World Journal of Cardiovascular Diseases
Vol.05 No.11(2015), Article ID:60975,6 pages
10.4236/wjcd.2015.511035
The Risk of Milrinone Use in Severe Neurological Insult and Stunned Myocardium
Elazbieta Wirkowski, Lester Kocztorz, Adel Hanna, Colleen Christiansen, Nicole Bielawski
Department of Neuroscience, Winthrop University Hospital, Mineola, New York, USA

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 14 September 2015; accepted 7 November 2015; published 10 November 2015

ABSTRACT
Acute neurological injuries cause catecholamine-induced cardiac damage and can result in or exacerbate preexisting cardiac failure. Milrinone (primacor) has been effective in the treatment of cardiac failure as well as cerebral vasospasm. We would like to present the outcomes of milrinone use in patients with major neurological injuries and preexisting compromised myocardial function. Four patients, age < 60 y/old, three women (two postpartum) and one man, will be presented. Two were diagnosed with a subarachnoid hemorrhage (SAH) coupled with severe vasospasm; one had status epilepticus (SE); and one had cerebral ischemia (CVA) due to carotid artery occlusion. All patients had cardiomyopathy with EF < 35%. All patients were on at least one press or prior to being put on milrinone. Three out of the four patients required an increase in norepinephrine or additional pressors after milrinone was added. All of the patients developed tachycardia, three severe and one mild. In two patients milrinone had to be discontinued due to the persistent hypotension. In our experience, use of milrinone drip in patients with preexisting cardiomyopathy led to hypotension and tachycardia, and brought about the need to increase or add new pressors, which in turn exacerbated catecholamine-induced myocardial injury.
Keywords:
Neurogenic Cardiomyopathy, Pressors, Subarachnoid Hemorrhage

1. Introduction
Acute neurological injuries cause catecholamine-induced cardiac damage and can result in or exacerbate preexisting cardiac failure. Maintaining proper cerebral perfusion pressure (CPP) is instrumental in the prevention of secondary brain injury. Milrinone is a potent phosphodiesterase type III inhibitor with inotropic and vasodilatory as well as possible anti-inflammatory properties on cerebral vessels [1] . These vasodilating and inotropic properties have been used to treat cardiac failure as well as augment cardiac output and improve cerebral perfusion pressure, particularly in its intravenous (I.V.) form [1] - [4] . Milrinone can be given intrarterially (I.A.) directly into the arteries of the brain to treat cerebral vasospasm [5] - [7] . The I.A. form of administration, contrary to I.V. form, has not been shown to cause systemic side effects such as hypotension or tachycardia. I.V. milrinone, when compared to dobutamine, was found to be more potent in increasing cardiac output (CO) and decreasing systemic vascular resistance (SVR), but the effect on stroke volume (SV) was not significantly better. The patients in this study required additional pressors, but hypotension and tachycardia were not reported as severe side effects. In addition, the patients in this study did not have any preexisting cardiac pathology [3] .
We would like to present the effects of milrinone use in four patients with major neurological injuries and preexisting compromised myocardial function that were treated in our ICU.
2. Case Study
2.1. Case #1
A 39-year-old woman was brought in, intubated and unresponsive, to our emergency department following tonic-clonic seizures and cardiac arrest. The patient was resuscitated but the time of returning circulation remained unclear. A brain tumor resection she underwent in her childhood resulted in epilepsy. She was known to be noncompliant with antiseizure medication and was abusing alcohol. She was two months postpartum at the time of this admission.
Because of her past medical history, as well as potential anoxic brain damage, she was admitted to the neuro-ICU (NICU) for further evaluation and treatment. She was unconscious, intubated, flexor extending with clonic tremor in all 4 extremities to noxious stimuli; she had reactive pupils and present corneals.
