Open Journal of Forestry
Vol.05 No.03(2015), Article ID:54576,4 pages
10.4236/ojf.2015.53022
Serpentinite Slurries against Forest Fires
R. D. Schuiling
Institute of Geosciences, Utrecht University, Utrecht, The Netherlands
Email: schuiling@geo.uu.nl
Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 8 February 2015; accepted 7 March 2015; published 12 March 2015
ABSTRACT
Forest fires are one of the commonest natural hazards. Forest fires make the largest contribution to CO2 emissions after the burning of fossil fuels. Here a new technology is proposed to extinguish forest fires not with water, but with a slurry of serpentine. Serpentinites are abundantly available in many countries on every continent. If serpentine is calcined, it weathers very fast and captures CO2. Calcination, however, requires a lot of heat, which makes it counterproductive to produce calcined serpentine for CO2 capture. In cases, however, where heat is the problem, like in forest fires, one can extinguish them to greater advantage by using serpentinite slurries instead of plain water. The calcined residue that is left as a thin cake on the burning material prevents oxygen to reach the burning material. It also prevents the escape of inflammable gases, and the calcination itself withdraws large quantities of heat from the fire. After the fire is extinguished, the calcined material in contact with the atmosphere will rapidly weather and capture CO2. This compensates part of the CO2 that is produced by the fire. In tests, where the efficacy of quenching fires with serpentine slurries was compared to the effect of water, it turned out that serpentinite slurries performed far better.
Keywords:
Forest Fires, Calcination, Endothermic Reaction, Serpentinite Slurries, CO2 Capture

1. Introduction
Rising CO2 levels in the atmosphere are considered by many to cause a climate change. Their role in causing ocean acidification is unambiguous. The Earth is continuously degassing, and CO2 is emitted by volcanoes and by the dissociation of limestones carried to great depth in subduction zones ( Schuiling, 2004 ). There must exists an efficient feedback system, otherwise the Earth atmosphere and climatic conditions would be like on Venus, which has an atmosphere of 85 bars CO2 pressure, and a surface temperature of 460˚C. This feedback mechanism is the weathering of basic silicates, which can take place on Earth thanks to the presence of liquid water, which is lacking on Venus.
The natural choice for the best method to counteract climate change and ocean acidification is mineral carbonation ( Schuiling and Krijgsman, 2006 ). During the reaction of basic silicates with carbonic acid and water (a process known as weathering), CO2 is transformed into bicarbonate ions. The bicarbonate solutions are carried by rivers to sea, where they form carbonate sediments (limestones and dolomites). These carbonates represent the ultimate and sustainable storage of CO2 (Figure 1).
During the entire geological history, volcanoes on Earth have emitted CO2. Weathering has kept the CO2 levels of the atmosphere within reasonable and livable bounds. There have been fluctuations, depending on the rate of geological processes like mountain building and volcanism ( Raymo and Ruddiman, 1992 ), but the advantage of weathering is that it has an inbuilt negative feedback. The higher the CO2 pressure in the atmosphere, the more acid the waters in equilibrium with this atmosphere, and the faster the rate of weathering by which atmospheric CO2 levels are reduced. At present, by burning in a few hundred years the fossil fuels that have taken hundreds of millions of years to form, the CO2 level of the atmosphere is rapidly rising and the oceans are acidifying, because the weathering process cannot keep pace with the greatly increased CO2 emission, which is 30 to 60 times larger than the natural emission of CO2.
Olivine and serpentine weather easily and are abundantly available, close to the surface, where they can be mined in open-pit mines. By milling these rocks, and spreading the crushed material over land, shallow sea and beaches, the process of weathering can be enhanced, leading to a new balance between emission and capture ( Schuiling et al., 2011 ). Next to these major applications, minerals like olivine or serpentine can serve the same purpose in a number of niche applications. One such niche application is the use of serpentine slurries to quench fires. The residual solids weather fast and capture CO2. This compensates for the CO2 that is produced during the fire. This way quenching forest fires with serpentine slurries becomes an environmental technology that can help to manage the climate. The environment not only provides the material to be used, but also imposes the conditions under which it will be applied ( Schuiling, 1998 ).
Figure 1. The cliffs of Dover are one of the places where nature has stored its CO2.
