Natural Resources
Vol.4 No.3(2013), Article ID:33598,6 pages DOI:10.4236/nr.2013.43029
Biological Activity of Spartium junceum L. (Fabaceae) Aromatic Water
![]()
1Department of Pharmacy and Biotechnology, Bologna University, Bologna, Italy; 2Department of Food Science, Naples University, Naples, Italy; 3Department of Chemistry, Calabria University, Arcavacata di Rende, Italy.
Email: *teresa.cerchiara2@unibo.it
Copyright © 2013 Teresa Cerchiara et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received February 6th, 2013; revised May 7th, 2013; accepted May 23rd, 2013
Keywords: Spartium junceum L.; Aromatic Water; Antibacterial Activity; Antioxidant Activity; Cytotoxicity
ABSTRACT
In this study, antimicrobial, antioxidant and cytotoxic activities of Spartium junceum L. aromatic water (SJAW), obtained by vacuum process distillation, were investigated. Antimicrobial activity of SJAW was evaluated against fungal organisms, Gram-positive and Gram-negative bacteria using agar-well diffusion method. Moreover, the ability of SJAW to sensitize Staphyloccus aureus to antibiotics was also evaluated. The antioxidant activity was examined by 2,2’-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity assay and the cytotoxic activity of SJAW main components was tested on melanoma RPMI 7932 and normal keratinocytes NCTC 2544 cell lines using the trypan blue method. The preparation did not show antibacterial activity against tested microorganisms and was unable, at tested concentrations/conditions, to sensitize Staphylococcus aureus to antibiotics clindamycin (2 μg) and tetracycline (30 μg). On the contrary SJAW possesses an appreciable antioxidant and cytotoxic activity suggesting its potential use for pharmaceutical applications.
1. Introduction
From ancient times, plants have been always used as remedies for many human and animal diseases. In literature are reported many papers on the activity of plants, essential oils and methanol or ethanol extracts to retard or inhibit the growth of bacteria, yeast and moulds [1-3]. Nowadays, the increasing resistance of microorganism to some antibiotics makes it necessary to continue the search of new antimicrobial active substances from plants. Moreover, plants are the main sources of natural antioxidants that are important for human health. Recently, natural antioxidants have been studied extensively for their capacity to protect organisms and cells from damage induced by oxidative stress, one of the major causes of ageing and degenerative diseases [4]. Spartium junceum L. is a small shrub particularly widespread in Mediterranean countries, where it grows spontaneously. Flowers of Spartium junceum L. have been reported to show mild sedative and diuretic activities [5], antiulcer [6,7], anti-inflammatory, analgesic [8] and antitumor [9]
activities. The aim of this study was to investigate antimicrobial, antioxidant and cytotoxic activities of SJAW.
2. Experimental
2.1. Materials
Flowers of Spartium junceum L. were collected from plants cultivated in the Orto Botanico of Calabria University (Italy) and identified by Dr N.G. Passalacqua of Museo of Storia Naturale of Calabria and Orto Botanico (Arcavacata Rende, CS, Italy), where a voucher specimen (CLU: 21990) has been deposited.
Nerolidol, 1-octen-3-ol, farnesene, linalool, lynalil acetate and 2,4-bis(1,1-dymethylethyl)phenol were purchased from Sigma Aldrich (Milan, Italy).
2.2. Spartium junceum L. Aromatic Water
The aromatic water was extracted by flowers of Spartium junceum L. using vacuum process distillation as reported in European Patent “Essential oil and aromatic water extraction process from vegetable matrices” by Liguori et al. [10]. Briefly, in a typical experiment, 130 g of flowers were collected in a flask and subjected to vacuum distillation by immersion in a previously heated oil bath (120˚C). The produced steam was condensed by decreasing the temperature providing 65 mL of aromatic water.
SPME-GC-MS analysis: The aromatic water was analyzed by GC/MS combined with solid phase microextraction (SPME), an extraction technique by which the analytes are directly extracted and concentrated on a fiber coating by direct immersion. The studies were conducted with SPME devices purchased from Supelco. A fused silica gel fiber coated with a 65 mm film of Carbowax/divinylbenzene (CW/DWB) was tested. To stabilize the phase and remove all contaminants, the fiber was conditioned before use for 0.5 h in the GC injector at 220˚C. After conditioning, the fiber was desorbed for 10 min and analysis was performed to evaluate the blank. The SPME procedure involved the immersion of the fiber in a vial containing 7 mL of aromatic water for 2 h. The last step was thermal desorption of the analytes into the injection port of the gas chromatograph for 10 min.
