Open Journal of Radiology
Vol.05 No.01(2015), Article ID:54247,6 pages
10.4236/ojrad.2015.51001
Transient Hypertensive Response to CO2 Aortography in a Patient with Pheochromocytoma
Nimesh Patel1, Kyung J. Cho2*
1University of Michigan Medical School, Ann Arbor, MI, USA
2Department of Radiology, Division of Vascular & Interventional Radiology, University of Michigan Health System, Ann Arbor, MI, USA
Email: *kyungcho@med.umich.edu
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 5 February 2015; accepted 23 February 2015; published 26 February 2015
ABSTRACT
We report on a 15-year-old male with 4-year history of hypertension and retroperitoneal masses referred to Interventional Radiology to evaluate the nature of the persistent masses and renal artery stenosis. Angiographic evaluation began with CO2aortogram during which the patient had a hypertensive response with bradycardia and arrhythmia to the intra-aortic injection of CO2. His hypertension, bradycardia, and arrhythmia responded to an intravenous nitroglycerin drip without administration of alpha or beta blockers. Due to this response and neovascularity of the mass, selective venous sampling of catecholamines and renin was performed which confirmed functioning paragangliomas and hemodynamically significant stenosis of left accessory renal artery, respectively. This is the first known case of hypertensive response to CO2 aortography in a patient with unsuspected functioning paragangliomas. We propose that patients with known or suspected diagnosis of catecholamine-secreting tumors undergoing CO2 angiography should be considered for pre-procedural treatment with alpha and beta blockage or have the appropriate medication on hand with close monitoring in the event of hypertensive response.
Keywords:
CO2, Aortography, Paraganglioma, Caval Venous Sampling, Renal Artery Stenosis

1. Introduction
CO2 has been increasingly used as a contrast agent since the advent of digital subtraction angiography has found to be comparable in accuracy to iodinated contrast agents [1] . One of the main advantages has its relative safety. It has no renal or hepatic toxicity even when selectively injected into the respective arterial or venous systems and is a natural byproduct of metabolism, therefore cannot cause hypersensitivity reaction [2] [3] .
Pheochromocytoma is an uncommon neuroendocrine tumor originating from the chromaffin cells in the medulla of the adrenal gland that secretes excessive amounts of catecholamines. Extra-adrenal pheochromocytomas (usually described as extra-adrenal paragangliomas) originate in the ganglia of the sympathetic nervous system that also produce excessive amounts of catecholamines, predominantly norepinephrine. A pheochromocytoma may cause a hypertensive crisis. It is usually benign but can be malignant in 10% of cases. Surgical removal of the tumor can cure hypertension associated with the tumor.
Hypertensive crisis induced by intra-arterial injection of ionic contrast medium in patients with catecholamine-secreting tumors is a well-known phenomenon [4] [5] . Furthermore, certain agents, such as glucagon, have been reported to induce catecholamine release from pheochromocytomas with subsequent multi-organ injury [6] . Later studies have shown that non-ionic contrast does not have this same effect when given intravenously during CT scans [3] [6] [7] .
To our knowledge, we are reporting the first case of CO2 angiography resulting in a hypertensive response with bradycardia and arrhythmia in a patient with catecholamine-secreting tumor.
