Open Journal of Forestry
Vol.07 No.01(2017), Article ID:72624,10 pages
10.4236/ojf.2017.71001
Genetic Variability in Myracrodruon urundeuva (Allemão) Engl. Progeny Tests
Daniela Silvia de Oliveira Canuto1, Alexandre Marques da Silva1, Miguel Luiz Menezes Freitas2, Alexandre Magno Sebbenn2, Mario Luiz Teixeira de Moraes1
1Ilha Solteira School of Engineering, São Paulo State University (UNESP), São Paulo, Brazil
2Forestry Institute, São Paulo, Brazil

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: April 28, 2016; Accepted: December 5, 2016; Published: December 8, 2016
ABSTRACT
Aroeira (Myracrodruon urundeuva) is a widely spread tree species, but due to its excellent wood quality it is now part of the list of vulnerable trees for extinction. Thus, strategies for the conservation of the genetic variability of the remaining natural populations are needed. Therefore, this study aimed to evaluate the genetic variability in M. urundeuva progeny tests, from natural populations located in areas with and without human disturbance. Seeds of six natural populations of M. urundeuva were collected from two conditions, with and without anthropogenic disturbance, and nine progeny tests were installed in the Education, Research and Extension Farm, located in Selvíria, Mato Grosso do Sul State, Brazil. The overall height, average crown diameter (DMC) and the diameter at breast height (DBH) were assessed as phenotypic traits. Estimates of variance components and genetic parameters were obtained by REML/BLUP method (maximum restricted likelihood/best unbiased linear prediction). The results showed that natural population with human disturbance presented less genetic variability than populations originating from areas without human disturbance (CVr until 0.87). The progeny tests evaluated had high accuracy (0.91 for DBH), which ensures the viability of the ex situ genetic conservation bank and its successful use in the breeding program, i.e., in the conversion of the progeny tests into M. urundeuva seed orchards. The most suitable character for selection was the DBH.
Keywords:
Conservation, Improvement, Seed Orchard

1. Introduction
Myracrodruon urundeuva (Allemão) Engl., Anacardiaceae, popularly known as aroeira, backwoods aroeira or urundeuva, is a deciduous species, heliophytic and selective xerofita (Lorenzi, 1992) . M. urundeuva occurs naturally in the extreme northwest of Argentina, in the south and east of Bolivia and Paraguay. In Brazil, it occurs in the following states: Paraíba, Pernambuco, Piauí, Rio de Janeiro, Rio Grande do Norte, Sergipe, São Paulo, Tocantins, Federation District and Mato Grosso do Sul (Carvalho, 2003) . It occurs also in different phytoecological regions, as semi deciduous forest; deciduous forest; scrubland and savanna; caatinga; kills drought; Mato Grosso do Sul and Mato Grosso Pantanal Chaco (Carvalho, 1994) and forest mono-dominated in Minas Gerais (Oliveira et al., 2015) . The wide geographic distribution of a species is an indicator of high levels of genetic diversity, which can give it the ability to occupy different habitats (Kageyama et al., 2003) .
The species has high-density wood, excellent mechanical performance and good chemical and physical defense, which explains the strength, hardness and durability. It is considered the toughest wood from Brazil. While one square centimeter concrete supports a load of 250 kgf wood, M. urundeuva can support 696 kgf, and it is registered at the Institute for Technological Research (IPT) in São Paulo, such as wood “durable” and put in select group of woods with secular durability, calls “imputrescible” (Ribeiro, 1989) . Moreover, M. urundeuva was identified as the most potential species for research on phytochemicals (Pereira et al. 2014) . Whereas M. urundeuva is endangered (BRASIL, 2008) , Souza et al. (2012) reported a significant use of this species for timber purposes, due to the popularity and the timber and non-timber value of M. urundeuva, can lead to local extinction. Due to these facts, M. urundeuva is experiencing a strong exploration process, in a predatory way, causing the devastation of their natural populations. In addition, it should be noted that selective logging for use in the timber industry virtually wiped out the large individuals, therefore considered endangered and categorized as vulnerable (Brandão, 2000; Mendonça & Lins, 2000) .
Studies to preserve and guarantee the genetic variability of these threatened populations have been intensified, and the tools used are the progeny tests, which is an ex situ strategy. Currently one of the major purposes of the progeny test is its transformation into an orchard, to provide seeds with genetic quality to restore the Legal Forest with superior material and adapted soil and climatic conditions associated with economic alternatives, to ensure the population’s quality of life place. Thus, the aim of this study was to evaluate the genetic variability in nine M. urundeuva progeny tests.