GC-MS analyses were performed using a 6890N Network GC System (Agilent Technologies Inc., Palo Alto, CA-USA) equipped with a HP-5MS (30 m × 0.25 mm I.D, dimethylsiloxane 5%; film thickness 0.25 mm) capillary column, and with a 5973 Network MSD mass spectrometer, operated in EI ionization mode (70 ev). GC-MS analyses were carried out in splitless mode (splitless time: 1 min), using helium as carrier gas (1 mL/min flow rate). The injection port was heated at 250˚C. The column was maintained at an initial temperature of 60˚C for 2 min, and then programmed at 7˚C/ min to a final temperature of 280˚C, where it was maintained for 10 min. Identification of the compounds was carried out by comparison with the MS and RT of authentic samples dissolved in water and analyzed under the same conditions (SPME-GC-MS).
HPLC/DAD analysis: The aromatic water was analyzed by HPLC coupled with a diode array detector (DAD). The Shimadzu HPLC system was equipped with LC-20AD pumps, SIL-20A autosampler (20 μL injection volume) and a model SPDM20A-CBM20A PDA/UV detector. Aromatic water components were separated on a C18 Discovery column (Supelco; 5 μm; L × I.D. 15 cm × 3.0 mm)) with a mobile phase flow rate of 1 mL/min. Chromatographic assays were performed at room temperature under the following conditions: initial = 50% H2O and 50% MeCN; 5 min = 50% H2O and 50 % MeCN; 15 min = 40% H2O and 60% MeCN; 50 min = 40% H2O and 60% MeCN; 60 min = 50% H2O and 50% MeCN. Identification of the compounds was carried out by comparing the retention times (Rt) and the corresponding UV absorbance spectra with those of the single reference standards. The data were integrated and analyzed using a Shimadzu LC-Solution.
2.3. Antimicrobial Activity
Evaluation of antimicrobial activity of SJAW was carried out as previously described by Villani et al. [11]. Particularly, the antimicrobial activity was estimated by an agar well diffusion assay. Briefly, a serial 2-fold dilution in phosphate buffer solution (50 mM pH 7.0) of SJAW was prepared, and 10 μL of each dilution were spotted onto agar soft media seeded with about 105 CFU/mL target microorganism. A total of 10 different dilutions, were analysed in the range of undiluted to 1:512. Malt Extract Agar (Oxoid) and the Tryptone Soya Agar (Oxoid) were used as media for fungal (Aspergillus brasiliensis-niger ATCC 16404; Candida albicans ATCC 10231) and bacterial (Enterococcus hirae ATCC 10541; Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 10536; Escherichia coli K12 ATCC 10538; Pseudomonas aeruginosa ATCC 15442) organisms, respectively. After incubation for 16 h at 37˚C (for bacteria) or 72 h at 28˚C (for fungi), antimicrobial activity was considered positive when a clear growth inhibition halo around the spot was presented.
2.4. Antibiotic Disk Assay
The ability of SJAW to sensitize Staphylococcus aureus against antibiotics was evaluated by a modification of the method recently described by Simões et al. [12]. Cells from an overnight growth culture of strains Staphylococcus aureus ATCC 6538 and AB-8802 (enterotoxinogenic strain) [13] were suspended in 0.5 mL of phosphate (KH2PO4; Na2HPO4) buffer (2 mM, pH 7) and mixed with 6 mL (0.7% w/v) of TSA (Oxoid) (final cell concentration was approximately 106 CFU/mL) tempered at 45˚C. Pure SJAW or diluted in ethanol (0.5% v/v) were added to the cell broth mixture yielding final concentrations of: a) SJAW 1% (estimated nerolidol concentration 2 mM); b) SJAW 5% (estimated nerolidol concentration 10 mM); c) SJAW 5% (estimated nerolidol concentration 10 mM) and 5% ethanol. Antibiotic discs containing TE-30 (tetracicline 30 μg-BBL) and DA-2 (clindamycin 2 μg-Oxoid) were placed on the surface of plates containing SJAW/SJAW ethanol and bacteria. After 24 h incubation period at 37˚C, zones of growth inhibition were measured. Tests without SJAW and ethanol were analysed as controls.