2. Case Summary
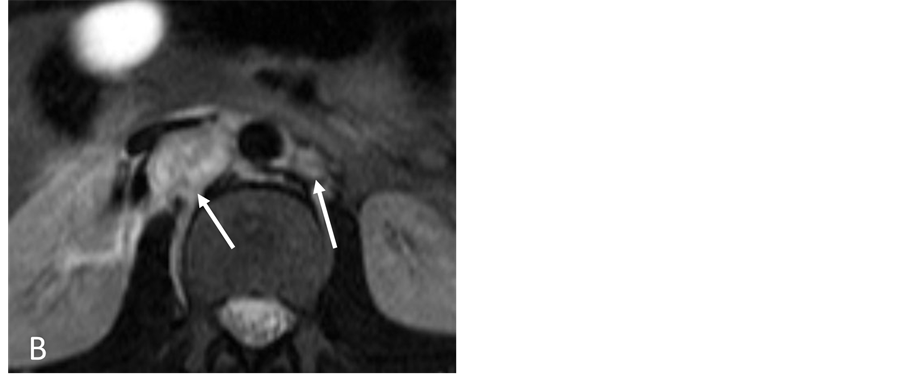
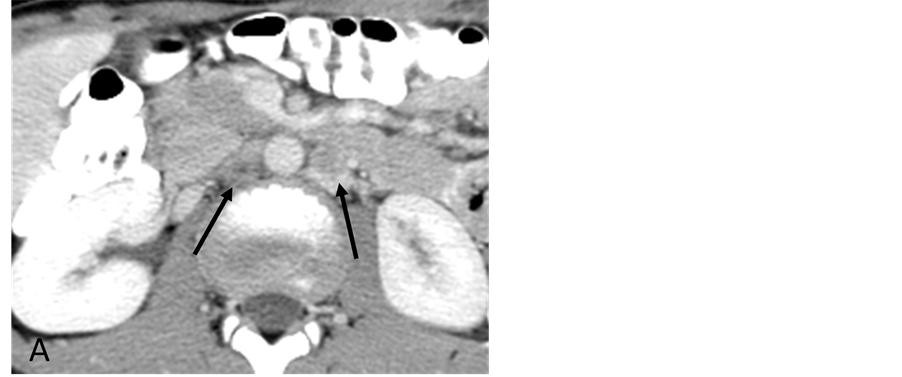
A 15-year-old man was referred to Interventional Radiology for evaluation of persistent retroperitoneal masses and hypertension with suspicion for renal artery stenos is. The past medical history was notable for a history of trauma and a retroperitoneal mass suspected to be a hematoma for a period of 5 years. Initial computed tomography (CT) scan demonstrated a low attenuation 1.6 cm × 1.5 cm retroperitoneal mass causing anterior deviation of the right renal artery suspicious for compression (Figure 1(A)). Renal ultrasound demonstrated normal kidneys bilaterally without signs of renal arterial stenosis. Further evaluation with magnetic resonance imaging (MRI)/magnetic resonance angiography (MRA) showed an irregular contrast enhancing 2.9 cm × 2.2 cm × 1.5 cm mixed signal retroperitoneal mass that demonstrated a mass effect on the inferior vena cava and right renal artery (Figure 1(B)). Despite elevated urine catecholamines, a subsequent iodine-131-meta-iodobenzylguani- dine (MIBG) scan was negative. Due to persistence of the mass and refractory hypertension requiring multiple antihypertensive medications, the patient was referred to Interventional Radiology for the evaluation ofsuspected renal artery stenosis.
Plasma catecholamine assay performed 4 years earlier, yielded epinephrine 67 pg/mL (n, 0 - 100 pg/mL), Dopamine, 108 pg/mL (n, 0 - 100 pg/mL), and norepinephrine, 1405 pg/mL (n, 0 - 500 pg/mL). Serum aldoste-
Figure 1. (A) Contrast-enhanced CT scan section through the kidneys showing two retroperitoneal masses (arrows); (B) On MRI the lesions have high signal intensity on T2-weighted images (arrows). The right retroperitoneal tumor is located posterior to the IVC and right renal artery.
rone was 2.6 ng/dL (n, recumbent, 1 - 16 ng/dL). Urine norepinephrine was 219 µg/TV (n, 0 - 100 µg/TV) and urine epinephrine was 52.4 µg/TV (n, 0.0 - 20.0 µg/TV).