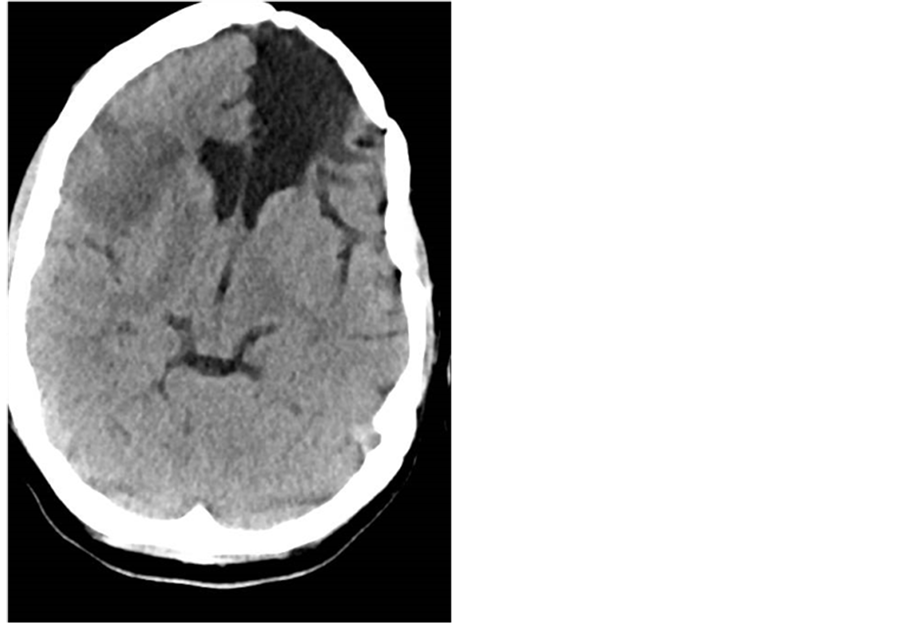
The patient was placed on continuous EEG monitoring, antiseizure medication, and was treated with hypothermia [Figure 1].
Figure 1. Head CT on arrival showing old left frontal encephalomalacia as well as new right MCA ischemia.
After a stat cardiac ECHO showed a left ventricle thrombus and EF < 15%, she was started on heparin and norepinephrine drip. On the second day, she went into cardiogenic shock and was started on milrinone. Her SBP improved but she remained tachycardic. On day four, the patient was weaned off of norepinephrine, but remained on milrinone for one more day. A repeated TTE showed EF of 15% - 20% and more left ventricular thrombi. The patient remained on anticoagulation, the hypothermia protocol was stopped, and the sedation was lifted gradually.
The TTE on day seventeen showed an improvement in EF 30% - 35% and resolution of the thrombi. Twenty-seven days after admission, the patient started to follow simple commands and was discharged to an inpatient rehabilitation facility. She was able to walk, talk and follow commands, but had visual deficits.
2.2. Case #2
This patient was a 44-year-old woman with no past medical history. She arrived in the ER complaining of the worst headache of her life, a brief episode of loss of consciousness witnessed by her husband, dizziness, nausea, vomiting, confusion and seizures. On admission, the patient was oriented to person, place and time, was awake, and had no neurological deficits.
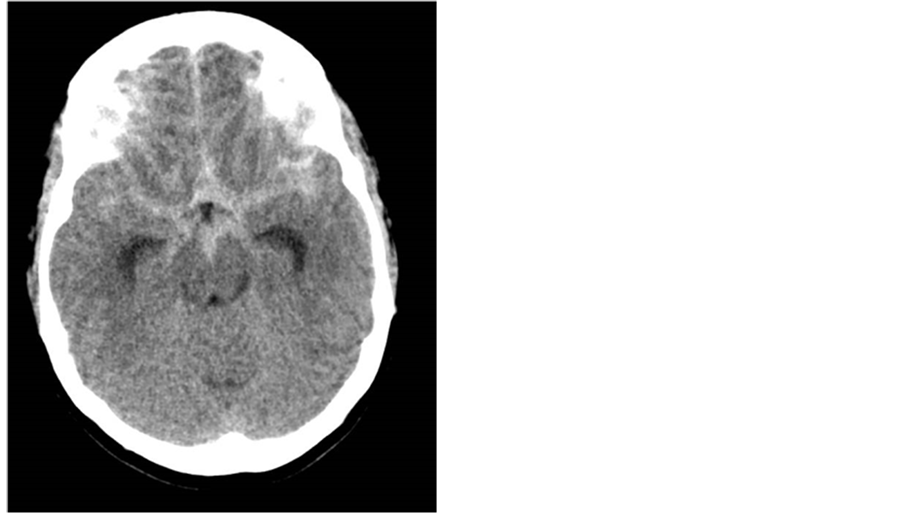
A head CT revealed a subarachnoid hemorrhage, and a formal cerebral angiogram showed an ACOM (anterior communicating artery) aneurysm not amenable to coiling [Figure 2].
Figure 2. Admission head CT showing diffuse SAH with hydrocephalus.
