Open Journal of Gastroenterology
Vol. 2 No. 1 (2012) , Article ID: 17187 , 8 pages DOI:10.4236/ojgas.2012.21001
Alteration of liver N-glycome in patients with hepatocellular carcinoma*
![]()
1Department of Microbiology, Peking University Health Science Center, Beijing, China
2Department for Molecular Biomedical Research, VIB, Ghent, Belgium
3Department for Biomedical Molecular Biology, Ghent University, Ghent, Belgium
Email: #zhuangbmu@126.com, #Cuiying.Chen@dmbr.UGent.be
Received 4 November 2011; revised 8 December 2011; accepted 19 December 2011
Keywords: N-Glycome; DSA-FACE; Fucosylation; HCC; Cirrhosis
ABSTRACT
Purpose: Alteration of liver function during progression of hepatocellular carcinoma (HCC) and cirrhosis affects the serum glycoprotein pattern. In this study, the changes in the N-glycome in liver tissue from patients with hepatocellular carcinoma and cirrhosis caused by hepatitis B virus infection were investigated to find out the relationship between this maker and liver disease. Methods: Twenty patients, 11 with cirrhosis and 9 with hepatocellular carcinoma, and 15 healthy donors were involved in this study. Liver protein N-glycans were profiled using the DSA-FACE technique developed in our laboratory. To further analyze the fucosylation status of these liver glycans Western lectin blots of total liver proteins were performed using Aspergillus oryzae lectin (AOL) as probe, which is a carbohydratebinding protein that recognizes specifically α-1,6-fucosylated glycans. Results: The N-glycome of liver proteins in patients with HBV related HCC and cirrhosis was analyzed. Compared with healthy donors, the N-glycome had significantly less (p < 0.05) high mannose (M8) in both groups of patients. The total core α-1,6-fucosy-lation in total liver glycoproteins was dramatically increased during the progress of hepatocellular carcinoma and cirrhosis compared to the controls. Conclusion: These results show that fucosylation not only increases in serum proteins but also in liver tissue itself of patients with HBV related HCC and cirrhosis.
1. INTRODUCTION
Hepatocellular carcinoma (HCC) is a primary cancer of the liver and is one of the leading causes of death worldwide [1]. HCC arises most commonly in cirrhotic livers following infection with hepatitis B virus (HBV) or hepatitis C virus (HCV) [2,3]. Like any other cancer, HCC develops when a mutation in the cellular machinery causes the cell to replicate at a higher rate and/or results in the cell avoiding apoptosis. In particular, chronic infections with HBV and/or HCV could evolve to malignancy by repeatedly causing the immune system to attack the liver cells.
Hepatocarcinogenesis is a complex process associated with accumulation of genetic and epigenetic changes that occur during initiation, promotion, and progression of the disease. Though extensive research over the past decade has identified a number of molecular biomarkers as well as cellular networks and signaling pathways affected in liver cancer [4], the pathophysiology of HCC is not understood clearly, but underlying liver dysfunction is a known predisposing condition.
Protein N-glycosylation, the most common co-translational modification, is the controlled enzymatic addition of sugars to a protein. N-glycans are synthesized in the endoplasmic reticulum (ER) and Golgi by the sequential addition of oligosaccharides, such as GlcNAc and fucose molecules, by the corresponding oligosaccharyl-transferases [5,6]. The N-glycan profile in serum is related to the glycosylation pathway in liver cells and B-cells, and changes in the levels of the enzymes of this pathway could lead to modifications in both the core and outer arm structures of glycans.
Because the sugar chains of glycoproteins are important for maintaining the ordered “social behavior” of differentiated cells in multicellular organisms, alterations in these sugar chains contribute to the molecular basis of abnormalities, such as invasion of tumor cells into the surrounding tissues and their metastasis. Alterations in the N-linked sugar chains are indeed found in various tumors [8-11]. Moreover, our and other studies recently showed that certain changes in the N-glycans of total serum glycoproteins are indicative of human HCC [7,8], non-alcoholic steatohepatitis (NASH) [9], liver fibrosis [10], and cirrhosis [11]. These observations are in line with the fact that a large fraction of serum proteins is produced by hepatocytes.
To further explore the glycoproteome, and to clearly link N-glycosylation changes in serum to the liver, not serum proteins but liver proteins themselves are studied. This has been the bottleneck of many studies since liver tissue is far harder to collect than serum and also technically liver glycosylation is more difficult to analyze. In this study, the N-linked glycans in liver samples of HBV-infected cirrhosis and HCC patients were analyzed. Our results demonstrate glycosylation alterations and increased fucosylation in glycans of the liver in patients with HCC, clearly linking alterations in serum N-glycome to the liver.
2. MATERIALS AND METHODS
2.1. Patients and Controls
This study included 20 patients (11 with cirrhosis and 9 with hepatocellular carcinoma caused by HBV infection) and 15 liver donors, all of whom underwent percutaneous liver biopsy at the Beijing Youan Hospital between 2008 and 2009. The study was approved by the Ethics Committee of Peking University Health Science Centre and by the Ethics Committee of Beijing Youan Hospital. Informed consent was obtained from all the volunteers.