2.5. Antioxidant Activity (DPPH Assay)
The antioxidant activity was determined by the 2,2’-diphenyl-1-picrylhydrazyl radical (DPPH, Sigma Aldrich) reduction assay as described by Brand-Williams et al. [14]. Different concentrations (10, 20, 50, 75 and 100 μg/ml) of SJAW as well as ascorbic acid (used as standard antioxidant compound) were mixed with a solution of DPPH (0.1 mM in methanol) at room temperature. The mixtures were kept in dark for 30 min and the absorbance was measured at 517 nm using a UV-Vis 1601 spectrophotometer (Shimadzu). Methanol was used as a blank solution and DPPH solution in methanol as the control. The test was carried out in triplicate. Results are expressed as percentage of inhibition of the DPPH radical according to the following equation:
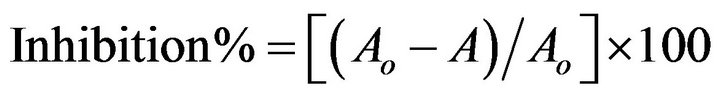
where Ao was the absorbance of DPPH without sample and A was the absorbance of sample with DPPH.
2.6. Cell Viability Assay
Human melanoma RPMI 7932 and normal keratinocytes NCTC 2544 cell lines were obtained from ICLC (Interlab Cell Line Collection), Genoa (Italy).
RPMI 7932 cells were grown in sterile RPMI 1640 medium supplemented with 10% (v/v) fetal bovine serum (FBS), 2 mM L-glutamine, 50 U/ml penicillin, 50 μg/ml streptomycin (all from Sigma) and were incubated at 37˚C in a fully humidified atmosphere of 5% CO2 and 95% air.
NCTC 2544 cells were grown in sterile MEM (EBSS) medium supplemented with 10% (v/v) fetal bovine serum (FBS), 2 mM L-glutamine, 1% non-essential amino acids, 50U/ml penicillin, 50 μg/ml streptomycin (all from Sigma) and were incubated at 37˚C in a fully humidified atmosphere of 5% CO2 and 95% air.
RPMI 7932 and NCTC 2544 cells were seeded on 6- welled plates at a density, respectively, of 5 × 104 and 3 × 104 cell/well in the culture medium. The commercially available identified components (nerolidol, 1-octen-3-ol, farnesene, linalool, lynalil acetate and 2,4-bis (1,1-dy-methylethyl)phenol were dissolved in dimethyl sulfoxide (DMSO) at the concentration of 0.1 μl/ml and were added in the RPMI 7932 and NCTC 2544 culture medium so that total incubation volume did not exceed 3 ml. The cells were incubated in the presence or absence of tested components. The cell number and viability were determined by the trypan blue exclusion method using a hemocytometer. The percentage inhibition of cell viability was calculated using cells treated with physiologic water as controls. Each experiment was repeated at least three times with duplicate samples. The data were expressed as mean ± standard deviation and statistical analysis for significance was performed by means of the Student’s test; values of p < 0.05 were considered statistically significant.
3. Results and Discussion
In this work, we studied biological activity of Spartium junceum L. aromatic water. Firstly, we analyzed the aromatic water extracted by flowers according to the methods reported in the literature [15]. The chemical composition of the extracted SJAW for vacuum distillation was determined as reported in our previous article [9]. Figure 1 shows a typical GC/MS chromatogram of SJAW and the components of the mixture were identified by SPME/GC/MS using as a reference an authentic samples of individual analytes (Table 1).
The quantitative measurement of some components of interest was made by HPLC/DAD and the results are reported in Table 2. The aromatic water composition was comparable with that reported in the literature [9] confirming nerolidol (53.2%) and 1-octen-3-ol (18.2%) as the major components.
The antimicrobial activity of SJAW was evaluated herein against fungal organisms (Aspergillus brasiliensis; Candida albicans), Gram-positive (Enterococcus hirae; Staphylococcus aureus) and Gram-negative (Escherichia coli; Pseudomonas aeruginosa) bacteria.
The mechanism of antibacterial action of sesquiterpenoids (including nerolidol) is yet not fully understood, but it is speculated to involve membrane disruption through lipophilic compounds [16]. According to Trombetta et al. [16], this action results in membrane expansion, increase of membrane fluidity and permeability, disturbance of membrane embedded proteins, inhibition of respiration, and alteration of ion transport processes. This action can contribute to the decrease of microbial resistance and avoid the spread of resistant strains [17]. Results obtained, in this study, by agar-well diffusion assay demonstrated that the SJAW did not have antimicrobial activity against tested microorganisms also when tested as undiluted (data not shown).