An External Ventricular Drain (EVD) was placed, and the patient was taken for an aneurysmal clipping. She had a baseline cardiac 2DECHO done that showed a normal EF 55% - 60%. The first 3 days after her surgery were uneventful, but on day four, in spite of nimodipine use, blood transfusion to stabilize hematocrit, and albumin to improve oncotic pressure, a Transcranial Doppler (TCD) showed increased velocities. On day five she developed an altered mental status, and on day six her urine output increased. The patient was initially placed on neosynephrine, with norepinephrine added to maintain cerebral perfusion. Her repeated 2DECHO showed decreased left ventricular systolic function with an EF drop to 40% - 50% and anteroseptal wall akinesis.
Due to a further increase in velocities on her TCD, a formal cerebral angiogram was performed next day, which showed moderate vasospasm, which was then treated with intra-arterial verapamil. The same day, the patient became drowsy. The cerebral angiogram was subsequently repeated, and again resulted in intra-arterial administration of verapamil. Due to the use of multiple pressors, the patient’s cardiac condition worsened and she developed Takotsubo syndrome. In an effort to reduce the use of pressors, neosynephrine was weaned off and I.V. milrinone was started. This resulted in a significant drop in blood pressure, EKG changes, a troponin leak, and significant tachycardia, which resulted in the discontinuation of milirinone use. The patient continued to receive verapamil intra-arterially, which led to the resolution of vasospasms. The patient’s medical and neurological condition improved, and she was eventually discharged to an acute rehab facility.
2.3. Case #3
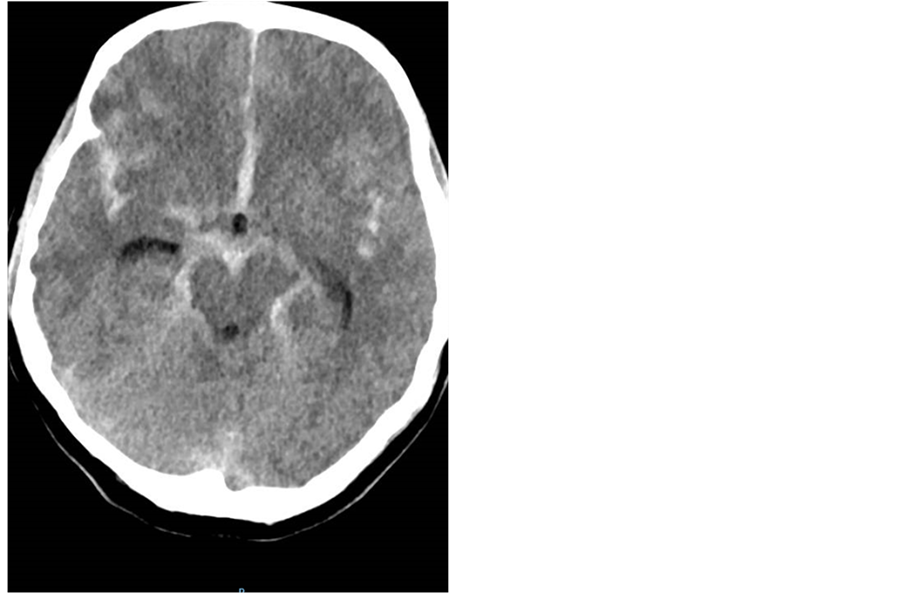
A 41-year-old, 6 months post partum woman with no past medical, presented unresponsive due to a subarachnoid hemorrhage (SAH) [Figure 3].
Figure 3. Head CT showing diffuse SAH with hydrocephalus.
Norepinephrine was started and an EVD (extra ventricular drain) was emergently placed. She had her right P-com aneurysm coiled, after which she woke up and was able to follow simple commands. She had elevated troponins due to catecholamine-related cardiac injury superimposed on possible postpartum cardiomyopathy. Her 2DECHO showed diminished EF of 35% - 40% with anteroseptal wall akinesis as well as inferolateral and basal segment wall hypokinesis. She developed pulmonary neurogenic edema. On day four she developed delayed cerebral ischemia (DCI) and milrinone was started. In spite of her receiving norepinephrine, she became hypotensive and tachycardic up to 160 BPM and vasopressin had to be added to two currently infusing pressors. The patient remained in severe vasospasm with deterioration in mental status in spite of euvolemia and blood pressure augmentation. She underwent an emergent cerebral angiogram to treat cerebral vasospasm with I.A. verapamil, but during the procedure became acutely hypotensive and eventually was pronounced brain dead.