2.2. Laboratory Tests
The main clinical and biological data of the patients are summarized in Table 1. All patients were diagnosed as HBV-related cirrhosis and HBV infection was diagnosed by serological detection of HBsAg, anti-HBsAg (HBsAb), HBeAg, anti-HBeAg (HbeAb), anti-HBcAg (HBcAb) and HBV DNA. The extent of liver damage was assessed by measurement of alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, albumin, total serum protein, and γ-glutamyltransferase (GGT).
The diagnosis of cirrhosis was made by histological examination, imaging procedures, and several liver function tests. Liver samples were evaluated independently by two experienced hepatopathologists who were unaware of the glycomics results. Liver cirrhosis patients (n = 10) were staged according to the Child-Pugh classifycation. Cirrhosis patients with HCC (n = 9) were diagnosed by a combination of histological examination of biopsies, ultrasonography and/or computed tomographic scanning, and measurement of AFP.
2.3. Preparation of Proteins from Liver Biopsies
About 50 mg of liver tissue was disrupted in a mixer on ice in 250 µl of lysis buffer: 1% NP40, 200 mM NaCl, 10 mM Tris-HCl pH 7.5, 5 mM EDTA, 10% glycerol,
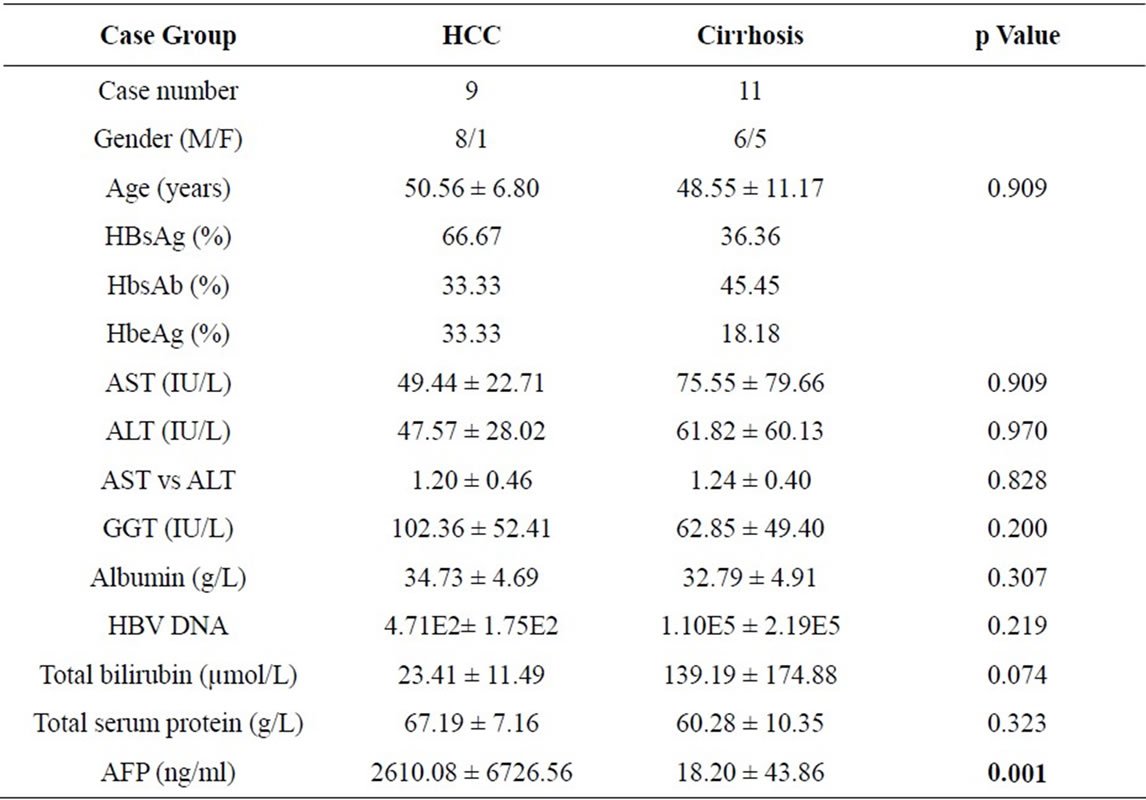
Table 1. Main clinical and biological data of the patients with HBV related cirrhosis and HCC.
and protease inhibitor cocktail (Complete, EDTA-free, Roche). Lysates were spun down for 20 min at 14,000 rpm at 4˚C. Supernatant was collected and the concentration of the proteins measured with a BCA protein Assay Kit (Pierce). Samples containing 150 µg proteins were concentrated in a vacuum dryer to 5 µl and used in DSA-FACE. For sialidase digestion, 1 µl of APTS-labeled sugar was used.
