Journal of Applied Mathematics and Physics
Vol.07 No.04(2019), Article ID:91584,34 pages
10.4236/jamp.2019.74052
Classical Quantum Field Theory Based on the Hypothesis of the Absolute Reference System
Konstantinos Patrinos
National Technical University of Athens, Athens, Greece

Copyright © 2019 by author(s) and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 1, 2019; Accepted: March 31, 2019; Published: April 3, 2019
ABSTRACT
The quantum field theory based on the hypothesis of the absolute reference system is a classical non-relativistic theory, which is compatible with current quantum theory. This conclusion arises when one compares the theoretical results of quantum electrodynamics using the basic principles of this hypothesis. Wave equation, which replaces this of Schrodinger, is the classical wave equation of a peculiar electromagnetic wave, derived from the study of particle structure.
Keywords:
Particle Mechanics, Field Theory, Quantum Electrodynamics, Quantum Mechanics, Experimental Confirmation of Particle Dynamics

1. Introduction
According to the hypothesis of the absolute reference system [1] , the wave-behavior of the particles is described by wave functions that are solutions of the classical differential equation of the electromagnetic wave and replace the corresponding solutions of the Schrodinger equation. These wave functions describe real electromagnetic waves originating from the particle photonic structure according to this hypothesis. The states of high energy particles come from the solutions of a modified Dirac equation, which is adapted to the classical perception of this hypothesis. In the first section, two issues of particle dynamics are discussed. One is the Compton effect, and the other is the experiment of Bertozzi, which is one of the historical experiments for confirmation one of the basic principle of the special theory of relativity. In the other sections, a study of the wave-behavior of the particles in quantum mechanics and quantum electrodynamics is discussed.
1.1. Compton Effect
We will examine now the scattering of a photon by an electron, in the reference system of the laboratory (which is the earth’s frame of reference), that is Compton effect (ref. [2] , paragraph 2.3.4, p. 44, Compton effect), from the point of view of the absolute reference system. We assume that the energy of the photon is  and the mass of the electron at rest is m. At the level XY, the electron momentum vector forms an angle
and the mass of the electron at rest is m. At the level XY, the electron momentum vector forms an angle 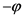 with the axis X, whereas the direction of the photon forms an angle
with the axis X, whereas the direction of the photon forms an angle 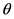 with the same axis.
with the same axis.
Based on what we have mentioned before about the absorption of a high energy photon from a free electron, the phenomenon studied will be accompanied by an increase in the mass of the electron equal to the equivalent mass of a bound photon 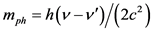 (due to the difference in the frequency of the photon incident to the electron and the corresponding outgoing) and also by a kinetic energy absorption equal to:
(due to the difference in the frequency of the photon incident to the electron and the corresponding outgoing) and also by a kinetic energy absorption equal to:
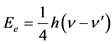 (1.1)
(1.1)
The total energy of the outgoing photon after the impact is equal to 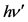 and the kinetic energy of the electron after the impact, as previously described, is equal to
and the kinetic energy of the electron after the impact, as previously described, is equal to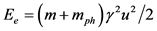 . The velocity u is measured with the instruments of the frame of reference of the laboratory and the contraction factor is
. The velocity u is measured with the instruments of the frame of reference of the laboratory and the contraction factor is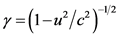 . The momentums of the incident and outgoing photons will be
. The momentums of the incident and outgoing photons will be  and
and 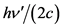 respectively; the momentum of the electron is p, while the frequency of the deposited mass of the bound photon is equal to
respectively; the momentum of the electron is p, while the frequency of the deposited mass of the bound photon is equal to .
.
Due to the conservation of momentum on the X axis, the following relation is taken:
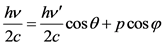 (1.2)
(1.2)
The conservation of the momentum on the Y axis:
 (1.3)
(1.3)
Of these two last relations:
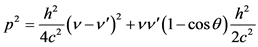 (1.4)
(1.4)
According to the previous mentioned and the relation (1.1):
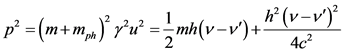 (1.5)
(1.5)
From these two latter relations, the change in the wavelength of the photon initially incident to the electron is calculated:
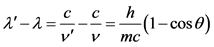 (1.6)
(1.6)
1.2. The Experiment of W. Bertozzi
An experiment of controlling the correctness of a proposed dynamics, such as the dynamics of the absolute reference system, is that of W. Bertozzi1, which was carried out in the early 1960s, and refers to the measurement of the maximum speed of high energy electrons by a linear accelerator (ref. [4] , chapter 1, Departures from Newtonian dynamics, “THE ULTIMATE SPEED”). The already accelerated electrons are released in small bundles (of time duration about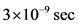 ), directed to the high-voltage negative end of the Van de Graaff accelerator. The path is described as “8.4 meter drift space” in Figure 1. Insulated leads at the ends of the path, collect the electrical signals of the beam.
), directed to the high-voltage negative end of the Van de Graaff accelerator. The path is described as “8.4 meter drift space” in Figure 1. Insulated leads at the ends of the path, collect the electrical signals of the beam.
These electrical signals are transmitted on a down-turn oscilloscope via two wires of the same length (so that the signals need equal time to reach the oscilloscope). In this way the pulses displayed on the oscillator give the real time of transmission of the electron beam along the “drift space”.
While electron velocity measurements are determined directly using the oscilloscope, kinetic energy is determined from potential difference produced in the Van de Graaff generator and electric field in Linac. This is a strictly predetermined procedure, which has been tested in the laboratory by magnetic deflection methods.
To test any dependency of the electron velocity from the force exerted, due to the very strong electric field, an additional measurement acquired by the high energy electrons is made to a further embodiment comprising an aluminum disk on which impinge the electrons at the end of their path and a thermocouple to measure the temperature increase of the aluminum disc, so the added energy in the form of heat will be proportional to the increase in temperature. In addition, an additional device for measuring the charge collected in the disk is used, in this way to determine the energy transferred from each electron. Such energy measurements were made in the estimated accelerator energies at 1.5 MeV and 4.5 MeV (tested by the above-mentioned magnetic deflection methods), whereby the corresponding values, obtained with the heat increase measurement method in the aluminum disc, were 1.6 MeV and 4.8 MeV.
The results of the experiment, listed in Table 1, are five measurements of the electron velocity at corresponding kinetic energy values.
The comparison of experimental results with theoretical calculations of the special theory of relativity and of the hypothesis of the absolute reference system, is certainly the basic criterion of convergence of the experiment with these considerations. The theoretical values of kinetic energy of the special theory of relativity derive from the relation:
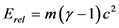 (1.7)
(1.7)
where m is the mass of the electron, u the electron velocity of the beam and