It was shown that sesquiterpenoid nerolidol was able to sensitize Staphylococcus aureus and Escherichia coli
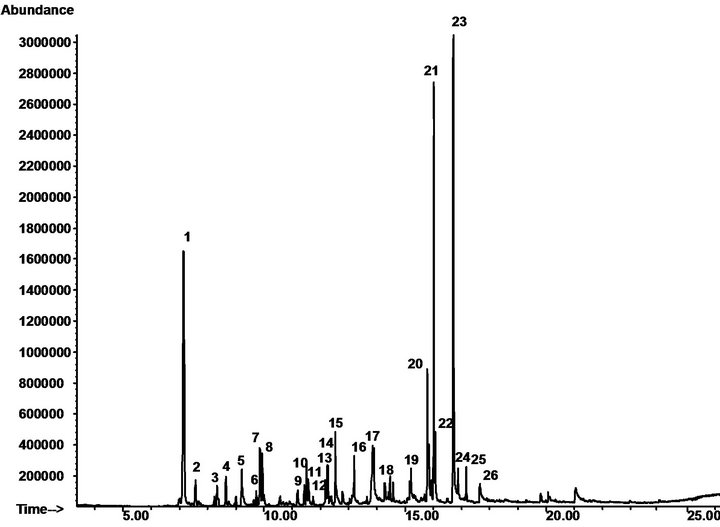
Figure 1. Typical GC/MS chromatogram of SJAW.

Table 1. Composition of Spartium junceum L. aromatic water by SPME-GC-MS analysis.
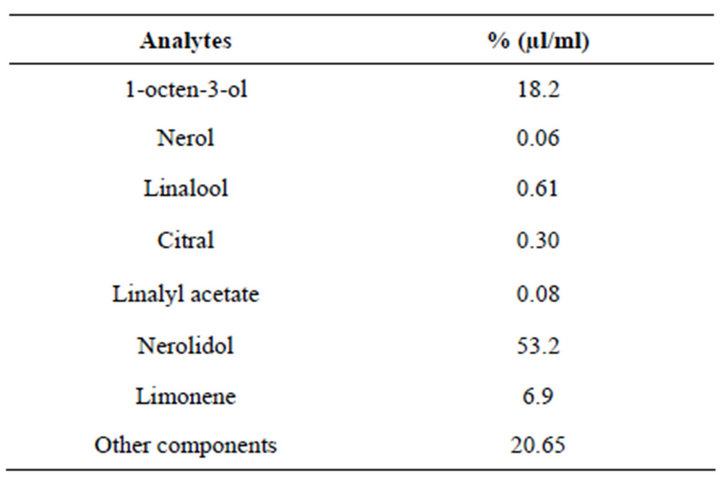
Table 2. Quantitative assay in Spartium junceum L. aromatic water by HPLC/DAD.
against antibiotics [12,18]. In this work, we investigated also on the ability of SJAW to sensitize two Staphylococcus aureus strains. The results of sensitization tests performed in this study are reported in Table 3. As Table 3 shows only in the case of combination SJWA-Ethanol (5% vol/vol each) a little increase (3 - 4 mm) of the halo diameter was shown.
These results are in contrast with those obtained by Brehm-Steche & Johnson [17]. These authors showed an enhanced antimicrobial activity effect of increasing concentrations of pure nerolidol (0, 1 and 2 mM) on susceptibility of S. aureus to ciprofloxacin, clindamycin and tetracycline. This behaviour can be attributed to the others compounds presented in the SJAW affecting the antimicrobial activity of nerolidol.
Figure 2 shows the antioxidant activity of SJAW as well as the standard antioxidant (ascorbic acid) evaluated by DPPH assay. SJAW showed a good antioxidant activity, although lower than that found for ascorbic acid. This activity can be attributed mainly to the high content of nerolidol, but also minor components of aromatic water such as linalool, limonene which show antioxidant properties as reported in literature [19-21].