2.4. Processing Liver Tissue Samples for Protein N-Glycome Analysis
The N-glycans present on the proteins in 5 ml of liver protein extract were released, labeled and analyzed by DSA-FACE as described previously [12,13], but with some minor modification. Briefly, 5 ml of liver protein was placed in a PCR tube containing 2 ml of buffer (10 mM NH4HCO3, pH 8.3, 5% SDS) and 3 ml of water. The tube was heated at 95˚C for 5 min and cooled for 15 min at 4˚C in a PCR thermocycler. Then, 3 ml of peptideN-glycosidase F (PNGase F) was added (2.2 U/ml in 10 mM NH4HCO3, pH 8.3, containing 3.33% NP40) (New England Biolabs, UK). The tubes were incubated in the thermocycler at 37˚C for 3 h, cooled to 4˚C, and then 100 ml of water was added. Six ml of the resulting solution was transferred to a new PCR tube and evaporated to dryness at 60˚C in the thermocycler (lid open). N-glycans were derivatized by adding 1 µl of labeling solution (1:1 mixture of 20 mM APTS; Molecular Probes, Eugene, OR) in 1.2 M citric acid and 1 M NaCNBH3 in dimethyl sulfoxide. The tightly closed tubes were incubated at 37˚C for 16 h. The APTS labeling reaction was stopped by adding 100 ml of water. Then, 2 µl of APTS-labeled solution was transferred to a new PCR tube for digestion with 0.25 mU Arthrobacter ureafaciens sialidase (Roche, Mannheim, Germany) in 5 µl of 5 mM NH4Ac, pH 5.0. The tube was incubated overnight at 37˚C in the thermocycler. 40 ml water was added and 10 ml of labeled N-glycans were analyzed by DNA Sequencer AssistedFluorophore Assisted Carbohydrate Electrophoresis (DSAFACE) using a capillary electrophoresis (CE)-based ABI 3130 sequencer. Data were analyzed using the GeneMapper v3.7 software (Applied Biosystems, Foster city, CA). The heights of the 10 peaks that were detected in all the samples were measured to obtain a numerical description of the profiles.
For structural analysis of APTS-labeled serum N-glycans, appropriate amounts were digested with exoglycosidases: Streptococcus pneumonia β-1,4-galactosidase (2 mU/digest), jack bean β-N-acetylhexosaminidase (10 mU/ digest), S. pneumoniae β-N-acetylhexosaminidase (10 mU/ digest), bovine kidney α-fucosidase (2 mU/digest) and almond meal α-1,3/4-fucosidase (1 μU/digest) (Prozyme, San Leandro, CA) and a-1,2-mannosidase from Trichoderma reesei (home made [14]). DSA-FACE was used to analyze the digestion products.
2.5. Structure Analysis of N-Glycans Using Exoglycosidase Digestion
The APTS-labeled desialylated N-glycans of 1 ml obtained from the DSA-FACE procedure was digested overnight with various mixtures of exoglycosidases in 10 mM NH4Ac (pH 5.0) at 37˚C. The products were separated by electrophoresis on an ABI 3130. The exoglycosidases were α-1,3/4-Fucosidase (almond meal) (Glyko, Novato, CA, USA), which cleaves non-reducing terminal α-1,3/4-linked fucose, and α-1,6-Fucosidase (bovine kidney) (Glyko, Novato, CA, USA), which cleaves non-reducing terminal α-1,6-linked fucose.
2.6. Western Lectin Blot
Samples of 25 µg proteins extracted from frozen liver tissues were separated by electrophoresis in 10% sodium dodecyl sulfate-polyacrylamide gel. The proteins were then transferred to a nitrocellulose membrane for lectin blot analysis. The membranes were blocked overnight at 4˚C with 3% bovine serum albumin (BSA) in Tris-buffered saline (TBS: 140 mM NaCl, 10 mM Tris-HCl) and then incubated for 1 h at room temperature with 5 µg/ml of biotinylated Aspergillus oryzae L-fucose-specific lectin (AOL) (Funakoshi Co., Ltd., Tokyo, Japan) in TBST buffer (TBS containing 0.05% Tween 20). After 4 washes of 5 min each with TBST, the membranes were incubated with 1/1000 diluted horseradish peroxidase-conjugated Streptavidin (R&D Systems, Minneapolis, MN, USA) for 1 h at room temperature. The membranes were washed 4 times again with TBST and developed with an ECL system (Amersham Biosciences Inc., Piscataway, NJ, USA).
2.7. Statistical Analysis
Statistical analyses were performed with SPSS 15.0 for Windows software (SPSS, Chicago, IL, USA). Results are presented as means ± SD. All reported p-values are two-tailed, using a t-test for independent samples.
3. RESULTS
3.1. Profiles of N-Glycans in Liver Tissue
Mature glycoproteins in the liver are synthesized in the ER from a variety of oligomannose N-linked glycans containing 5 - 9 mannose residues (high-mannose) and then transported to the Golgi. Further removal of mannose residues in the Golgi leads to a “core” structure containing three mannose and two N-acetylglucosamine residues (GlcNAc). Elongation of this core with a variety of monosaccharides leads to formation of the three major classes of N-linked saccharides: high-mannose oligosaccharides, complex oligosaccharides (in which antennae are initiated with GlcNAc), and hybrid oligosaccharides (in which only mannose is attached to the Man-α-1,6- arm of the core and one or two antennae are on the Man- α-1,3-arm).