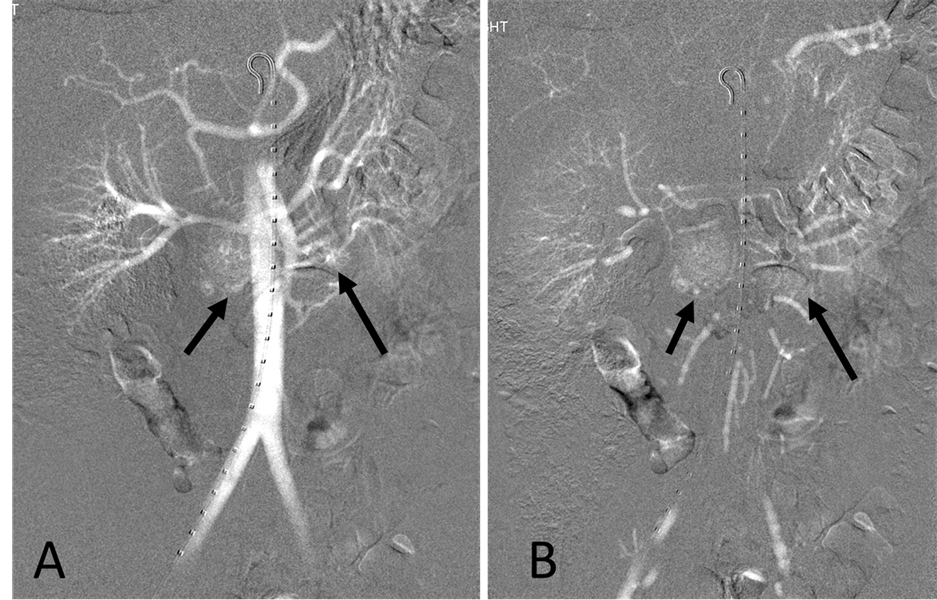
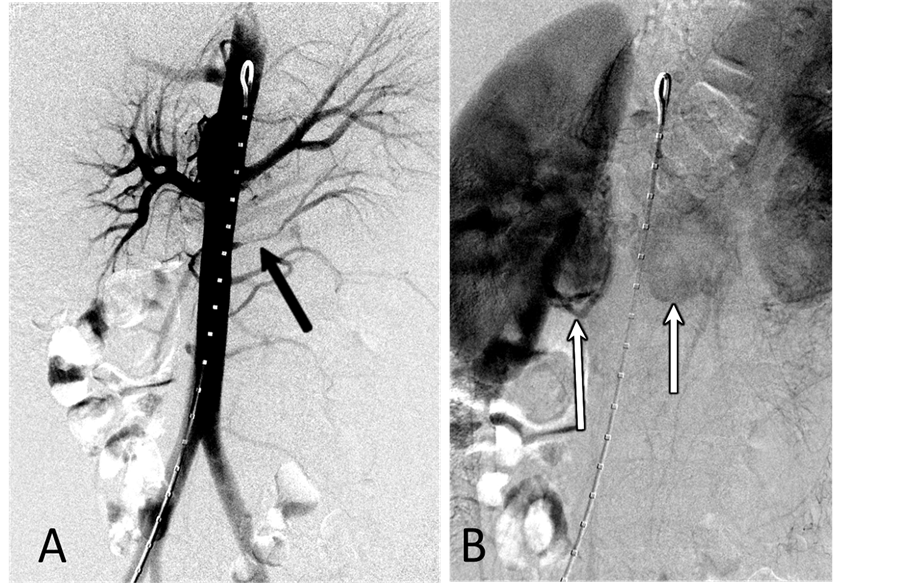
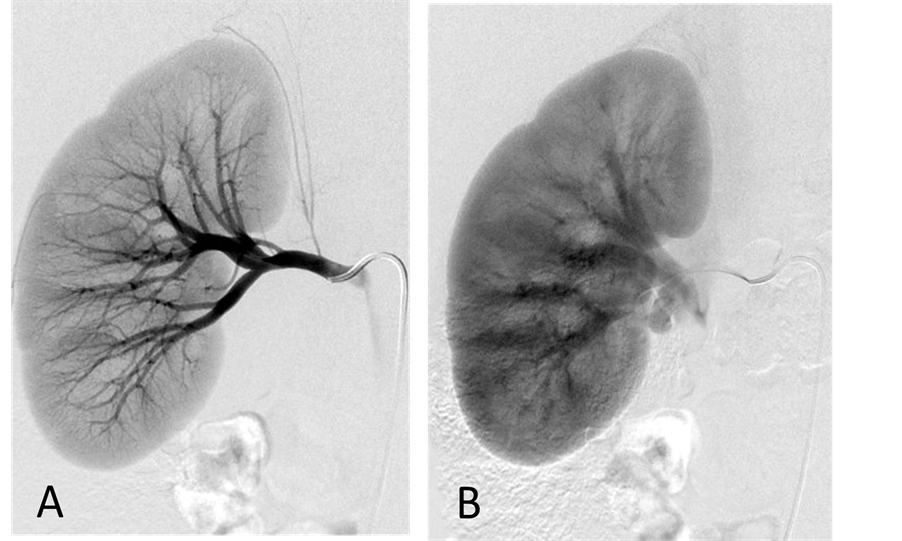
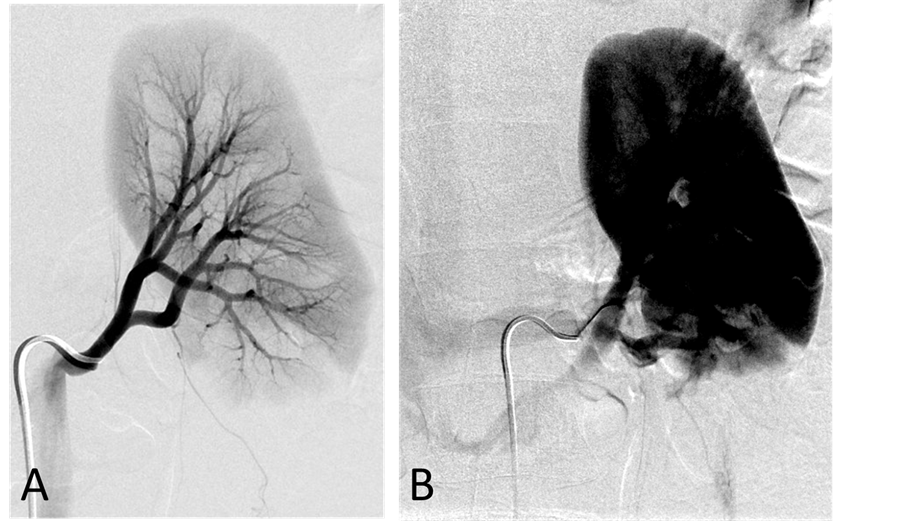
With the patient under general anesthesia, a 5-Fr Omni Flush catheter (Angio Dynamics, Queensbury, NY, USA) was placed in the aorta above the celiac axis from a percutaneous retrograde transfemoral arterial approach, and aortograms were performed, first with 30 cc of CO2 and then in the anterior-posterior (AP) and left anterior-oblique (LAO) projections with Visipaque (GE Healthcare, Cork, Ireland, UK). During each of the runs, there was a moderate increase in blood pressure from baseline systolic pressures of 120 s to 145 - 150 s, lasting 5 minutes, accompanied by bradycardia and transient arrhythmia. A nitroglycerin drip was started; however, the patient did not require any alpha or beta blockers. Next, a 5-Fr shepherd hook catheter (Angio Dynamics, Queensbury, NY, USA) was positioned and digital subtraction angiography (DSA) was performed, sequentially, in the right and left renal arteries, superior mesenteric artery, and finally the celiac axis. The aortograms revealed two vascular retroperitoneal tumors to the right and left of the aorta, at the level of L1-2, measuring 22 mm × 27 mm and 26 mm × 27 mm, respectively (Figure 2(A) and Figure 2(B)). The right retroperitoneal mass was located posteroinferior to the right renal artery and posterior to the inferior vena cava (IVC). The left retroperitoneal mass was located between the left main renal artery and the inferior accessory renal artery. The inferior accessory renal artery supplying the lower pole of left kidney showed diffuse narrowing due to either compression or catecholamine release by the tumor (Figure 3(A) and Figure 3(B)). Subsequently, bilateral renal arteriograms were performed without a hypertensive response to the injection of contrast material. The right main renal artery was displaced by the tumor with mild arterial narrowing (Figure 4(A) and Figure 4(B)). Left renal arteriogram showed a patent main renal artery with a circumaortic renal vein (Figure 5(A) and Figure 5(B)).