However, the transferred kinetic energy in the target molecules, i.e. the experimentally measured heat, is calculated based on the relative description of the electron collisions of the beam with the atoms of the material. In the absorbtion of a free high energy photon (i.e. of an, elementary, plane electromagnetic wave) from an electron, half of this energy is transferred as kinetic energy, while the other half is available for formation of additional elementary mass. Indeed, in the collision of the beam electrons with the target, the predominant image of the interactions is that of the polarized photons, as an image of elementary plane-waves, that act as interaction photons. Under these conditions the kinetic energy transferred to the target atom, in the form of heat, will be equal to:
Figure 1. The apparatus schematic diagram of measuring of the electron experimental flight time and of the electrons energy. The electrons have already accelerated due to the existence of a strong electric field of the Van de Graaff generator.
Table 1. Experimental results of W. Bertozzi’s measurements, as set out in his work entitled “The Ultimate Speed”, in 1964.

In an intermediate state, where the electrons of the beam move at speeds that are not very close to the velocity of light in the vacuum, a part of the total number of force carriers will transfer the total kinetic energy to the target atoms, while the remaining force carriers the half of the kinetic energy. In this case, the energy transferred in the form of heat to the target atom will have a value between 

Table 2 includes in the first column the ratio of speeds
The values of the first two lines in the table correspond to speeds equals to 87% and 91% of the light velocity in the vacuum, not so close to 100%. Since, according to the hypothesis of the absolute reference system, the heat transferred to the target has values between 