To verify the structure of liver N-glycans, we compared and overlaid liver N-glycan profile to that of serum and RNase B. As shown in Figure 1, RNase B contains only five different high mannose 5, 6, 7, 8 and 9 (represented as M5, M6, M7, M8 and M9), while serum has most complex oligosaccharides: biantennary N-glycans (NGA2F, NA2 and NA2F) and triantennary N-glycans (NA3 and NA3F) as our previous report [7]. We found
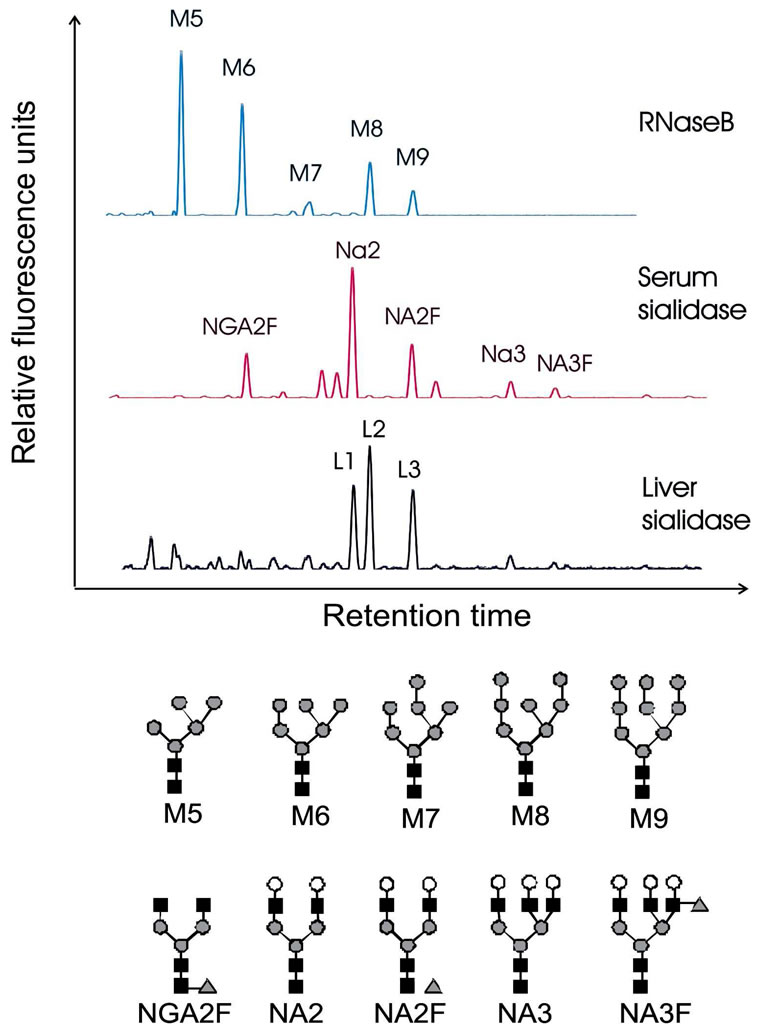
Figure 1. Desialylated N-glycan profiles from liver glycoproteins (lower panel). N-glycan profiles from RNase B (upper panel) and serum (middle panel) were used as controls. RNase B contains varies high mannose from M5 to M9. Serum contains varies complex N-glycans. High mannose presented in RNase B and the most abundant glycans detected in serum are indicated. The structures of the N-glycan peaks are shown below the panels. NGA2F is an agalacto, core-α-1,6-fucosylated biantennary glycan; NA2 is a bigalacto, biantennary glycan; NA2F is a bigalacto, core-α-1,6-fucosylated biantennary; NA3 is tri-antennary; NA3F is a branching a-1,3-fucosylated triantennary. The symbols used in the structural formulas are the following: “black square” stands for N-acetylglucosamine (GlcNAc); “open circle” stands for galactose; “grey triangle” stands for a-1,3/6-linked fucose; “grey circle” stands for mannose.
that the pattern of the desialylated N-glycans in liver N-glycoproteins was different from that of the serum N-glycan finger print (Figure 1). There were three most abundant N-glycans, represented as L1, L2 and L3 (Figure 1). Overlaying with N-glycan profiles of serum and RNase B, L1 migrated at the same site as NA2 and L2, and as high as mannose 8 (M8), while L3 run as high as mannose 9 (M9) and NA2F. These structures were further studied by using exoglycosidases. If L2 and L3 are high mannose N-glycans (M8 and M9, respectively), they shall disappear and generate M5 when being digested by α-1,2-mannosidase. Indeed, L2 was totally disappeared and shifted three residues forwards to M5, confirming L2 represent high mannose 8 (M8). However, L3 was diminished partially and shifted four residues to M5 (Figure 2). Following digestion with bovine kidney
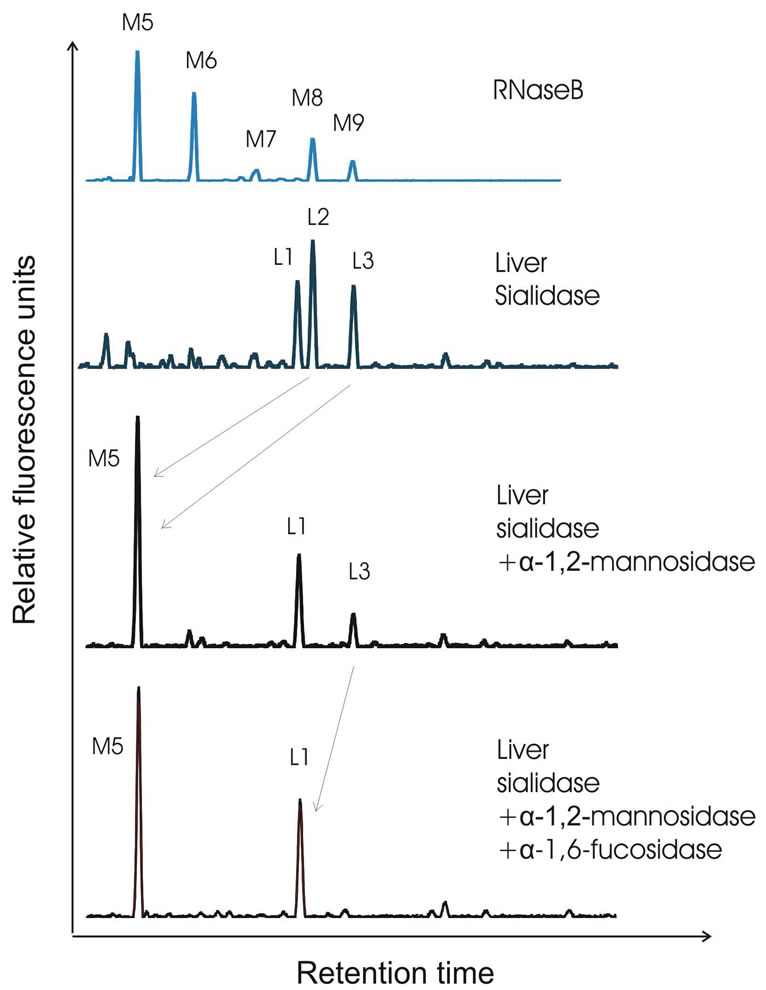
