. Merchant, et al., “Influence of Genistein Isoflavone on Matrix Metalloproteinase-2 Expression in Prostate Cancer Cells,” Journal of Medicinal Food, Vol. 9, No. 4, 2006, pp. 491-497.
- J. K. Kumi-Diaka, K. Merchant, A. Haces, et al., “Genistein-Selenium Combination Induces Growth Arrest in Prostate Cancer Cells,” Journal of Medicinal Food, Vol. 13, No. 4, 2010, pp. 842-850. doi:10.1089/jmf.2009.0199
- J. Kumi-Diaka, S. Saddler-Shawnette, A. Aller, et al., “Potential Mechanism of Phytochemical-Induced Apoptosis in Human Prostate Adenocarcinoma Cells: Therapeutic Synergy in Genistein and Beta-Lapachone Combination Treatment,” Cancer Cell International, Vol. 4, No. 1, 2004, p. 5. doi:10.1186/1475-2867-4-5
- S. Balabhadrapathruni, T. J. Thomas, E. J. Yurkow, et al., “Effects of Genistein and Structurally Related Phytoestrogens on Cell Cycle Kinetics and Apoptosis in MDAMB-468 Human Breast Cancer Cells,” Oncology Reports, Vol. 7, No. 1, 2000, pp. 3-12.
- H. S. Seo, J. Ju, K. Jang et al., “Induction of Apoptotic Cell Death by phyToestrogens by Up-Regulating the Levels of Phospho-p53 and p21 in Normal and Malignant Estrogen Receptor α-Negative Breast Cells,” Nutrition Research, Vol. 31, No. 2, 2011, pp. 139-146. doi:10.1016/j.nutres.2011.01.011
- N. Zhou, Y. Yan, W. Li, et al., “Genistein Inhibition of Topoisomerase II Alpha Expression Participated by Sp1 and Sp3 in HeLa cell,” International Journal of Molecular Sciences, Vol. 10, No. 7, 2009, pp. 3255-3268. doi:10.3390/ijms10073255
- Z. Li, J. Li, W. Li, et al., “Genistein Induces G2/M Cell Cycle Arrest via Stable Activation of ERK1/2 Pathway in MDA-MB-231 Breast Cancer Cells,” Cell Biology and Toxicology, Vol. 24, No. 5, 2008, pp. 401-409. doi:10.1007/s10565-008-9054-1
- D. Feldman, R. J. Skowronski and D. M. Peehl, “Vitamin D and Prostate Cancer,” Advances in Experimental Medicine and Biology, Vol. 375, 1995, pp. 53-63. doi:10.1007/978-1-4899-0949-7_5
- R. J. Skowronski, D. M. Peehl and D. Feldman, “Vitamin D and Prostate Cancer: 1, 25 Dihydroxy Vitamin D3 Receptors and Actions in Human Prostate Cancer Cell Lines,” Endocrinology, Vol. 132, No. 5, 1993, pp. 1952- 1960. doi:10.1210/en.132.5.1952
- R. J. Skowronski, D. M. Peehl and D. Feldman, “Actions of Vitamin D3, Analogs on Human Prostate Cancer Cell Lines: Comparison with 1,25-Dihydroxyvitamin D3,” Endocrinology, Vol. 136, No. 1, 1995, pp. 20-26. doi:10.1210/en.136.1.20
- M. Guzey, S. Kitada and J. C. Reed, “Apoptosis Induction by 1alpha,25-Dihydroxyvitamin D3 in Prostate Cancer,” Molecular Cancer Therapeutics, Vol. 1, No. 9, 2002, 667- pp. 677.
- D. M. Peeh, R. J. Skowronski, G. K. Leung, et al., “Antiproliferative Effects of 1,25 Dihydroxy Vitamin D3 on Primary Cultures of Human Prostatic Cells,” Cancer Research, Vol. 54, No. 3, 1994, pp. 805-810.
- R. J. Skowronski, D. M. Peehl and D. Feldman, “Actions of Vitamin D3, Analogs on Human Prostate Cancer Cell Lines: Comparison with 1,25-Dihydroxyvitamin D3,” Endocrinology, Vol. 136, No. 1, 1995, pp. 20-26. doi:10.1210/en.136.1.20
- G. J. Kelloff, “Perspective on Cancer Chemoprevention Research and Drug Development,” Advances in Cancer Research, Vol. 78, pp. 199-334. doi:10.1016/S0065-230X(08)61026-X
- A. Rao, R. D. Woodruff, W. N. Wade, T. E. Kute and S. D. Cramer, “Genistein and Vitamin D Synergistically Inhibit Human Prostatic Epithelial Cell Growth,” Journal of Nutrition, Vol. 132, No. 10, 2002, pp. 3191-3194.