The theoretical values derived from the special theory of relativity are confirmed only in the first line of the table, while the rest are much smaller than the corresponding experimental ones. These values might be somewhat acceptable if they were larger than the corresponding experimental ones. Therefore the experimental results are in accordance with the special theory of relativity only in that the higher speed in nature is that of the velocity of light in the vacuum. However, it is not in agreement with the corresponding experimental values of energy.
Table 2. Table of experimental and theoretical values of the special theory of relativity and absolute reference system.
2. The Wave Behavior of Particles
If photons are the structural component of matter, then a self-evident conclusion is that particle movements should obey a wave equation similar to the differential equation of the electromagnetic wave. It seems, according to the relative theoretical analysis of this section, in the light of the hypothesis of the absolute reference system, that the theoretical results of this assumption are acceptable, since they are in accordance with those of modern physics.
2.1. Particle-Frequency and Wavelength
A particle, as previously described, is composed of a number of bound photons and its total energy will be determined as the sum of total energies of these photons. The total energy derived from the mass frequencies of all the bound photons of the particle shall be equal to:

where 




where 


The kinetic energy of the particle will result from the difference:

We define the quantity 



Since the mass frequencies of the bound photons are different from each other, the transfer frequencies of all the bound photons, based on the latter relation, will be different from each other. Therefore, the sum of the transfer frequencies of all bound photons will be equal to the particle frequency. This frequency (which is actually a sum of frequencies) accompanies the movement of each particle and is a characteristic of its reference system.
Therefore the kinetic energy in relation to the particle frequency is:

Also the corresponding particle wavelength 


where k (the wavenumber) is equal to
2.2. Wave Function of a Free Particle
The wave function 

which propagates at a velocity
This wave function as a solution of the wave equation will be of the form3:

where the constant A is the amplitude of the particle wave, 




the equation of momentum in relation to energy for a free particle 

which is the aforementioned differential wave equation.
The operator of momentum is the same as that of the Schrodinger quantum mechanics, but the operator of energy is differentiated by a factor equal to 1/2, since the operator of energy of the Schrodinger quantum mechanics is equal to

If the constant A of the solution of the differential equation of electromagnetic wave is the electric field amplitude derived from the sum of all the electric fields of all the elemental photonic electromagnetic waves accompanying the movement of the particle, then the quantity A2 is proportional to the energy density of these electric fields, and also it is proportional to the density of the mass inside the particle-space. Since the amount of this energy is constant for each particle, in a parallel beam of same particles with the same number of bound photons per particle, the magnitude 

The particle current corresponding to the wave function 

and therefore:

so, based on the continuity equation


According to relations





When the particle moves within a potential, then as we shall see later, the solution of the differential equation will surely vary as compared to that of the free particle. In this case, for a plurality of same particles, under the influence of this potential, the quantity 




As we will see in Section 4, on quantum field theory based on the hypothesis of the absolute reference system, the Klein-Gordon equation and the Dirac equation, which are used in the case where the velocity of the particle is comparable to the velocity of light in the vacuum, are acceptable by the hypothesis of the absolute reference system. All the theoretical results of quantum field theory (for example, the theoretical results of quantum electrodynamics) are accepted by the hypothesis of the absolute system, if interpreted in the basis of this theory.
2.3. Wave Function of a Particle in the Presence of an External Potential
Starting from the assumption that an initially free charged particle, for example a free electron or a parallel electron beam, enters a space with stable electrical potential, with some initial conditions of the problem, we reach a differential equation. The solution of this equation gives the ability to determine the number of particles, as a function of the location. If the dynamic energy V of an electron is positive but less than the initial kinetic energy T, then the kinetic energy in the space of existing potential is 


The equations to which it generally obeys a particle motion to an external potential V is the energy conservation equation 



By including the time evolution of this particle’s state, we arrive at a more general form of the wave function, 




It also, of course, satisfies the wave-equation:

2.4. Particle Motion in Closed Orbits
For an electron moving in an atomic scale space, under the influence of the Coulomb field, the corresponding force exerted on it will have values that correspond to the same scale (that is, this force can not be enormous), and therefore, its velocity will not be comparable to the speed of light in the vacuum. So, the calculated contraction factor value is very close to 1 (
According to mentioned in the previous subsection 2.1 about the kinetic energy in relation to particle frequency, according to the relation (2.5) presented in subsection 2.1 the frequency of the particle is proportional to kinetic energy. Therefore, in a closed periodic motion of an electron, the average value of the kinetic energy, over a time period equal to that required for a complete closed orbit of the particle, will be proportional to the average value of this frequency. This results in the following relation:

where



and respectively is defined as “equivalent time period of oscillation” of the particle-wave the amount