Figure 2. Exoglycosidase sequencing of N-glycans from liver glycoproteins. The upper panel shows profile from RNase B as high mannose reference. The 2nd panel shows desialylated Nglycan profile from liver. The 3rd panel shows separation of desialylated N-glycans digested with α-1,2-mannosidase. And the 4th panel shows separation of desialylated N-glycans digested with α-1,2-mannosidase and bovine kidney α-1,6-fucosidase. Peak L2 contains high mannose 8 structure (M8) as it shifted three mannoses forwards after α-1,2-mannosidase treatment. Peak L3 is mix of two structures: high mannose glycan (M9) as it shifted four mannoses forwards after α-1,2-mannosidase treatment and α-1,6-fucosylated structure as it shifted one residue forwards to L1. The arrow lines indicate the changes in the glycan peaks due to glycosidase digestion.
α-1,6-fucosidase which can remove only one α-1,6- fucose from the core N-acetylglucosamine, remained part of L3 was then disappeared and shifted one residue forward to L1, indicating L3 contains a mixture of M9 and core-fucosylated N-glycan (Figure 2). It was further verified that the structures of L1 and L3 were hybrid biantennary N-glycans of NA2 and NA2F, as they were shifted two residues forward after b-galactosidase digestion (Figure 3). The levels of L1, L2 and L3 were not changed after treatment with N-acetylhexosaminidase, indicating they contain neither agalacto nor branched fucose structures. Taking together, L1 and L2 represented NA2 and M8, respectively. As L3 was digested partially with α-1,2-mannosidase and α-1,6 fucosidase, it is clear that L3 represented a mixture of two structures: high mannose (Man9) and a-1,6 fucosylated N-glycan NA2F.
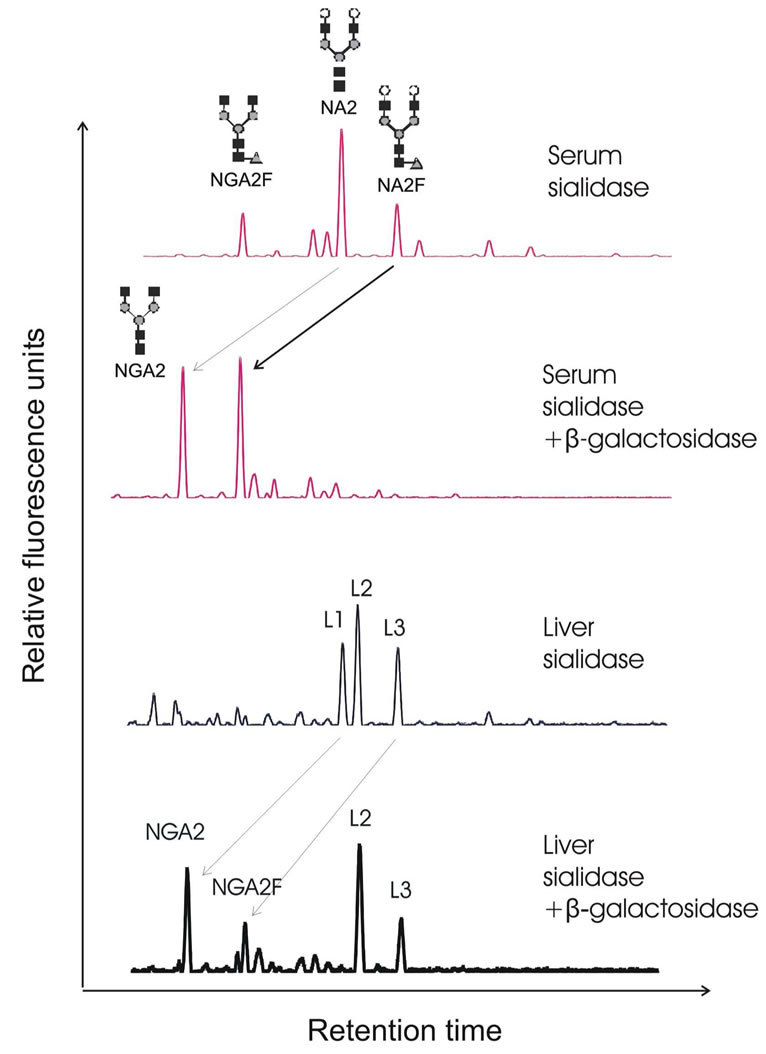
Figure 3. Exoglycosidase sequencing of N-glycans from liver glycoproteins. The upper two panels show separations of desialylated serum N-glycans and digested with b-galactosidase as reference. The lower two panels show separations of desialylated liver N-glycans and digested with b-galactosidase. Peak L1 and L3 shifted two residues forwards to NGA2 and NGA2F after removal of two galactoses. The arrows indicate the changes in the glycan peaks due to glycosidase digestion. The symbols used in the structural formulas are the following: “black square” stands for N-acetylglucosamine (GlcNAc); “open circle” stands for galactose; “grey triangle” stands for α-1,6-linked fucose; “grey circle” stands for mannose.