Selective venous sampling for catecholamines and renal vein renin were performed from a percutaneous retrograde transfemoral venous approach with a 5-Fr Cobra catheter with two side holes punched near its tip (Cook, Bloomington, IN, USA) to elicit the etiology and location of the masses. Blood samples were obtained along the right and left sides of the IVC above and below the right and left renal veins, respectively. Additional blood samples were obtained from the right and left renal veins, and left retroaortic renal vein. The catecholamine concentrations (dopamine and norepinephrine) were elevated along the IVC receiving effluent blood from
Figure 2. (A) CO2 digital subtraction aortogram, arterial phase, showing hypervascular retroperitoneal tumors (arrows) (B) late arterial phase. There is increased CO2 accumulation throughout the tumors (arrows).
Figure 3. (A) Aortogram with iodinated contrast medium in left anterior oblique projection. The tumors show fine neovascularity. The inferior accessory renal artery (arrow) abutting the inferior margin of the tumor shows diffuse narrowing; (B) Capillary phase. There is irregular, increased contrast accumulation throughout the tumors (arrows).
Figure 4. (A) Right renal arteriogram, arterial phase. There is mild compression of the main renal artery by the tumor associated with mild narrowing of the renal artery at the hilus; (B) Right renal arteriogram, capillary phase. No tumor blush is seen.
the retroperitoneal masses. The concentration of epinephrine in the IVC was normal. The values of catecholamines are shown in Table 1. Left renal vein renin value was 117.1 ng/mL/hour (n, 0.5 - 3.5 ng/mL/hour), right renal vein renin level, 75.9 ng/mL/hour, and IVC, 91.4 ng/mL/hour. The left renal: right renal renin ratio was 1.5, lateralizing renin excess to the left kidney which is most likely due to narrowing of the left inferior accessory renal artery caused by the tumor.
Figure 5. (A) Left renal arteriogram,arterial phase. The main and intrarenal arteries are patent; (B) Left renal arteriogram, capillary and venous phase. The nephrographic defect in the lower pole of the kidney is the part of the kidney supplied by the accessory renal artery arising from the aorta. There is circumaortic renal vein as renal vein variation.
Table 1. Plasma catecholamine levels in the inferior vena cava and renal veins.
Normal serum catecholamine values: Epinephrine = 0 - 100 pg/mL; Dopamine = 0 - 100 pg/mL; Norepinephrine = 0 - 500 pg/mL.
The patient subsequently underwent surgical resection of the masses at the outside hospital. The procedure was completed at an outside hospital; therefore, histology of the resected tumor is not available. It was documented as a pheochromocytoma in the patient’s medical record and supported by normalization of pre-operative elevation of catecholamine following removal of the tumor. On postoperative day #7, the plasma free normetanephrine was 0.65 nmol/L (n < 0.90 nmol/L) and plasma free metanephrine was lower than 0.20 nmol/L (n, < 0.50 nmol/L). Plasma norepinephrine, epinephrine, and Dopamine were also within normal limits.
The patient has not had any headache, dizziness, sweating, or abdominal pain suggestive of recurrence of his pheochromocytoma for two years and ten months after resection of pheochromocytoma. His lab studies for catecholamines were normal. He remained normotensive.
3. Discussion
Since the advent of digital subtraction angiography, CO2 has commonly been used as a safe alternative contrast agent for diagnostic angiography and endovascular procedures in patients with or without renal failure and contrast allergies. To the best of our knowledge, this is the first case of a hypertensive response to the intra-aortic injection of CO2 for aortography in a patient with catecholamine-secreting paraganglioma.