Since the electron must behave, based on its structure, like a wave, in order for the movement of its closed orbit to be stable, this wave should be a stationary wave. Therefore, an additional binding condition is introduced which governs the periodic motion under consideration and is that the length of the closed orbit should be an integer multiple of the equivalent particle-wavelength.
If the 




and finally a general equation is:

This result, taking into account the equation of motion of the electron, according to the relative examples exposed in the next section, leads to the conclusion that the energy of the electron and also all physical quantities involved in this problem are quantized.
2.5. Uncertainty Principle
In the subsection 2.2 it is stated that the quantity 



By normalizing the function 

the amount 



The mathematical development of the subject here was done by Andre Kessler4. It turns out that the Fourier conjugate of a very localized waveform will be spread out. Thus, if position and momentum (or energy and time, etc.) are Fourier conjugates, and if you know the position to a high degree of accuracy, then you don’t know the momentum very well and vice versa.
If 

The inverse Fourier transform is:

The momentum will be given by the relation

where 



Like earlier, we should expect the classical position to be the average value, and other values to be less probable, and therefore the probability of position is expressed by a normal distribution. If we let 

where 

Since the function 



The coefficients 

this gets us:
Since the 





After the integration of the second member of the last equation, the following equation is taken:

In the last equation, the coefficients and exhibitors must be equal. The two equations:
end up in exactly the same relation:

Due to the relation


This, of course, the latter is true only if the probability distribution is normal. If it isn’t 

or else:

This is the Heisenberg uncertainty principle for position and momentum.
The time-dependent part of wave function 

The inverse Fourier transform can be written as follows:

here the quantity 


where


As above, in the general case:

In the case of a harmonic oscillator the total energy is equal to 

This last relation is the Heisenberg uncertainty principle for energy and time.
3. Examples of Electron Motion in Various Potentials
We will then look at some of the best-known examples of quantum mechanics in the light of the hypothesis of the absolute reference system. The results obtained by solving these examples seem to be fully in agreement with the corresponding results of Schrodinger’s quantum mechanics.
If we consider as initial condition the relation

3.1. Circular Motion of Electron in Coulomb Potential
A simplified example of an electron’s motion in a Coulomb field is that of circular motion. In this case the field comes from a unique proton. In the general case, such as the Hydrogen atom, the orbit of the electron is elliptical, but this subject will be examined in a next example.
The total energy of the electron in our example is:

where m is the mass, u is the electron velocity, e is the elementary charge, and r is the position of the electron in a cartesian coordinate system, with origin the center of mass of the proton-electron system. Based on the centripetal force exerted on the electron

Since the speed remains constant, based on the relation (2.23) presented in subsection 2.4, the average value of velocity-squared is

The frequency 

and therefore on the basis of the foregoing, the total energy, speed, the radius of the circular track and the angular momentum, are given by the following relations:




In the same relations one ends up, applying Bohr’s theory of circular motion.
3.2. Infinite Potential Well
We assume that the potential at the X axis is zero in the range 


The kinetic energy of the electron is:

where m is the mass of the electron. Since the speed remains constant, based on the relation (2.23) presented in subsection 2.4, the average of the velocity-squared is

The speed is equal to 

The kinetic energy is:

The momentum is:

3.3. Harmonic Oscillator
The potential of the one-dimensional harmonic oscillator, in the X direction, is given by the parabolic form:

assuming x equal to




where

Therefore the total energy of the oscillator is given by:

The values 



3.4. Hydrogen Atom
Our basic hypothesis here is that the electron trajectory is elliptical, and one focal point of the ellipse is the center of mass of the hydrogen atom (that is, lies in the nucleus). The orbital position of the electron in polar coordinates (reference [8] , paragraph 3-7, THE KEPLER PROBLEM: INVERSE SQUARE LAW OF FORCE), is given by the relations:
where a and b are the lengths of the semi-major and semi-minor axis of the ellipse respectively. The angular momentum is conserved, and is equal to:

The force exerted to the electron is:

where e is the charge of the electron. According to the last relation the angular momentum squared is:

The kinetic energy is:
According to the relations:
and relation (3.20), the relation for kinetic energy becomes:

The dynamic energy, according to relation (3.22), is:

The total energy of the electron as a sum of kinetic and dynamic energy (and according to the relation (3.22)) is:

From the last relation, it appears that the total energy of the electron is inversely proportional to the length of the large axis of the elliptical trajectory.
Then, in order to use the Equation (2.23) presented in subsection 2.4, we will calculate the time 

The area of the ellipse, taking into account the area speed, is:

Since the area of ellipse is

Since the frequency of periodic electron motion in the closed elliptical trajectory is

The time average of kinetic and dynamic energy (that is, the values of 

so, based on the virial theorem 



which is the expected value, since, due to the virial theorem, the total energy is

so, the quantized semi-major axis of the elliptical trajectory is:

The quantized total energy of the electron is:

We will now examine the quantized term of kinetic energy

where 




Due to relation (3.22) and the relation (3.32), the resulting angular momentum is:

where 

From the last two equations we get the equation:

If 




that is, it takes values:
We assume now that the elliptical orbit of the electron is on the level XY of a Cartesian coordinate system XYZ. If the plane of the trajectory has been rotated at an angle

where we have considered as an integer quantum number of the projection of angular momentum on the Z axis the number





3.5. A simple Potential Step
We assume the existence of an electric potential










We also assume that the kinetic energy of an electron of the incident-beam is E. Due to the energy conservation, the total energy of an electron of the refracted beam is






while the wave number 

We initially consider




In the case where


Another way to deal with the same problem is to calculate the reflection and refraction from the expressions for the particle currents6. Since the above wave functions refer to the incident, reflected, and refracted beam, the quantity 






where, the


In the area of the negative semi-axis the total wave function (incident and reflected beam) is given by the relation:

while in the area of the positive semi-axis (refracted beam only):

The continuity boundary conditions at 




from which relations emerge:


From the last two relations and from relations (3.44) and (3.45), we end up with the previous relations for reflection and transmission.
3.6. A Rectangular Potential Step
we consider in this example a fixed potential in the X direction in the region 













In the case where the kinetic energy E of an electron of the incident beam is greater than the dynamic energy 



where 



Also, following the analogous procedure for the transmission, we get the relation:

We will now follow the methodology on particle currents and boundary conditions of continuity, as in the previous example. In this case the reflection and transmission are derived from the corresponding current ratios, according to the following relations:


The total wave function in region a is defined as




From these equations, four relations between the complex amplitudes are taken, which are the following:
From the last four equations we get the equality:

From this last equality and from the relations (3.55) and (3.56), we reach the same relation for the reflection, that is the relation (3.53). In the same way, the transmission, given by the relation (3.54), is also calculated.
In the case where the kinetic energy E of an electron of the incident beam is less than the dynamic energy 







Replacing the k and





while for




4. Quantum Electrodynamics
We will examine in this section quantum electrodynamics, according to classical theory based on the absolute reference system. Can a theory of classical physics to include quantum field theory and give corresponding theoretical results?
The answer is affirmative, and this will be seen in this study, in this section. The waveforms used in this section, as in the previous ones, are solutions of this wave equation and do not express probability amplitudes, as they derive from a probabilistic view. In contrast, that waveforms express the propagation of a peculiar particle electromagnetic wave, according to the assumption of absolute reference system, which is a classic non-relativistic view of nature in the broader field of statistical physics.
The free-particle wave-equation for high velocities, according to the hypothesis of the absolute reference system, is the wave-equation, in which the force currier is massive and the factor 




This equation looks like the Klein-Gordon equation7. The wave function that is a solution of this equation will be that of the plane wave8, which is in the form of:

where N is the normalization factor, which will be discussed below. By substituting this wave function in the previous differential equation we get the following relation:
so, due to the energy relation

According to this last relation the wave function (4.2) takes the form:

The operator which acts on the wave function 


while the differential operator, which yields as an eigenvalue the energy E is

Multiplying the two members of the Equation (4.1) by the quantity
Adding the last two equations and multiplying with

The particle density and the particle current are given by the relations:


According to relations 



because the speed 

4.1. Yukawa Potential
Now we will extend to the study of the interaction between heavier particles and the corresponding dynamics. This study has to be done from the point of view of the hypothesis of the absolute system of reference. Such a potential we will first examine is that which comes from relatively heavier particles, such as protons and neutrons. In particular, we will calculate the potential comes from the exchange of intermediate particles (force carriers) that give rise to forces between such heavy particles (see [2] , paragraph 1.5.2, The Yukawa Theory).
The set of carrier particles in the field around a proton, in addition to the photons that are carriers of electrostatic interactions, consists of larger photon packets, which are the intermediate particles of interactions between nucleons. These particles, which are the carriers of strong interactions, are the explanation of the small radius of force action between the nucleons inside the atomic nucleus. The radius of action of static interactions depends, on the mass of the carrier of the quantum field, and an explanation for this was given by Yukawa in 1935, in his effort to describe the above-mentioned forces.
We assume initially that the mass of an intermediate particle that is exchanged is m. From a physical point of view, this exchange gives momentum that justifies the existing force of interaction. Along with the capturing of this mass, the heavy body takes extra energy equal to the total kinetic energy of this intermediate particle. This energy is:

where 


The differential equation of wave motion results from the replacement, in the last relation, of p and E with the corresponding operators 

This last equation has two partial solutions. One partial solution is given by the Equation (4.4), but, at the present, we are not interested as much in the propagation of the particle-wave, as much we are interested in the examined here static potential. The other solution is of the form:

and is independent of time. The 
The physical analog of Yukawa potential in electromagnetism is that resulting from the substitution of the constant g with the charge q. However, because of the very small mass of the interaction photon, the exponential part of the potential is very close to 1 and therefore the electrical potential is of the known form
According to the history of nuclear forces the Yukawa hypothesis predicted as carrier of strong interactions a spinless quantum of mass 

4.2. Perturbation Theory
Let us assume that a time depended potential 



where 






and 
Also the “probable wave-function” 

so




Assuming that before action of the potential 









so we get the following differential equation:

By substituting the expression 15 in this last differential equation, since

By multiplication of the last equation from the left with 


Using the orthonormality relation for 


where we have defined

and what is sometimes called the transition matrix element:

According to the first order approximation, the Equation (4.22) gives us

Using that 


for


For a potential that is time-independent the expression, for the transition amplitude, becomes

where
We define the mean transition rate in the limit for large T as

When the wave functions are those of the plane wave, that is

For the plane waves this gives

where, for plane waves


The phase space factor for a process with n final state particles is

The cross-section is

4.3. Coulomb Scattering
We will examine the case where the outer field comes from a point charge at the beginning of the axes, while the velocity of the incoming charged particle is low, that is

The wave function of the initial state and the conjugate wave function of the final state are:
The transition amplitude is:
according to the relations:
By setting

The time-averaged transition rate is:

Due to normalization of the plane wave function over a box with volume V, we have

while the phase space factor is

According to the relation (4.33) the cross-section is

Since



The kinetic energy of the incoming charged particle is


This is the well-known Rutherford scattering formula.
4.4. Dirac Equation
The Dirac equation with small variations, as will be shown below and with the help of what has already been mentioned in this section, is perfectly compatible with the hypothesis of the absolute reference system. We want to find a squared equation, which gives the wave Equation (4.1). This equation has the following form:

This is certainly a well-known problem whose solutions for 


We also define the four components of the matrix
so the Equation (4.42) becomes,

where we have used the symbol 


We can use Dirac spinors to write plane wave solutions9 of the Equation (4.43). Consider
where 

By substituting this last wave function in the (4.43) equation we get the following solutions:

The second of the Equations (4.44) gives:
where



Since the space of the absolute reference system is the three-dimensional Euclidean space, the four-dimensional space-time will be an extension of this Euclidean space in the four dimensions. In particular, the space-time metric tensor will be the Kronecker






A very important remark is that the Dirac equation does not need to be relativistically covariant under a Lorentz transformation in the hypothesis of the absolute reference system, since this physics is not relativistic.
Considering all of this, the components of 







The Dirac equation for anti-particles is

The plane wave solutions for the particles take the form

where the spinors 


and



According to Dirac algebra we have
The Hermitian conjugate of 
so the Hermitian conjugate of Dirac equation is
Now we multiply the Dirac equation from the left by 