3.2. Altered Glycosylation in the Liver of Patients
We previously reported changes in serum N-linked glycosylation in conjunction with the development of hepatocellular carcinoma. To see whether N-glycans are altered in liver tissue during HCC development, we determined the liver desialylated N-glycan profiles in HCC patients (n = 9), cirrhosis patients (n = 11) and healthy liver donors (n = 15). The N-glycan profile was altered in the livers of cirrhosis and HCC patients compared to the donors (Figure 4(a)). The level of L2, which is a high mannose N-glycan (M8), was significantly (p < 0.0001) decreased in the liver of cirrhosis and HCC patients compared to the donors (Figure 4(b)). This result shows that glycosylation is altered in liver tissue during development of HCC.

Figure 4. Altered N-glycosylation in liver during progress of cirrhosis and HCC. Desialylated N-glycan profiles in the liver revealed a decreased level of L2 in cirrhosis and HCC group (a). The value of log (L1/L2) showed significantly difference between control, cirrhotic and HCC groups (b). The vertical axis represents the glycan values of log (L1/P2). Error bars represent 95% confidence intervals for means. Statistical significance of differences between groups is indicated by the p value.
3.3. Increased Fucosylation during Progression of HCC
Fucosylation is one of the most common modifications of oligosaccharides on glycoproteins and glycolipids. To determine the level of fucosylation in HCC liver, the probes of lectins, which are carbohydrate-binding or carbohydrate cross-linking proteins, was used for western blot analyses. A probe of AAL (Aleuria aurantia lectin) isolated from Aleuria aurantia mushrooms, which binds preferentially to fucose linked (α-1,6) to N-acetylglucosamine, or to fucose linked (α-1,3) to N-acetyllactosamine-related structures was used. The binding of AAL to liver glycoproteins was considerably increased in livers with HCC and cirrhosis compared to control liver, indicating that fucosylation increases during progression of HCC and cirrhosis (Figure 5(a)). To verify whether this was due to an increase in core α-1,6-fucosylation of N-glycans, we performed western blot with a probe of AOL (Aspergillus oryzae), which has strongest preference for the α-1,6-fucosylated glycans [15]. The result showed a higher level of total core fucose residues in livers with cirrhosis and HCC than in healthy control livers (Figure 5(b)). The total glycoproteins were detected using Concanavalin A (ConA) lectin, which binds specifically to certain structures in glycoproteins, mainly internal and nonreducing terminal alpha-mannosyl groups. There was no significantly different total in glycan proteins among control, HCC and liver cirrhosis groups (Figure 5(c)).

Figure 5. SDS-PAGE of proteins from liver and western blots probed with lectin. The upper panel shows a western AAL and AOL blots of serum proteins (a) and (b). The lower panel shows a western blot of liver proteins probed with ConA (c) and a gel stained with Coomassie Blue (CBB) of the same proteins (d). The lanes are as follows: 1 - 3: controls; 4 - 6: cirrhosis; 7 - 9: HCC. The data were reproducible in three independent experiments. The molecular weight of proteins were indicated.
3.4. Correlation of the Fucosylation Level with AFP
The AST/ALT ratio is considered a sensitive marker of cirrhosis progression in viral hepatitis [16]. γ-glutamyltransferase (GGT) has also been shown to have good sensitivity at the stage when viral hepatitis starts to cause structural damage [17]. However, we did not found significant differences between the HCC and cirrhosis groups in AFP, GGT or the AST/ALT ratio (Table 1). Pearson correlation showed that log (L1/L2) was not correlated with GGT and AST/ALT ratio, but positively correlated with AFP (r = 0.646; p < 0.0001).
4. DISCUSSION
Alteration of liver function during progression of HCC and cirrhosis affects the metabolism of glycoproteins and their concentration and secretion [18]. By analyzing the N-glycan profiles of liver tissue, we found that fucosylation increased in the livers of HCC and cirrhosis patients.
Glycoproteins are first formed in the ER from a variety of oligomannose N-linked glycans (high-mannose) and then transported to the Golgi. Removal of some mannose residues and further modification with a variety of different monosaccharides in the Golgi leads to form the three major classes of N-linked saccharides: high-mannose oligosaccharides, complex oligosaccharides and hybrid oligosaccharides. Whether an N-glycan is high-mannose or complex is thought to depend on its accessibility to the various saccharide-modifying proteins in the Golgi. This study detected a decrease in high-mannose N-glycans (M8 and M9) in the livers of HCC patients in comparison to healthy donors. This observation suggests that original high-mannose forms are more likely to be trimmed and then modified by the addition of other types of groups during progression of HCC. The altered modification of glycoproteins during progression of HCC could affect the secretion of N-glycosylated proteins by liver cells.