Arteriography is infrequently performed in patients with clinical and biochemical evidence of pheochromocytoma or functioning paragangliomas (extra adrenal catecholamine-secreting tumors collectively referred to as pheochromocytoms), given the advent of safe and accurate imaging modalities such as CT and MIBG scans. However, when the diagnosis is still equivocal, angiography with venous sampling is a highly sensitive and specific test that can be employed [7] . In this case, given: CT findings, negative MIBG scan, and history of abdominal trauma; the masses were first hypothesized to be retroperitoneal hematomas therefore leading to a delay in the correct diagnosis and treatment. Regardless of location, pheochromocytomas are usually hypervascular tumors exhibiting neovascularity and tumor stain. The cystic nature of the tumor or central necrosis of the tumor can demonstrate a central avascular zone surrounded by a dense rim of contrast accumulation, giving a “ring sign”. Functional paragangiomas may be found in chromaffin tissue throughout the body including; along the sympathetic chain, in the neck and thorax, along the wall of urinary bladder, the organ of Zuckerkandl (located inferior to the origin of the inferior mesenteric artery), and pelvis. High local levels of catecholamines can be found in the adrenal and renal veins and along the IVC, depending on the location of the tumors [8] . Generally, extra-adrenal pheochromocytomas secrete predominantly dopamine and norepinephrine, whereas adrenal pheo- chromocytomas secrete epinephrine. Given the extensive yet equivocal workup the patient received, adrenal and renal vein sampling was performed to determine the diagnosis of pheochromocytoma. The normalization of catecholamine levels 7 days post operatively and resolution of persistent hypertension further confirmed the diagnosis.
Hypertensive crisis induced by ionic contrast material administered intra-arterially or intravenously, in patients with pheochromocytomas is a well-known phenomenon [4] [5] . Furthermore, certain agents, such as glucagon, have been reported to induce catecholamine release from pheochromocytomas with subsequent multi-organ injury during CT [6] . In patients with pheochromocytoma of the urinary bladder, an increase in intravesical pressure during micturition can also lead to catecholamine release and hypertensive symptoms [9] . When angiography is performed in unsuspected cases of pheochromocytomas, using contrast materials may cause hypertension. The exact mechanism for this is unknown and may be due to either contrast-induced catecholamine release or release due to arterial pressure changes from contrast injection. Studies have shown that non-io- nic contrast does not have this same effect when given intravenously during CT scans; however, there are no studies regarding the safety of non-ionic contrast in angiography [10] [11] . Pre-procedural treatment with alpha blockers has been used during ionic and non-ionic contrast studies to prevent hyperadrenergic crisis, though there have been reports of breakthrough symptoms. Phenoxybenzamine, a non-selective, irreversible alpha antagonist is commonly used; however it requires the administration of a beta adrenergic antagonist to prevent reflex tachycardia [12] [13] . Doxazosinemesylate, a α1-selective alpha blocker, has also been used more recently and does not cause the same post-procedural hypotension noted with phenoxybenzamine [14] . During the patient’s hypertensive response with bradycardia and arrhythmia to CO2 angiography, a nitroglycerin drip was used successfully without the need for an alpha or beta blockade. Despite this, we believe that the standard protocol for treatment of hypertensive crisis following contrast administration, as mentioned above, should be on hand prior to performing the procedure.
To our knowledge, there have been no studies describing the safety of CO2 angiography in diagnosing catecholamine-secreting tumors. Given its relative safety as a contrast agent as evidenced by its use in patients with renal failure or contrast allergies, it would be assumed that CO2 angiography would not induce a hyperadrenergic response, yet this case shows that it is possible. The rare occurrence of this complication may be a result of a relatively low utilization of CO2 in comparison to iodinated contrast when performing angiography and venous sampling, or the high accuracy of CT in diagnosing these tumors leading to a small number of angiographic testing overall.
Due to the potential neurotoxicity and coronary ischemia, CO2 should not be used in the arterial circulation above the diaphragm. However, CO2 can be safely used as an intravenous contrast agent in the venous circulation including the central veins, the IVC, the SVC, and the portal venous system. The potential complications of CO2 as a contrast agent are air contamination resulting from incorrect use of the delivery system and pulmonary vapor lock due to the inadvertent administration of excessive volume of CO2.
4. Conclusion
In conclusion, despite the known safety of CO2 as a contrast agent in the arterial circulation below the diaphragm, CO2 aortography can cause a hypertensive response in patients with pheochromocytomas. We therefore propose that centers should consider pre-procedural therapy with the current standards for treatment of hyperadrenergic crisis, alpha and beta antagonists, or have the medications on hand with close patient monitoring in case of a hypertensive event.