2.4. Case #4
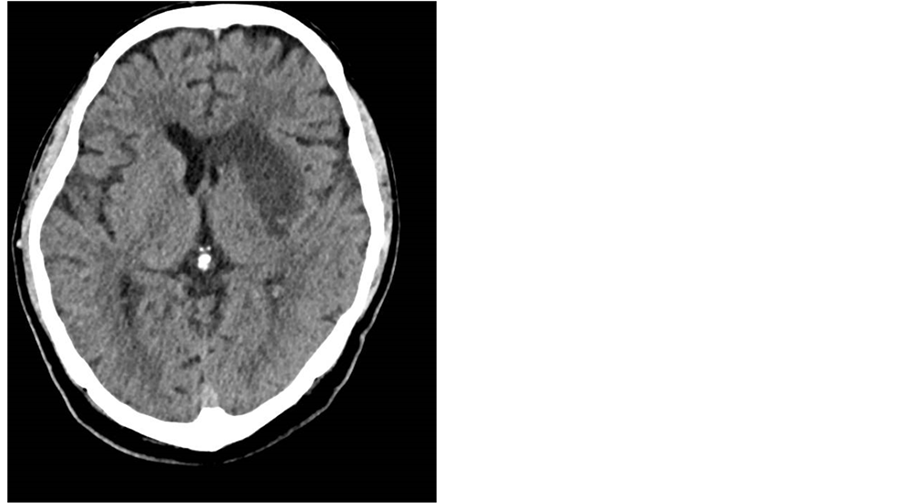
A 53-year-old man with a history of cardiomyopathy of unclear etiology, presented with acute onset right arm weakness and expressive aphasia. He received IV TPA and underwent intra-arterial clot retrieval from the left MCA (middle cerebral artery). Due to his known low EF of 15% - 20% and history of congestive heart failure (CHF), milrinone was started. On the following day he developed atrial fibrillation, with rapid ventricular response (RVR) up to 150 BPM, and hypotension. The patient was anticoagulated, phenylehrine was replaced by neosynephrine and cardizem drip was started. His milrinone drip was discontinued, which resulted in the stabilization of his tachycardia and hypotension. The patient was eventually able to be weaned off all pressors as well as the cardizem drip. His neurological condition improved, and he was discharged to an acute rehabilitation facility [Figure 4].
Figure 4. Head CT showing subacute left MCA ischemic CVA.
3. Discussion
We presented four patients, younger than 60 y/old, three women (two postpartum) and one man. Two were diagnosed with SAH (subarachnoid hemorrhage) with severe delayed cerebral ischemia (DCI), one was status epilepticus (SE) and one presented with occlusion of the carotid artery and cerebral ischemia. All patients had cardiomyopathy with EF < 35%, which worsened during the course of treatment. All patients were on at least one pressor prior to being put on milrinone. Three out of the four patients required an increase in norepinephrine or additional pressors after milrinone was added. All of the patients developed tachycardia, three severe and one mild. In two of the patients, milrinone had to be discontinued due to persistent hypotension and tachycardia. All 3 patients who survived were discharged to rehab with Rankin score of less than 3.
Milrinone, a phosphodiesterase inhibitor, combines inotropic and vasodilatory qualities and seems to be ideal in treatment of vasospasm after SAH or augmenting cardiac output, improving cerebral perfussion in patients with decreased cardiac ejection fraction. Milrinone can be administered intravenously or directly into brain arteries in its intra-arterial form. Most studies describe the benefits of direct intra-arterial use of milrinone in the cerebral vasospasm. Fraticelli et al. used 34 selective intrarterial milrinone to treat 72 vasospastic territories. They observed an increase in arterial diameter, but also a moderate increase in heart rate. Their conclusion was that milrinone administered I.A. was effective and safe in treatment of cerebral vasospasm [2] .
The Montreal Neurologic Institute Protocol for SAH uses I.V. milrinone as the first line medication to treat vasospasm. If there is no effect, only then is intra-arterial administration considered. In the study, milrinone drip was used for an average of 9 days and was tolerated well. Unfortunately, only 4% of their population had CHF on admission, and the sample size was small [1] .