Consequently, we realize that if we define a current as

then this current satisfies a continuity equation,

which is always positive.
Substituting the plane wave solution
Consequently, in order to have one particle per volume V we choose

5. Conclusions
Quantum theory, based on the assumption of the absolute reference system, in the context of quantum mechanics and quantum field theory, is simpler than current quantum theory and gives, with minor differences, the same theoretical results. To sum up, the study so far, based on the hypothesis of the absolute reference system, concludes with the following conclusions:
1) The wave behavior of particles based on the classical wave equation, instead of the Schrodinger equation, gives theoretical results in agreement with those of quantum electrodynamics. It is also noteworthy that based on the hypothesis of the absolute reference system, there are no negative energy values such as those derived from the Klein-Gordon equation, and the Dirac theory.
2) The theoretical energy values in the W. Bertozzi experiment (section 1.2) agree with the corresponding experimental ones, in contrast to the theoretical energy values derived from the special theory of relativity for this experiment, which are not in agreement with the experimental results.
3) The dynamics of the hypothesis of the absolute reference system is confirmed experimentally.
Conflicts of Interest
The author declares no conflicts of interest regarding the publication of this paper.
Cite this paper
Patrinos, K. (2019) Classical Quantum Field Theory Based on the Hypothesis of the Absolute Reference System. Journal of Applied Mathematics and Physics, 7, 747-780. https://doi.org/10.4236/jamp.2019.74052
References
- 1. Patrinos, K. (2019) The Physics of an Absolute Reference System. Journal of Applied Mathematics and Physics, 7, 431-475. https://doi.org/10.4236/jamp.2019.73033
- 2. Perkins, D.H. (1987) Introduction to High Energy Physics. Addison-Wesley Publishing Company, Boston.
- 3. Bertozzi, W. (1964) Education Development Center, Newton, Mass. 1962 “The Ultimate Speed”. American Journal of Physics, 32, 551-555. https://doi.org/10.1119/1.1970770
- 4. French, A.P. (1968) Special Relativity. The MIT Introductory Physics Series, W.W. Norton & Company, New York.
- 5. Patrinos, K. (2018) The Physics of an Absolute Reference System. Konstantinos Patrinos, Athens.
- 6. Jackson, J.D. (1975) Classical Electrodynamics. Second Edition, Wiley Eastern Limited, New York.
- 7. Born, M. and Wolf, E. (1975) Principles of Optics “Electromagnetic Theory of Propagation, Interference and Diffraction of Light”. Fifth Edition, Pergamon Press, Oxford.
- 8. Goldstein, H. (1980) Classical Mechanics. Second Edition, Addison-Wesley Publishing Company, Boston.
- 9. Ryder, L.H. (1985) Quantum Field Theory. Cambridge University Press, Cambridge.
- 10. Landau, L. and Lifshitz, E. (1975) The Classical Theory of Fields. Pergamon Press Ltd., Oxford.
- 11. Feynman, R.P. (1961) Quantum Electrodynamics. A Lecture Note and Reprint Volume, W. A. Benjamin, New York.
NOTES
1The Ultimate Speed, W. Bertozzi, Education Development Center, Newton, Mass. 1962. For more information see reference [3] .
2About experimental confirmation of particle dynamics from the point of view of the hypothesis of absolute reference system see reference [5] , sections 1.3, 2.4 and 3.9.
3ref. [6] , Chapter 7, Plane Electromagnetic Waves and Wave Propagation, section 7.1, Plane Waves in a Nonconducting Medium.
4“Derivation of the Heisenberg Uncertainty Principle’’, Andre Kessler, Department of Mathematics, Massachusetts Institute of Technology.
5When an external magnetic field is applied, sharp spectral lines like the 
6The same way of dealing with this example, based on Schrodinger’s quantum mechanics, is set out in a lecture-note entitled “Reflection and Transmission at a Potential Step’’, MIT OpenCourseWare, http://ocw.mit.edu, 6.007 Electromagnetic Energy: From Motors to Lasers, Spring 2011.
7ref. [9] , Chapter 2, Single-particle relativistic wave equations, section 2.2, Klein-Gordon equation.
8ref. [10] , CHAPTER 6, paragraph 48, Monochromatic plane waves.
9ref. [11] , Thirteenth Lecture, Solution of the Dirac Equation for a Free Particle.





