The appearance of core fucosylation on many glycolproteins present in the circulation correlates with HCC [19]. It has been reported that core-1,6-fucosylation of haptoglobin [20], α-fetal protein (AFP) [21] and α1 antitrypsin [22] in serum is increased in liver diseases. Our previous study also showed alteration of the total serum N-glycome in HCC [7,23] and cirrhosis [11,24]. Therefore, the detection of an increase in total core α-1,6- fucose in the liver of HCC and cirrhosis patients compared to healthy volunteers, which indicates that the alteration in the total serum N-glycome in liver HCC and cirrhosis is, at least partly, due to modification of Nglycoproteins in the liver. The alteration of fucosylation could affect secretion of hepatic glycoproteins into the bile duct. Fucosyltransferase (Fut8) knockout mice have a lower level of α1-antitrypsin and α1 acid glycolprotein in bile than wild type mice, which suggests that core fucosylation could be a signal for secretion in the bile duct [25]. Increased fucosylation in HCC has been attributed to an increase in both the α-1-6 fucosyltransferase (Fut8) and its substrate, GDP-fucose [26]. However, expression of Fut8 and GDP-fucose synthesis in liver were barely detectable by qPCR. Nevertheless, fucosylation is strongly associated with cancer, and more information about the mechanism involved and detection of other fucosylated glycoproteins would be highly desirable.
5. ACKNOWLEDGEMENTS
We thank Dr. Amin Bredan for editing the manuscript. This work was supported by a grant from Ghent University (BOF No. 01106205), Flanders-China Bilateral project (011S605), and was also funded by the National Science Foundation of China (Grant No. 30771996) and the National Projects on Major Infectious Diseases, Ministry of Science and Technology of China (Grant No.2008ZX10002-012).
REFERENCES
- Bruix, J., Boix, L., Sala, M. and Llovet, J.M. (2004) Focus on hepatocellular carcinoma. Cancer Cell, 5, 215-219. doi:10.1016/S1535-6108(04)00058-3
- Liaw, Y.F. (2005) Prevention and surveillance of hepatictis B virus-related hepatocellular carcinoma. Seminars in Liver Disease, 25, 40-47. doi:10.1055/s-2005-915649
- Koike, K. (2005) Molecular basis of hepatitis C virus-associated hepatocarcinogenesis: Lessons from animal model studies. Clinical Gastroenterology and Hepatology, 3, S132-S135. doi:10.1016/S1542-3565(05)00700-7
- Aravalli, R.N., Steer, C.J. and Cressman, E.N. (2008) Molecular mechanisms of hepatocellular carcinoma. Hepatology, 48, 2047-2063. doi:10.1002/hep.22580
- Dwek, R.A. (1998) Biological importance of glycosylation. Developments in Biological Standardization, 96, 43- 47.
- Trombetta, E.S. and Parodi, A.J. (2005) Glycoprotein reglucosylation. Methods, 35, 328-337. doi:10.1016/j.ymeth.2004.10.004
- Liu, X.E., Desmyter, L., Gao, C.F., Laroy, W., Dewaele, S., Vanhooren, V., Wang, L., Zhuang, H., Callewaert, N., Libert, C., Contreras, R. and Chen, C. (2007) N-glycomic changes in hepatocellular carcinoma patients with liver cirrhosis induced by hepatitis B virus. Hepatology, 46, 1426-1435. doi:10.1002/hep.21855
- Goldman, R., Ressom, H.W., Varghese, R.S., Goldman, L., Bascug, G., Loffredo, C.A., Abdel-Hamid, M., Gouda, I., Ezzat, S., Kyselova, Z., Mechref, Y. and Novotny, M.V. (2009) Detection of hepatocellular carcinoma using glycomic analysis. Clinical Cancer Research, 15, 1808- 1813. doi:10.1158/1078-0432.CCR-07-5261
- Chen, C., Schmilovitz-Weiss, H., Liu, X.E., Pappo, O., Halpern, M., Sulkes, J., Braun, M., Cohen, M., Barak, N., Tur-Kaspa, R., Vanhooren, V., Van Vlierberghe, H., Libert, C., Contreras, R. and Ben-Ari, Z. (2009) Serum protein N-glycans profiling for the discovery of potential biomarkers for nonalcoholic steatohepatitis. Journal of Proteome Research, 8, 463-470. doi:10.1021/pr800656e
- Vanderschaeghe, D., Laroy, W., Sablon, E., Halfon, P., Van Hecke, A., Delanghe, J. and Callewaert, N. (2009) GlycoFibroTest is a highly performant liver fibrosis biomarker derived from DNA sequencer-based serum protein glycomics. Molecular & Cellular Proteomics, 8, 986- 994. doi:10.1074/mcp.M800470-MCP200
- Vanderschaeghe, D., Laroy, W., Sablon, E., Halfon, P., Van Hecke, A., Delanghe, J. and Callewaert, N. (2004) Noninvasive diagnosis of liver cirrhosis using DNA sequencer-based total serum protein glycomics. Nature Medicine, 10, 429-434. doi:10.1038/nm1006