2. Materials and Methods
Seeds of six natural populations of M. urundeuva were collected in two situations, without human disturbance (natural forest) and with human disturbance (modified areas with agriculture and grazing) (Table 1). Which were seedlings produced and installed in nine progeny tests in Education, Research and Extension Farm, located in Selvíria, Mato Grosso do Sul State, Brazil, thus, constituting the M. urundeuva ex situ bank.
With the seeds from natural populations of M. urundeuva without human disturbance of Paulo de Faria, four progeny tests were installed. The other populations been installed in only one provenance and progeny test. All experiments were conducted, using the randomized block design. The number of replications; the number of progenies and the number of plants per plot are presented in Table 2.
Table 1. Geographical Location of natural populations of Myracrodruon urundeuva with human disturbance (CPA) and without human disturbance (SPA).
Table 2. Characteristics of Progeny Tests (TP) of Myracrodruon urundeuva installed in Selvíria, Mato Grosso do Sul.
R: number of replications; P: number of progeny; N: number of plants by progenies.
In the test 1, 3, 4, 5, 8 and 9, M. urundeuva was planted in consortium with other species: 1-Anadenthera falcata and Guazuma ulmifolia; 3 and 4-Ricinus communis, Zea mays and Cajanus cajan; 5-Eucalyptus spp; 8-Trema micranta, Peltophorum dubium and Eucalyptus spp, 9-Cordia trichotoma, because M. urundeuva needs shade in early development. The progeny tests 2, 6 and 7 were planted only using M. urundeuva. All trials were measured in 2007 for the quantitative traits: total tree height (H); average crown diameter (DMC) and diameter at breast height (DBH).
Quantitative variables were analyzed by the method of linear mixed model (univariate additive)-REML/BLUP, applied to progeny tests in randomized block design, many plants per plot, one site and a single population, following the procedure proposed by
Resende (2002a)
: y = Xb + Za + Wc + e; where: y = data vectors; b = vector of the effects of blocks (fixed); a = vector of additive genetic effects (random); c = vectors of the effects of plot (random); e = vector of random errors effects. X, Z e W = incidence matrices for b, a and c, respectively. Estimates of variance components and genetic parameters were obtained by REML/BLUP method (restricted maximum/best linear prediction likelihood untainted), using genetic-statistical software SELEGEN- REML/BLUP
(Resende, 2002b)
. The estimated parameters were the coefficients: heritability in individual, strict sensus (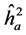 ), average among progenies (
), average among progenies (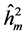 ) and additive within plot (
) and additive within plot (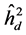 ); genetic variation at the individual level (CVgi) and plot level (CVgp); relative variation (CVr) and accuracy (Ac) to the traits H, DMC and DBH.
); genetic variation at the individual level (CVgi) and plot level (CVgp); relative variation (CVr) and accuracy (Ac) to the traits H, DMC and DBH.
3. Results and Discussion
The highest average annual increment (IMA) for the height (Table 3) for populations without human disturbance was found in the TP-3 (0.89 m) and the lowest in TP-4 (0.58 m), and for the population with human disturbance, the highest was in TP-9 (1.20 m) and the lowest was in TP-8 (0.43 m). Sebbenn and Etorri (2001) , studying a progeny
Table 3. Average estimates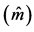 , mean annual increment (IMA), experimental variation coefficient (CVexp), change index (IV), F-test (F) and correlation due to the common environment of the plot
, mean annual increment (IMA), experimental variation coefficient (CVexp), change index (IV), F-test (F) and correlation due to the common environment of the plot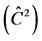 , for traits height (H), DMC and DBH.
, for traits height (H), DMC and DBH.
test of M. urundeuva consortium with Peltophorum dubium and Esenbechia leiocarpa found IMA H of 0.89 m. Bertoni and Dickfeldt (2007) evaluating M. urundeuva planted in alternating areas in the State Park of Porto Ferreira, Sao Paulo State, Brazil, found for H an IMA 0.72 m.
In general, environmental control was good for all trials (CVexp < 20%, Pimentel Gomes (1985) . But the variation index (IV) showed a lower percentage because the value depends only on CVexp, as a proportion of the residual variation of experimental medium (Pimentel Gomes, 1987) and the variation index proposed by Pimentel Gomes (1991) or experimental precision coefficient Storck et al. (2000) consider the number of replications used in the trial, apart from a residual nature variation.