References
- Seeger, J.M., Self, S., Harward, T.R., et al. (1993) Carbon Dioxide Gas as an Arterial Contrast Agent. Annals of Sur- gery, 217, 688-698. http://dx.doi.org/10.1097/00000658-199306000-00011
- Hawkins, I.F. (1982) Carbon Dioxide Digital Subtraction Arteriography. American Journal of Roentgenology, 139, 19- 24. http://dx.doi.org/10.2214/ajr.139.1.19
- Hawkins, I.F., Wilcox, C.S., Kerns, S.R., et al. (1994) CO2 Digital Angiography: A Safer Contrast Agent for Renal Vascular Imaging? American Journal of Kidney Diseases, 24, 685-694. http://dx.doi.org/10.1016/S0272-6386(12)80232-0
- Gold, R.E., Wisinger, B.M., Geraci, A.R. and Heinz, L.M. (1972) Hypertensive Crisis as a Result of Adrenal Veno- graphy in a Patient with Pheochromocytoma. Radiology, 102, 579-580. http://dx.doi.org/10.1148/102.3.579
- Christenson, R., Smith, C.W. and Burko, H. (1976) Arteriographic Manifestations of Pheochromocytoma. American Journal of Roentgenology, 126, 567-575. http://dx.doi.org/10.2214/ajr.126.3.567
- Hosseinnezhad, A.I., Black, R.M., Aeddula, N.R., Adhikari, D. and Trivedi, N. (2011) Glucagon-Induced Pheochro- mocytoma Crisis. Endocrine Practice, 17, e51-e54. http://dx.doi.org/10.4158/EP10388.CR
- Allison, D.J., Brown, M.J., Jones, D.H. and Timmis, J.B. (1983) Role of Venous Sampling in Locating a Phaeochro- mocytoma. British Medical Journal (Clinical Research Ed.), 286, 1122-1124. http://dx.doi.org/10.1136/bmj.286.6371.1122
- Davies, R.A., Patt, N.L. and Sole, M.J. (1979) Localization of Pheochromocytoma by Selective Venous Catheterization and Assay of Plasma Catecholamines. Canadian Medical Association Journal, 120, 539-542.
- Li, W., Yang, B., Che, J.P., Yan, Y., Liu, M., Li, Q.Y., Zhang, Y.Y. and Zheng, J.H. (2013) Diagnosis and Treatment of Extra-Adrenal Pheochromocytoma of Urinary Bladder: Case Report and Literature Review. International Journal of Clinical and Experimental Medicine, 6, 832-839.
- Mukherjee, J.L., Peppercorn, P.D., Reznek, R.H., et al. (1997) Pheochromocytoma: Effect of Nonionic Contrast Me- dium in CT on Circulating Catecholamine Levels. Radiology, 202, 227-231. http://dx.doi.org/10.1148/radiology.202.1.8988215
- Bessell-Browne, R. and O’Malley, M.E. (2007) CT of Pheochromocytoma and Paraganglioma: Risk of Adverse Events with i.v. Administration of Nonionic Contrast Material. American Journal of Roentgenology, 188, 970-974. http://dx.doi.org/10.2214/AJR.06.0827
- Rossi, P., Young, I.S. and Panke, W.F. (1968) Techniques, Usefulness, and Hazards of Arteriography of Pheochromo- cytoma: Review of 99 Cases. The Journal of the American Medical Association, 205, 547-553. http://dx.doi.org/10.1001/jama.1968.03140340017004
- van der Hosrst-Schrivers, A.N., Kerstens, M.N. and Wolffenbuttel, B.H. (2006) Preoperative Pharmacologicalmanagement of Phaeochromocytoma. Netherlands Journal of Medicine, 64, 290-295.
- Prys-Roberts, J.R. and Farndon, J.R. (2002) Efficacy and Safety of Doxazosin for Perioperative Management of Pa- tients with Pheochromocytoma. World Journal of Surgery, 26, 1037-1042. http://dx.doi.org/10.1007/s00268-002-6667-z
NOTES
*Corresponding author.