2. Study Approach
2.1. Heat Withdrawal
If one knows the heat capacity of water and serpentine as a function of temperature ( Robie et al., 1978 ), it is straightforward to calculate the total heat that is withdrawn from a fire when a certain volume of a mixture of serpentine and water is sprayed over it. One needs, of course, also take into account the heats of vaporization of the water, and the heat of dissociation of the serpentine. The last can be calculated if the mineralogical composition of the end-product is known. Equilibrium phases would be olivine, enstatite and water vapor, but in reality this equilibrium is not reached in the short period in the fire, but rather some more or less amorphous or poorly crystalline intermediate materials. This means that the calculated heat of dissociation should be increased by an unknown, but rather modest amount equivalent to the difference in heat content between crystalline compounds and their amorphous equivalents. This makes serpentine slightly more efficient than would follow from the equilibrium calculation, where data for crystalline materials and pure water were used. Mixes of 40 wt% of serpentine and 60% of water are still quite liquid and easy to pump. The total heat withdrawn by this mixture when heated from room temperature to 900 K was compared to that of pure water over the same temperature interval. It turns out that the serpentine slurry withdraws 27% more heat than an equivalent volume of water.
2.2. Quenching Tests
Several quenching tests were carried out at the test site of Brand beveiliging (Fire Protection) BV in Wijchen/the Netherlands to observe the effects of spraying fires with serpentine slurries, and compare these to quenches with water. It was found that fires were considerably faster quenched with serpentinite than with water. With serpentine slurries, the effect was quasi-immediate. Moreover, a fire quenched with water had the tendency to repeatedly start flaming again after the spraying was stopped. This is obvious in tv reports on large forest fires that are sprayed with water. In contrast, fires that were sprayed with serpentinite slurries were quenched permanently. For this experiment, serpentine powder from a serpentinite quarry near Leoben (Austria) was used. A previous quenching test was carried out with a serpentine-rich residue from the PASEK olivine mine in NW Spain, with similar results.
The difference in behavior of fires when quenched with either water of serpentinite slurries is certainly not entirely due to their different heat content. It was observed that the serpentinite covers the burning material with a thin baked layer. This skin prevents the escape of inflammable gases and the access of oxygen, and this effect is more important than the larger heat withdrawal. After the fire is quenched, this thin baked layer will rapidly dissolve, while it reacts very fast with CO2 and rainwater.
2.3. After the Fire
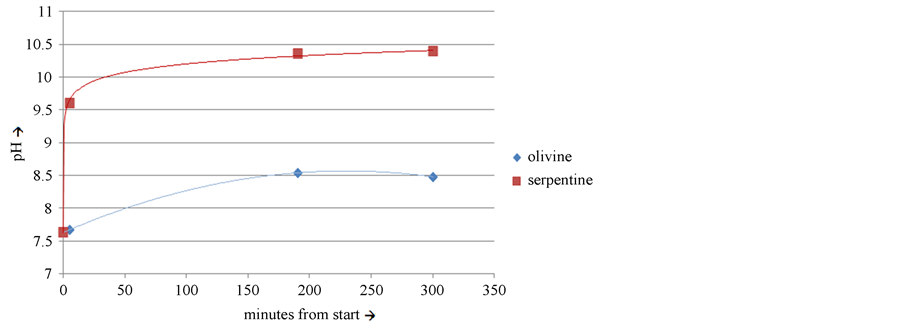
Once the fire is quenched, a baked serpentine residue is left. This is very reactive to mixtures of CO2 and water. Experiments with calcined serpentine that was alternately shaken in a closed bottle, and left standing open to the air showed that the reaction with water, and the absorption of CO2 was much more intense than the same treatment with fine-grained olivine. In fact, when calcined serpentine powder was first brought in contact with water, the pH rose within 5 minutes to 9.6 (Figure 2).
This is another major advantage of the use of serpentine slurries, they compensate rapidly a significant part of the CO2 that was emitted by the fire. An additional beneficial effect may be the release of magnesium, increasing the soil pH and helping trees to grow faster.
3. Results and Discussion
3.1. Applications
Possibly the best use of serpentinite slurries would be in forest fires, both on the ground as from the air. Dropping a load of serpentinite slurries from a plane or helicopter has the advantage that the slurry can have a higher percentage of serpentine than when it must be pumped, because for dropping a higher viscosity is permitted, as even a more viscous mud can be scooped out of a basin.
Another type of fire where the application of serpentine slurries might come in useful is in tunnels.
Figure 2. pH increase of suspensions of calcined serpentine and fine grained olivine.