Moreover as reported in our previous article, SJAW exhibited a marked growth inhibitory effect on RPMI 7932, while it was not cytotoxic to the control NCTC 2544 cells [9]. So, to identify the active components involved in the growth inhibition of the melanoma cells, we tested commercially available pure compounds (linalool, farnesene, 1-octen-3-ol, linalyl acetate and 2,4-bis (1,1-dymethylethyl)phenol). Among the compounds tested, nerolidol, farnesene and linalool are significantly cytotoxic against melanoma cells RPMI 7932, while only linalool exhibited a low growth inhibitory effect on normal cells NCTC 2544 showing its selectively cytotoxic activity against melanoma cell lines (Figure 3). Recently, the antiproliferative activity of linalool on amelanotic melanoma cells [22] as well as leukaemia cells [23] are
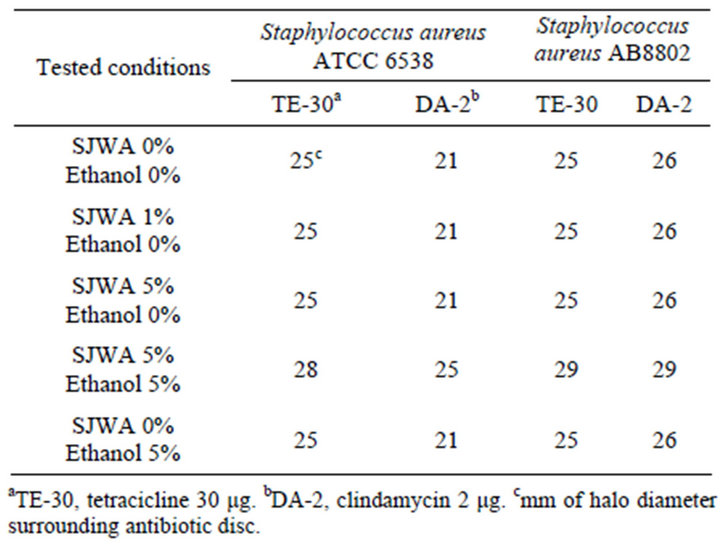
Table 3. Antimicrobial activity of SJAW.
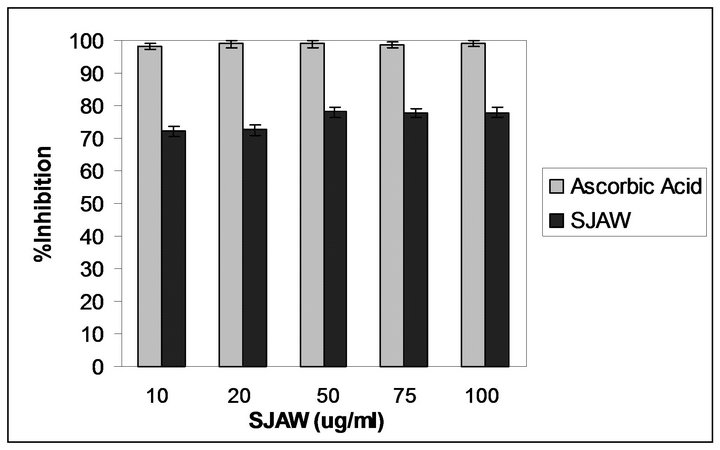
Figure 2. DPPH scavenging activity of SJAW and ascorbic acid. Values are means ± SD (n = 3).
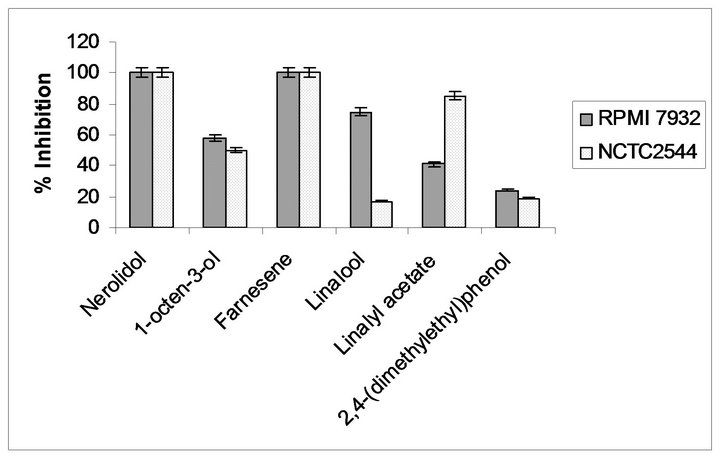
Figure 3. Cytotoxic effect of various components on RPMI 7932 and NCTC 2544 cell lines. Data were expressed as mean ± SD (n = 3).
reported in literature, but further studies are needed to clarify molecular mechanism involved in the growth inhibition of the melanoma cell lines.