Naidech et al. compared dobutamine and milrinone use in 11 neurogenic stunned myocardium patients with SAH, concluding that milrinone was better in patients who had a severely depressed systolic function, but normal vascular resistance and no hypotension. They did not observe severe tachycardia as a side effect of milrinone treatment. Their patients did not have preexisting cardiac pathology [3] .
Prophylactic use of milrinone in children with low cardiac output due to congenital heart disease was not effective in preventing mortality or low cardiac output syndrome [4] .
Zeiler et al. presented 2 cases with delayed cerebral ischemia post SAH which deteriorated with norepinephrine infusion due to the progression of vasospasm. One patient’s exam improved after starting milrinone and stopping norepinephrine, while the second patient deteriorated clinically in spite of milrinone. Again, hypotension and tachycardia were not mentioned as side effects, and patients did not have preexisting cardiac problems [8] .
4. Conclusion
In our experience, the use of I.V. milrinone led to hypotension and tachycardia, and brought about the need to increase preexisting or add new pressors, which in turn exacerbated catecholamine-induced myocardial injury. It did not prevent or ameliorate severe vasospasm. Our patients had compromised myocardial function prior to being admitted. Two had postpartum cardiomyopathy, and one had congenital cardiomyopathy along with neurogenic cardiomyopathy, all of which could have contributed to the side effects that we observed. We presented the above cases to caution against the use of milrinone for augmentation of CPP (cerebral perfusion pressure) in patients with preexisting cardiacmuscle compromise.
Cite this paper
ElazbietaWirkowski,LesterKocztorz,AdelHanna,ColleenChristiansen,NicoleBielawski, (2015) The Risk of Milrinone Use in Severe Neurological Insult and Stunned Myocardium. World Journal of Cardiovascular Diseases,05,307-312. doi: 10.4236/wjcd.2015.511035
References
- 1. Lannes, M., Teitelbaum, J., Cortes, M. and Cardoso, M. (2002) Milrinone and Homeostasis to Treat Cerebral Vasospasm Associated with Subarachnoid Hemorrhage: The Montreal Neurological Hospital Protocol. Neurocritical Care, 16, 354-362.
http://dx.doi.org/10.1007/s12028-012-9701-5 - 2. Fraticelli, A., Cholley, B.P., Losser, M.-R., et al. (2008) Milrinone for the Treatment of Cerebral Vasospasm after Aneurysmal Subarachnoid Hemorrhage. Stroke, 39, 893-898.
http://dx.doi.org/10.1161/STROKEAHA.107.492447 - 3. Naidech, A., Du, Y., Kreter, K., et al. (2005) Dobutamine versus Milrinone after SAH. Neurosurgery, 56, 21-27.
- 4. Burkhardt, B.E., Rucker, G. and Stiller, B. (2015) Prophylactic Milrinone for the Prevention of Low Cardiac Output Syndrome and Mortality in Children Undergoing Surgery for Congenital Heart Disease. Cochrane Database of Systematic Reviews, 3, CD009515.
http://dx.doi.org/10.1002/14651858.cd009515.pub2 - 5. Sherif, C., Wambacher, B., Loyoddin, M., Karaic, R., et al. (2014) Repeated Combined Endovascular Therapy with Milrinone and Nimodipine for the Treatment of Severe Vasospasm: Preliminary Results. Neurovascular Events after Subarachnoid Hemorrhage, Acta Neurochirurgica Supplement, 120, 203-207.
- 6. Shankar, J.J., dos Santos, M.P., Deus-Silva, L. and Lum, C. (2011) Angiographic Evaluation of the Effect of Intra-Arterial Milrinone Therapy in Patients with Vasospasm from Aneurysmal Subarachnoid Hemorrhage. Neuroradiology, 53, 123-128.
http://dx.doi.org/10.1007/s00234-010-0720-7 - 7. Sadamasa, N., Narumi, K.Y.O., Chin, M. and Yama-gata, S. (2014) Milrinone via Lumbar Subarachnoid Catheter for Vasospasm after Aneurysmal Subarachnoid Hemorrhage. Neurocritical Care, 21, 470-475.
http://dx.doi.org/10.1007/s12028-014-9996-5 - 8. Zeiler, F.A., Silvaggio, J., Kauf-mann, A.M., Gillman, L.M. and West, M. (2014) Norepinephrine as a Potential Aggravator of Symptomatic Cerebral Vaso-spasm: Two Cases and Argument for Milrinone Therapy. Case Reports in Critical Care, 2014, Article ID: 630970.
http://dx.doi.org/10.1155/2014/630970