3.2. Global CO2 Emissions from Forest Fires
The global CO2 emissions from forest fires are considerable. It is difficult to obtain data on how much CO2 is annually emitted by forest fires. There are some data from the USA, where it was calculated that the CO2 emission by bush fires was between 4% and 6% of the total CO2 emission from fossil fuels in the USA ( Thompson, 2007 ), which would mean about 275 million tons. This is of the same order as the total CO2 emissions by traffic in the USA. Liu Yongqiang et al. 2011 , arrive at a lower estimate of 90 million tons. A study for boreal Eurasia ( Shvidenko et al., 2011 ) arrives at 440 million tons of CO2 emissions annually from forest fires in boreal Eurasia. It is difficult to extrapolate these data to the whole world, but it is likely that the total of CO2 emissions from forest fires will exceed 3 Gt of CO2, as the largest contributions are from savannah fires in Africa, and forest fires in S America and SE Asia. In fact, Van der Werf et al., 2009 estimate that forest fires worldwide contribute 20% of global CO2 emissions, making it the second largest source after the burning of fossil fuels, but their estimate includes forest losses by deforestation.
If fire fighting by the use of serpentine slurries instead of water could save 10% of the forests lost by fire, this would mean a reduction in the order of 300 million tons of CO2 emitted annually. For the total reduction in CO2 emission one should add the amount of CO2 that is rapidly captured by the serpentine after it was calcined in the fires.
Using the common and cheap natural material serpentine instead of expensive fire retardants like diammonium phosphate which contributes nothing to CO2 capture is an obvious improvement over current fire fighting practice.
In countries with a warm and dry season, and frequent forest fires in summer, small permanent basins (ponds) should be constructed near large forests, where serpentine slurries are kept ready for emergencies. Another advantage in countries with a shortage of water is the fact that quenching with serpentinite slurries needs much less water than quenching with water only.
4. Conclusion
Serpentine slurries provide a cheap and effective environmental technology to quench widespread fires. Likely applications are in forest fires or tunnel fires. After the fire is ended, the baked residue of the serpentine dissolves fast by reaction with CO2 and water. By this reaction the greenhouse gas CO2 is converted to, and stored as innocent bicarbonate solutions. This compensates the CO2 that was emitted by the fire. The reduction of the number and extent of forest fires by using serpentine slurries to extinguish them together with the CO2 compensation by the rapid weathering of the calcined serpentine can lead to a significant reduction in global CO2 emissions. CO2 reduction is not the only advantage of this approach; it probably will save lives as well.
Acknowledgements
Thanks are due to J. van Ooij, K. Sickmann and M. Warnar, who participated in this research as part of their junior thesis at the Junior College Utrecht. I also wish to thank Javier Martinez Rubio from the PASEK olivine mine (Spain), and Jan Koller from the Isomag serpentinite quarry (Austria) for making serpentine samples available.
References
- Liu, Y . Q., Stanturf, J., Tian, H. Q., & Qu, J. (2011). CO2 Emissions from Wildfires in the U.S. Present Status and Future Trends.
- Raymo, M. E., & Ruddiman, W. F. (1992). Tectonic Forcing of Late Cenozoic Climate. Nature, 359, 117-121. http://dx.doi.org/10.1038/359117a0
- Robie, R. A., Hemingway, B. S., & Fisher, J. R. (1978). Thermodynamic Properties of Minerals and Related Substances at 298.15 K and 1 Bar (105 Pascals) Pressure and at Higher Temperatures. Geological Survey Bulletin, 1452, 456 p.
- Schuiling, R. D. (1998). Geochemical Engineering: Taking Stock. Journal of Geochemical Exploration, 62, 1-28. http://dx.doi.org/10.1016/S0375-6742(97)00042-3
- Schuiling, R. D. (2004). Thermal Effects of Massive CO2 Emissions Associated with Subduction Volcanism. Comptes Rendus Geoscience, 336, 1053-1059. http://dx.doi.org/10.1016/j.crte.2004.04.008
- Schuiling, R. D., Tickell, O., & Wilson, S. A. (2011). Climate Change and the KISS Principle. Poster Goldschmidt Conference Prague, 14-19 August 2011.
- Schuiling, R. D. and Krijgsman, P. (2006). Enhanced Weathering; an Effective and Cheap Tool to Sequester CO2. Climatic Change, 74, 349-354. http://dx.doi.org/10.1007/s10584-005-3485-y
- Shvidenko, A., Schepaschenko. D., Sukhinin, A., McCallum, I., & Maksyutov, S. (2011). Carbon Emissions from Forest Fires in Boreal Eurasia between 1998-2010. The 5th International Wildland Fire Conference, South Africa.
- Thompson, A. (2007). Wildfires Release as Much CO2 as Cars. Live Science.
- Van der Werf, G. R., Morton, D. C., DeFries, R. C., Olivier, J. G. J., Kasibhatla, P. S., Jackson, R. B., Collatz, G. J., & Randerson, J. T. (2009). CO2 Emissions from Forest Loss. Nature Geoscience, 2, 737-738. http://dx.doi.org/10.1038/ngeo671