- C. M. Yashar, W. J. Spanos, et al., “Potential of the Radiation Effect with Genistein in Cervical Cancer,” Gynecologic Oncology, Vol. 99, No. 1, 2005, pp. m199-m205. doi:10.1016/j.ygyno.2005.07.002
- F. Lian, M. Bhuiyan, Y. W. Li, N. Wall, M. Kraut and F. H. Sarkar, “Genistein-Induced G2-M Arrest, p21WAF1 Upregulation, and Apoptosis in a Non-Small-Cell Lung Cancer Cell Line,” Nutrition and Cancer, Vol. 31, No. 3, 1998, pp. 184-191. doi:10.1080/01635589809514701
- C. Law, “Basic Research Plays A Key Role in New Patient Treatments,” Journal of the National Cancer Institute, Vol. 88, No. 13, 1996, p. 869. doi:10.1093/jnci/88.13.869
- C. McNeil, “Topotecan after FDA and Asco What’s Next?” Journal of the National Cancer Institute, Vol. 88, No. 12, 1996, pp. 788-789. doi:10.1093/jnci/88.12.788
- H. Ulukan and P. W. Swaan, “Camptothecin: A Review of Their Chemotherapeutic Potential,” Drugs, 62, 14, 2002, pp. 2039-2057.
- M. E. Wall, M. C. Wani, C. E. Cook, et al., “Plant AntiMutagenic Agents, 1. General Bioassay and Isolation Procedures,” Journal of Natural Products, Vol. 51, No. 5, 1988, pp. 866-873. doi:10.1021/np50059a009
- R. Padzur, “FDA Approval for Topotecan Hydrochloride”. http://www.cancer.gov/cancertopics/druginfo/fda-topotecan-hydrochloride
- M. R. Redinbo, L. Stewart, P. Kuhn, et al., “Crystal Structures of Human Topoisomerase I in Covalent and Non-Covalent Complexes with DNA,” Science, Vol. 279, No. 5356, 1998, pp. 1504-1513. doi:10.1126/science.279.5356.1504
- D. J. Adams, M. L. Wahl, J. L. Flowers, et al., “Camptothecin Analogs with Enhanced Activity against Human Breast Cancer Cells. II. Impact of the Tumor pH Gradient,” Cancer Chemotherapy and Pharmacology, Vol. 57, No. 2, 2006, pp. 145-154. doi:10.1007/s00280-005-0008-5
- W. ten Bokkel Huinink, S. R. Lane and G. A. Ross, “Long-Term Survival in a Phase III, Randomized Study of Topotecan versus Paclitaxel in Advanced Epithelial Ovarian Carcinoma,” Annals of Oncology, Vol. 15, No. 1, 2004, pp. 100-103. doi:10.1093/annonc/mdh025
- P. A. Ohneseit, D. Prager, R. Kehlbach and H. P. Rodeman, “Cell Cycle Effects of Topotecan Alone and in Combination with Irradiation,” Radiotherapy & Oncology, Vol. 75, No. 2, 2005, pp. 237-245. doi:10.1016/j.radonc.2005.03.025
- M. Timur, S. H. Akbas and T. Ozben, “The Effect of Topotecan on Oxidative Stress in MCF-7 Human Breast Cancer Cell Line,” Acta Biochimica Polonica, Vol. 52, No. 4, 2005, pp. 897-902.
- C. Erlichman, S. A. Boerner, C. G. Halgren, et al., “The HER Tyrosine Kinase Inhibitor CI1033 Enhances Cytotoxicity of 7-Ethyl-10-hydroxycamptothecin and Topotecan by Inhibiting Breast Cancer Resistance ProteinMediated Drug Efflux,” Cancer Research, Vol. 61, No. 2, 2001, pp. 739-748.
- V. Hormann, J. Kumi-Diaka, M. Durity, et al., “Anticancer Activities of Genistein-Topotecan Combination in Prostate Cancer Cells,” Journal of Cellular and Molecular Medicine, Vol. 20, No. 10, 2012, pp. 1-6. doi:10.1111/j.1582-4934.2012.01576.x
- K. S. Louis and A. C. Siegel, “Cell Viability Analysis Using Trypan Blue: Manual and Automated Methods,” Methods in Molecular Biology, Vol. 740, 2011, pp. 7-12. doi:10.1007/978-1-61779-108-6
- J. Kumi-Diaka and A. Butler, “Caspase 3 Protease Activation during the Process of Genistein-Induced Apoptosis in TM4 Testicular Cells,” Biology of the Cell, Vol. 92, No. 2, 2000, pp. 115-124. doi:10.1016/S0248-4900(00)89019-X