4. Conclusion
SJAW did not show any antibacterial activity against tested microorganisms and it is unable to significantly sensitize Staphylococcus aureus strains to antibiotics. However, the aromatic water possesses a good antioxidant and antitumor activity suggesting its potential use for pharmaceutical applications.
5. Acknowledgements
This work was supported by the Ministero dell’Istruzione, dell’Università e della Ricerca (MIUR, Roma, Italy) (Project No. 987, “Development and optimization of processes for the production of materials from broom fibers”) and by the University of Calabria. The authors are thankful to Dr. N. G. Passalacqua and Orto Botanico for providing flowers.
REFERENCES
- M. J. Gonzàlez and J. M. Marioli, “Antibacterial Activity of Water Extracts and Essential Oils of Various Aromatic Plants against Paenibacillus larvae, the Causative Agent of American Foulbrood,” Journal of Invertebrate Pathology, Vol. 104, No. 3, 2010, pp. 209-213. doi:10.1016/j.jip.2010.04.005
- S. T. Chang, P. F. Chen and S. C. Chang, “Antibacterial Activity of Leaf Essential Oils and Their Constituents from Cinnamomum osmophloeum,” Journal of Ethnopharmacology, Vol. 77, No. 1, 2001, pp. 123-127. doi:10.1016/S0378-8741(01)00273-2
- O. A. Kumar, L. M. Naidu and K. G. R. Rao, “In Vitro Antibacterial Activity in the Extracts of Andrographis paniculata Burm. F.,” International Journal of PharmTech Research, Vol. 2, No. 2, 2010, pp. 1383-1385.
- E. Koksal, E. Bursal, E. Dikici, F. Tozoglu and I. Gulcin, “Antioxidant Activity of Melissa officinalis Leaves,” Journal of Medicinal Plants Research, Vol. 5, No. 2, 2011, pp. 217-222.
- T. Baytop, “Phytotherapy in Turkey, Past and Present,” Istanbul University Press, Istanbul, 1984.
- E. Yesilada, Y. Takaishi, T. Fujita and E. Sezik, “AntiUlcerogenic Effects of Spartium junceum Flowers on in Vivo Test Models in Rats,” Journal of Ethnopharmacology, Vol. 70, No. 3, 2000, pp. 219-226. doi:10.1016/S0378-8741(99)00180-4
- E. Yesilada and Y. Takaishi, “A Saponin with AntiUlcerogenic Effect from the Flowers of Spartium junceum,” Phytochemistry, Vol. 51, No. 7, 1999, pp. 903-908. doi:10.1016/S0031-9422(99)00198-3
- L. Menghini, P. Massarelli, G. Bruni and R. Pagiotti, “Anti-Inflammatory and Analgesic Effects of Spartium junceum L. Flower Extracts: A Preliminary Study,” Journal of Medicinal Food, Vol. 9, No. 3, 2006, pp. 386- 390. doi:10.1089/jmf.2006.9.386
- T. Cerchiara, S. V. Straface, G. Chidichimo, E. L. Belsito, A. Liguori, B. Luppi, F. Bigucci and V. Zecchi, “Spartium junceum Aromatic Water: Chemical Composition and antitumor Activity,” Natural Product Communications, Vol. 7, No. 1, 2012, pp. 137-140.
- A. Liguori, E. L. Belsito, A. Leggio and G. Chidichimo, “Essential oil and Aromatic Water Extraction Process from Vegetable Matrices,” European Patent No. 07020630, 5-2114, 2007.
- F. Villani, O. Pepe, G. Mauriello, G. Salzano, G. Moschetti and S. Coppola, “Antimicrobial Activity of Staphylococcus xylosus from Italian Sausage against Listeria monocytogenes,” Letters of Applied Microbiology, Vol. 18, No. 3, 1994, pp. 159-161. doi:10.1111/j.1472-765X.1994.tb00833.x
- M. Simões, S. Rocha, M. A. Coimbra and M. J. Vieira, “Enhancement of Escherichia coli and Staphylococcus aureus Antibiotic Susceptibility Using Sesquiterpenoids,” Medicinal Chemistry, Vol. 4, No. 6, 2008, pp. 616-623. doi:10.2174/157340608786242016
- G. Blaiotta, D. Ercolini, C. Pennacchia, V. Fusco, A. Casaburi, O. Pepe and F. Villani, “PCR Detection of Staphylococcal Enterotoxin Genes in Staphylococcus spp. Strains Isolated from Meat and Dairy Products. Evidence for New Variants of seG and seI in S. aureus AB-8802,” Journal of Applied Microbiology, Vol. 97, No. 4, 2004, pp. 719-730. doi:10.1111/j.1365-2672.2004.02349.x
- W. Brand-Williams, M. E. Cuvelier and C. Berset, “Use of Free Radical Method to Evaluate Antioxidant Activity,” Lebensmittel-Eissenschaft und Technologie, Vol. 28, No. 1, 1995, pp. 25-30.