- Vanhooren, V., Laroy, W., Libert, C. and Chen, C. (2008) N-Glycan profiling in the study of human aging. Biogerontology, 9, 351-356. doi:10.1007/s10522-008-9140-z
- Laroy, W., Contreras, R. and Callewaert, N. (2006) Glycome mapping on DNA sequencing equipment. Nature Protocols, 1, 397-405. doi:10.1038/nprot.2006.60
- Maras, M., Callewaert, N., Piens, K., Claeyssens, M., Martinet, W., Dewaele, S., Contreras, H., Dewerte, I., Penttilä, M. and Contreras, R. (2000) Molecular cloning and enzymatic characterization of a Trichoderma reesei 1,2-alphaD-mannosidase. Journal of Biotechnology, 77, 255-263. doi:10.1016/S0168-1656(99)00222-9
- Matsumura, K., Higashida, K., Ishida, H., Hata, Y., Yamamoto, K., Shigeta, M., Mizuno-Horikawa, Y., Wang, X., Miyoshi, E., Gu, J. and Taniguchi, N. (2007) Carbohydrate binding specificity of a fucose-specific lectin from Aspergillus oryzae: A novel probe for core fucose. The Journal of Biological Chemistry, 282, 15700-15708. doi:10.1074/jbc.M701195200
- Giannini, E., Risso, D. and Testa, R. (2001) Transportability and reproducibility of the AST/ALT ratio in chronic hepatitis C patients. The American Journal of Gastroenterology, 96, 918-919. doi:10.1111/j.1572-0241.2001.03646.x
- Silva, I.S., Ferraz, M.L., Perez, R.M., Lanzoni, V.P., Figueiredo, V.M. and Silva, A.E. (2004) Role of gammaglutamyl transferase activity in patients with chronic hepatitis C virus infection. Journal of Gastroenterology and Hepatology, 19, 314-318. doi:10.1111/j.1440-1746.2003.03256.x
- Miyoshi, E., Moriwaki, K. and Nakagawa, T. (2008) Biological function of fucosylation in cancer biology. Journal of Biochemistry, 143, 725-729.
- Comunale, M.A., Lowman, M., Long, R.E., Krakover, J., Philip, R., Seeholzer, S., Evans, A.A., Hann, H.W., Block, T.M. and Mehta, A.S. (2006) Proteomic analysis of serum associated fucosylated glycoproteins in the development of primary hepatocellular carcinoma. Journal of Proteome Research, 5, 308-315. doi:10.1021/pr050328x
- Miyoshi, E. and Nakano, M. (2008) Fucosylated haptoglobin is a novel marker for pancreatic cancer: Detailed analyses of oligosaccharide structures. Proteomics, 8, 3257-3262. doi:10.1002/pmic.200800046
- Breborowicz, J., Mackiewicz, A. and Breborowicz, D. (1981) Microheterogeneity of alpha-fetoprotein in patient serum as demonstrated by lectin affino-electrophoresis. Scandinavian Journal of Immunology, 14, 15-20. doi:10.1111/j.1365-3083.1981.tb00179.x
- Naitoh, A., Aoyagi, Y. and Asakura, H. (1999) Highly enhanced fucosylation of serum glycoproteins in patients with hepatocellular carcinoma. Journal of Gastroenter ology and Hepatology, 14, 436-445. doi:10.1046/j.1440-1746.1999.01882.x
- Fang, M., Zhao, Y.P., Zhou, F.G., Lu, L.G., Qi, P., Wang, H., Zhou, K., Sun, S.H., Chen, C.Y. and Gao, C.F. (2010) N-glycan based models improve diagnostic efficacies in hepatitis B virus-related hepatocellular carcinoma. International Journal of Cancer, 127, 148-159. doi:10.1002/ijc.25030
- Gui, H.L., Gao, C.F., Wang, H., Liu, X.E., Xie, Q., Dewaele, S., Wang, L., Zhuang, H., Contreras, R., Libert, C. and Chen, C. (2010) Altered serum N-glycomics in chronic hepatitis B patients. Liver International, 30, 259-267. doi:10.1111/j.1478-3231.2009.02170.x
- Nakagawa, T., Uozumi, N., Nakano, M., Mizuno-Horikawa, Y., Okuyama, N., Taguchi, T., Gu, J., Kondo, A., Taniguchi, N. and Miyoshi, E. (2006) Fucosylation of N-glycans regulates the secretion of hepatic glycoproteins into bile ducts. The Journal of Biological Chemistry, 281, 29797-29806. doi:10.1074/jbc.M605697200
- Noda, K., Miyoshi, E., Gu, J., Gao, C.X., Nakahara, S., Kitada, T., Honke, K., Suzuki, K., Yoshihara, H., Yoshikawa, K., Kawano, K., Tonetti, M., Kasahara, A., Hori, M., Hayashi, N. and Taniguchi, N. (2003) Relationship between elevated FX expression and increased production of GDP-L-fucose, a common donor substrate for fucosylation in human hepatocellular carcinoma and hepatoma cell lines. Cancer Research, 63, 6282-6289.
NOTES
*This work was supported by a grant from Ghent University (BOF No. 01106205), Flanders-China Bilateral project (011S605), and was also funded by the National Science Foundation of China (Grant No. 30771996) and the National Projects on Major Infectious Diseases, Ministry of Science and Technology of China (Grant No. 2008ZX 10002-012).
#Corresponding author.