The TP-4 showed significant difference at 1% of probability among progenies, indicating genetic variation among the progeny for the traits height; in other progeny tests occurred homogeneity of values, which was acquired with the development of the progeny, as in earlier ages had differences. When the TP-8 and the TP-3 were evaluated at younger ages, Fonseca (2003) ; Freitas, Moraes and Buzetti (2002) and Freitas et al. (2006) detected significant differences at 5% of probability for height.
Correlation due to the common environment of the plot 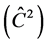 in TP-4 and TP-8 showed to be the most suitable trials for the selection for height, because there is little environmental influence on the variation observed in the plots. According to
Sturion and Resende (2005)
, the values of around 0.10 are observed in experiments with perennials plants.
in TP-4 and TP-8 showed to be the most suitable trials for the selection for height, because there is little environmental influence on the variation observed in the plots. According to
Sturion and Resende (2005)
, the values of around 0.10 are observed in experiments with perennials plants.
The highest means of DMC were found in the TP-5 and TP-7, the CVexp ranged from 8.4 to 22.8%, which shows appropriate experimental environmental control in the trials. The variation rates (IV), which takes into account the number of replications, ranged from 0.70 to 7.60, these values can be considered low for experiments where there is competition (Pimentel Gomes and Garcia, 2002) .
The F test indicated that there is genetic variability among progenies in TP-4, 6 and 8 for the DMC, with significant differences at the level of 1% and 5%. The TP-2 and 6 showed lowest value for the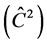 , 0.07 and 0.03, respectively. The other progeny tests presented values higher than 10%.
, 0.07 and 0.03, respectively. The other progeny tests presented values higher than 10%.
For DBH the highest averages were found in TP-6 (7 cm) and 7 (12.94 cm). The highest IMA value was 0.85 cm for TP-2/ These values are superior to that found by Guerra et al. (2009) , obtained average DBH of 6.87 cm and 5.98 cm for M. urundeuva originating from Aramina-SP and Selvíria-MS, at 15 years of age.
The CVexp ranged from medium to high, but the IV indicated good environmental control. TP-4, 5 and 6 showed significant differences between progenies for DBH. The correlation due to the common environment of the plot revealed that the DBH is the most favorable trait for selecting.
The average heritability among progenies (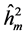 ) was greater than the individual heritability (
) was greater than the individual heritability (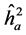 ) and additive within plots (
) and additive within plots (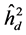 ) for all trait in all of progeny tests (Table 4). This results are in agreement with the results found by
Siqueira et al. (2000)
, in Balfourodendron riedelianum,
Sebbenn and Ettori (2001)
in M. urundeuva,
Sebbenn et al. (2003)
in Araucaria angustifolia,
Souza et al. (2003)
in Astronium fraxinifolium,
Costa et al. (2005)
in Ilex paraguariensis,
Moraes et al. (2007)
in Hevea brasiliensis,
) for all trait in all of progeny tests (Table 4). This results are in agreement with the results found by
Siqueira et al. (2000)
, in Balfourodendron riedelianum,
Sebbenn and Ettori (2001)
in M. urundeuva,
Sebbenn et al. (2003)
in Araucaria angustifolia,
Souza et al. (2003)
in Astronium fraxinifolium,
Costa et al. (2005)
in Ilex paraguariensis,
Moraes et al. (2007)
in Hevea brasiliensis,
Table 4. Genetic parameter estimates for the traits height, DMC and DBH for testing progenies Myracrodruon urundeuva with and without human disturbance.
Missio et al. (2005) in Pinus caribaea var. hondurensis, and Oliveira et al. (2004) in Spondias tuberosa.
The 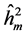 was higher in progeny tests originated from areas with human disturbance, except for the DBH. This results for all the traits studied indicate that the selection can be more effective by selection among progenies
(Costa et al., 2010)
. According to
Falconer (1987)
, small populations are expected to present lower heritability than large populations. The average heritability among progeny show great generic control of traits, indicating the possibility to obtain genetic gains by selecting the best families for seed production.
was higher in progeny tests originated from areas with human disturbance, except for the DBH. This results for all the traits studied indicate that the selection can be more effective by selection among progenies
(Costa et al., 2010)
. According to
Falconer (1987)
, small populations are expected to present lower heritability than large populations. The average heritability among progeny show great generic control of traits, indicating the possibility to obtain genetic gains by selecting the best families for seed production.