- E. L. Belsito, C. Carbone, M. L. Di Gioia, A. Leggio, A. Liguori, F. Perri, C. Siciliano and M. C. Viscomi, “Comparison of the Volatile Constituents in Cold-Pressed Bergamot Oil and a Volatile Oil Isolated by Vacuum Distillation,” Journal of Agricultural and Food Chemistry, Vol. 55, No. 19, 2007, pp. 7847-7851. doi:10.1021/jf070997q
- D. Trombetta, F. Castelli, M. G. Sarpietro, V. Venuti, M. Cristani, C. Daniele, A. Saija, G. Mazzanti and G. Bisignano, “Mechanisms of Antibacterial Action of Three Monoterpenes,” Antimicrobial Agents and Chemotherapy, Vol. 49, No. 6, 2005, pp. 2472-2478. doi:10.1128/AAC.49.6.2474-2478.2005
- A. C. Vatopoulos, V. Kalapothaki and N. J. Legakis, “Bacterial Resistance to Ciprofloxacin in Greece: Results from the National Electronic Surveillance System,” Emerging Infectious Disease, Vol. 5, No. 3, 1999, pp. 471-476. doi:10.3201/eid0503.990325
- B. F. Brehm-Steche and E. A. Johnson, “Sensitization of Staphylococcus aureus and Escherichia coli to Antibiotics by the Sesquiterpenoids Nerolidol, Farnesol, Bisabolol, and Apritone,” Antimicrobial Agents and Chemotherapy, Vol. 47, No. 10, 2003, pp. 3357-3360. doi:10.1128/AAC.47.10.3357-3360.2003
- M. F. Ferreira, C. M. Palmeira, M. M. Oliveira, D. Santos, A. M. Simoes, S. M. Rocha, M. A. Coimba and F. Peixoto, “Nerolidol Effects on Mitochondrial and Cellular Energetics,” Toxicology in Vitro, Vol. 26, No. 2, 2012, pp. 189-196. doi:10.1016/j.tiv.2011.11.009
- S. Pacifico, B. D’Abrosca, A. Golino, C. Mastellone, S. Piccolella, A. Fiorentino and P. Monaco, “Antioxidant Evaluation of Polyhydroxylated Nerolidols from Redroot Pigweed (Amarathus retroflexus) Leaves,” LWT—Food Science and Technology, Vol. 41, No. 9, 2008, pp. 1665- 1671.
- P. Pripdeevech, W. Chumpolsri, P. Suttiarporn and S. Wongpornchai, “The Chemical Composition and Antioxidant Activities of Basil from Thailand Using Retention Indices and Comprehensive Two-Dimensional Gas Chromatography,” Journal of the Serbian Chemical Society, Vol. 75, No. 11, 2010, pp. 1503-1513. doi:10.2298/JSC100203125P
- M. R. Loizzo, R. Tundis, F. Menichini, A. M. Saab, G. A. Statti and F. Menichini, “Antiproliferative Effects of Essential Oils and Their Major Constituents in Human Renal Adenocarcinoma and Amelanotic Melanoma Cells”, Cell Proliferation, Vol. 41, No. 6, 2008, pp. 1002-1012. doi:10.1111/j.1365-2184.2008.00561.x
- Y. Gu, Z. Ting, X. Qiu, X. Zhang, X. Gan, Y. Fang, X. Xu and R. Xu, “Linalool Preferentially Induces Robust Apoptosis of a Variety of Leukemia Cells via Upregulating p53and Cyclin-Dependent Kinase Inhibitors,” Toxicology, Vol. 268, No. 1-2, 2010, pp. 19-24. doi:10.1016/j.tox.2009.11.013
NOTES
*Corresponding author.