The highest heritability coefficients for height were obtained in TP-4 (Table 3). The TP-4 presented highest heritabilities in the first yea (0.8, 0.36 and 0.07, for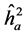 ,
, 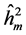 ,
, 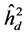 , respectively) and in the second year (0.35, 0.75 and 0.28 to
, respectively) and in the second year (0.35, 0.75 and 0.28 to


In progeny tests derived from the Ecological Station of Paulo de Faria (TP-1, 2, 3 and 5) the
The presence of genetic variability can be confirmed and quantified by the coefficient of genetic variation, which expresses the magnitude of genetic variation relative to the nature of the medium (Resende et al., 1991) . The greatest genetic variation values were found in the DMC and DBH in TP-4 (17.3 and 2.8, respectively), indicating that there is a greater amount of genetic variation among progenies from areas without human disturbance. Incontrast, occurred less genetic variation when compared to populations from forest fragments.
The coefficient of genetic variation in individual level (CVgi) was in general twice time large than the CVgp (Table 3) for height, DMC and DBH. According to Aguiar et al. (2010) , the larger value for the greater ease to find individuals that may provide higher gains in the selection.
The CVr indicates the most appropriate trait for selection, and the TP-5, 7 and 9 the height, the TP-2, 3, 6, 8 and 9 the DMC, the TP-1 and 4 DBH. According Vencovsky and Barriga (1992) , traits that have values close to one are suitable for carrying out a selection.
The accuracy, which measures the confidence level of the estimates, was high in TP-4 for DBH, in TP-4, 5 and 7 for height and TP-4, 6 and 8 for the DMC, according with the magnitude criterion of Resende and Duarte (2007) . Thus, the M. urundeuva progeny tests provide genetic variability among progenies enough to conduct genetic conservation and breeding programs.
4. Conclusion
The evaluations of silvicultural traits in the nine M. urundeuva progeny tests allowed the observation that the trees had a satisfactory development.
Genetic variation of the populations with human disturbance is lower than populations originated from areas without human disturbance.
The progeny tests showed a high accuracy, which ensures the viability of the genetic conservation bank ex situ and success in the breeding programs, by the transformation of trials seed orchards. The most appropriate trait for selection is the diameter at breast height.
Acknowledgements
To the Pro-Rectory of Research for the Young Investigator grant. To aid the search FAPESP No. 05/60113-7 and CNPq No. 470713/2006-2. CNPq for the PhD scholarship. The TT2 FAPESP Grant No. 06/55191-1. The technical agricultural João Antonio de Souza Queiroz and employees of UNESP Jose Cambuim and Alonso Angelo da Silva for the help in the development of this research.
Cite this paper
de Oliveira Canuto, D. S., da Silva, A. M., Freitas, M. L. M., Sebbenn, A. M., & de Moraes, M. L. T. (2017). Genetic Variability in Myracrodruon urundeuva (Allemão) Engl. Progeny Tests. Open Journal of Forestry, 7, 1-10. http://dx.doi.org/10.4236/ojf.2016.71001
References
- 1. Aguiar, A. V., Sousa, V. A., & Shimizu, J. Y. (2010). Genetic Selection Pinus gregii Progeny to the Establishment of Seed or Chards. Pesquisa Florestal Brasileira, 30, 107-117.
https://doi.org/10.4336/2010.pfb.30.62.107 [Paper reference 1] - 2. Bertoni, J. E., & Dickfeldt, E. P. (2007). Planting of Myracrodruon urundeuva Fr. All. (Aroeira) in Alteredarea of Forest: Seedling Development and Forest Restoration. Revista do Instituto Florestal, 19, 31-38. [Paper reference 1]
- 3. Brandão, M. (2000). Caatinga. In M. P. Mendonça, & L. V. Lins (Orgs.), Redlist of Endangered Species of Minas Gerais Flora (pp. 75-85). Belo Horizonte: Fundação Biodiversitas e Fundação Zôo-Botanica de Belo Horizonte. [Paper reference 1]
- 4. BRASIL (2008) Normative Instruction No. 06 of 23 September. Ministry of the Environment.
http://www.mma.gov.br/estruturas/179/_arquivos/179_05122008033615.pdf [Paper reference 1] - 5. Carvalho, P. E. R. (2003). Aroeira-Verdadeira (Circular técnico 82). Colombo: Empresa Brasileira de Pesquisa Agropecuária—Centro Nacional de Pesquisas Florestais. [Paper reference 1]
- 6. Carvalho, P. E. R. (1994). Brazilian Forest Species: Silvicultural Recommendations, Potentiality and Use of Wood (640 p). Colombo: Empresa Brasileira de Pesquisa Agropecuária—Centro Nacional de Pesquisas Florestais. [Paper reference 1]
- 7. Costa, R. B., Resende, M. D. V., Contini, A. Z., Roa, R. A., & Martins, W. J. (2005). Genetic Evaluation of Leucaena leucocephala [(Lam.) De Wit] Progenies in Indigenous Reserve, in Caarapó City, MS. Floresta e Ambiente, 12, 15-21. [Paper reference 1]
- 8. Costa, R. B., Resende, M. D. V., Gonçalves, P. S., Roa, R. A. R., & Feitosa, K. C. O. F. (2010). Genetic Parameters and Values Prediction for Growth and Latex Production Traits in Rubber Tree Progenies. Bragantia, 69, 49-56.
https://doi.org/10.1590/S0006-87052010000100007 [Paper reference 1] - 9. Ettori, L. C., Figliolia, M. B., & Sato, A. S. (2006). Ex Situ Conservation of Genetic Resources of Native Species: The Current Situation in the Forest Institute. In A. R. Higa, & L. D. Silva (Eds.), Groves of Native Species Seeds (pp. 203-226). Curitiba: FUPEF. [Paper reference 1]
- 10. Falconer, D. S. (1987). Introduction to Quantitative Genetics (279 p). Viçosa: Imprensa Universitária. [Paper reference 2]
- 11. Fonseca, A. J., Moraes, M. L. T., Aguiar, A. V., & Lacerda, A. C. M. B. (2003). Genetic Variation in Progenies of Two Populations of Myracrodruon urundeuva Fr. All. Inagroforestry System. Revista do Instituto Florestal, 15, 97-107. [Paper reference 1]
- 12. Freitas, M., Aukar, A., Sebbenn, A., Moraes, M., & Lemos, E. (2006). Geneticvariation in Progenies of Myracrodruon urundeuva F.F. & M.F. Allemao in Three Cultivation Systems. Revista árvore, 30, 319-329.
https://doi.org/10.1590/S0100-67622006000300001 [Paper reference 1] - 13. Freitas, M., Moraes, M., & Buzetti, S. (2002). Geneticvariation in Progenies of Myracrodruon urundeuva Fr. All. Indifferent Systems of Plantation. Revista do Instituto Florestal, 14, 133-141. [Paper reference 1]
- 14. Guerra, C., Moraes, M., Silva, C., Canuto, D., Andrade, J., Freitas, M., & Sebbenn, A. M. (2009). Selection Strategies within Progenies in Two Myracrodruon urundeuva Fr. All. Populations. Scientia Forestalis, 37, 079-087. [Paper reference 1]
- 15. Kageyama, P. Y., Cunha, G. C., Barreto, K. D., Camargo, F., & Sebbenn, A. M. (2003). Genetic Diversity and Spatial Autocorrelation in Ocotea odorifera (Lauraceae) Populations. Scientia Forestalis, 64, 108-119. [Paper reference 1]
- 16. Lorenzi, H. (1992). Brazilian Trees: Manual of Identification Manual and Cultivation of Arboreal Plants in Brazil (p. 149). Nova Odessa: Plantarum. [Paper reference 1]
- 17. Mendonça, M. P., & Lins, L. V. (Orgs.) (2000). Red List of Endangered Species of Flora of Minas Gerais (157 p). Belo Horizonte: Fundação Biodiversitas. [Paper reference 1]
- 18. Missio, R. F., Silva, A. M., Dias, L., Moraes, M., & Resende, M. (2005). Estimates of Genetic Parameters and Prediction of Additive Genetic Values in Pinus kesya Progenies. Crop Breeding and Applied Biotechnology, 5, 394-401.
https://doi.org/10.12702/1984-7033.v05n04a04 [Paper reference 1] - 19. Moraes, M., Missio, R. F., Silva, A. M., Cambuim, J., Santos, L. A., & Resende, M. (2007). Effect of Selective Thinning on the Genetic Parameters Estimates in Pinus caribaea Morelet var. Hondurensis Progenies. Scientia Forestalis, 35, 55-65. [Paper reference 1]
- 20. Oliveira, F. P., Fernandes Filho, E. I., Souza, A. L., & Soares, V. P. (2015). Mapping of Forests Monodominated by Myracrodruon urundeuva through Landsat/TM5 and Rapideye Images. Floresta e Ambiente, 22, 322-333.
https://doi.org/10.1590/2179-8087.090114 [Paper reference 1] - 21. Oliveira, V. R., Resende, M., Nascimento, C., Drumond, M. A., & Santos, C. (2004). Genetic Variability of Provenances and Progenies of Umbu Tree by Mixed Linear Model Methodology (REML/BLUP). Revista Brasileira de Fruticultura, 26, 53-56.
https://doi.org/10.1590/S0100-29452004000100015 [Paper reference 1] - 22. Pereira Jr., L. R., Alberício Pereira de Andrade, A., Araújo, K. D., Barbosa, A. S., & Barbosa, F. M. (2014). Caatinga Species as an Alternative to the Development of New Phytochemicals. Floresta e Ambiente, 21, 509-520. [Paper reference 1]
- 23. Pimentel Gomes, F. (1987). Experimental Statistic Scourse (12th ed., 466 p). São Paulo: Nobel. [Paper reference 1]
- 24. Pimentel Gomes, F. (1991). Index Variation: An Advantageous Substituteco Efficient of Variation (4 p). Piracicaba: IPEF. [Paper reference 1]
- 25. Pimentel Gomes, F. (1985). Experimental Statistic Scourse. Piracicaba: ESALQ/USP. [Paper reference 1]
- 26. Pimentel Gomes, F., & Garcia, C. H. (2002). Statistics Applied to Agronomic and Forestry Experiments: Exhibition with Examples and Guidelines for Application Use (309 p). Piracicaba: FEALQ. [Paper reference 1]
- 27. Resende, D. V., & Duarte, J. B. (2007). Precision and Quality Control in Varietytrials. Pesquisa Agropecuária Tropical, 37, 182-194. [Paper reference 1]
- 28. Resende, M. (2002a). Biometric and Statistical Genetics in Breeding Perennials (975 p). Brasília: EMBRAPA Informação Tecnológica. [Paper reference 1]
- 29. Resende, M. (2002b). Software SELEGEN-REML/BLUP (67 p). Colombo: EMBRAPA-CNPF. [Paper reference 1]
- 30. Resende, M., Souza, S. M., Higa, A. R., & Stein, P. P. (1991). Study of Genetic Variation and Selection Methods in Acacia Mearnsii Progeny Test in Rio Grande do Sul. Boletim de Pesquisa Florestal, 22/23, 45-59. [Paper reference 1]
- 31. Ribeiro, J. H. (1989). Aroeira: Durable and a Life. Globo Rural, 5, 85-90. [Paper reference 1]
- 32. Sebbenn, A. M. (2003). Samplesize for ex Situ Conservation in Forestry Tree Species with Mixed-Mating System. Revista do Instituto Florestal, 15, 147-162. [Paper reference 1]
- 33. Sebbenn, A. M., & Ettori, L. C. (2001). Ex Situ Genetic Conservation of Esenbeckia leiocarpa, Myracrodruon urundeuva and Peltophorum dubium in Combined Test of Progenies. Revista do Instituto Florestal, 13, 201-211. [Paper reference 2]
- 34. Siqueira, A., Sebbenn, A. M., Ettori, L. C., & Nogueira, J. (2000). Genetic Variation among and within Populations for ex Situ Conservation of Balfourodendron riedelianum (Engler) Engler. Revista do Instituto Florestal, 12, 89-103. [Paper reference 1]
- 35. Souza, R. F., Gomes, D. S., Leite, A. P., Santos, S. S., Alves, C., & Lucena, R. (2012). Ethnobotanical Study Myracrodruon urundeuva Allemãoin Thevalley of Piancó (Paraíba, Nordeste, Brasil). Revista de Biologia e Farmácia, 7, 72-83. [Paper reference 1]
- 36. Souza, C. S., Aguiar, A. V., Silva, A. M., & Moraes, M. (2003). Genetic Variation in Progenies of Astronium fraxinifolium Schott in Two Planting Systems. Revista do Instituto Florestal, 15, 137-145. [Paper reference 1]
- 37. Storck, L., Garcia, D. C., Lopes, S. J., & Estefanel, V. (2000). Vegetable Experimentation (198 p). Santa Maria: UFSM. [Paper reference 1]
- 38. Sturion, J. A., & Resende, M. (2005). Efficiency of Experimental Design and Testing Capability in Breeding Erva-Mate (Ilex paraguariensis St. Hil.). Boletim de Pesquisa Florestal, 3-10. [Paper reference 1]
- 39. Vencovsky, R., & Barriga, P. (1992). Genetics Biometrics in Plant Breeding (496 p). Ribeirão Preto: Sociedade Brasileira de Genética. [Paper reference 1]


