World Journal of Neuroscience
Vol.4 No.2(2014), Article ID:44715,7 pages DOI:10.4236/wjns.2014.42010
The Sources of Extracellular Adenosine in Hippocampus and Neostriatum of the Rat Brain
M. A. Pak1, Y. Yetkin2*
1Physiologisches Institut, Heinrich-Heine-Universität Düsseldorf Postfach 101007, 40001 Düsseldorf, Germany
2Department of Physiology, Medical Faculty, Yüzüncü Yil University, Van, Turkey
Email: *yyetkin@yyu.edu.t
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 25 November 2013; revised 2 January 2014; accepted 15 January 2014
ABSTRACT
Endogenous adenosine can enter the extracellular space either by direct release or via formation from adenine nucleotides. We have compared the effects of substances blocking the ecto-5'-nucleotidase with (, (-methylene adenosine 5'-diphosphate (AOPCP) or adenosine kinase with iodotubercidin (Itu) on field potentials in the hippocampus and the neostriatum in vitro evoked by stimulation of the stratum radiatum or the cortico-striatal pathway respectively. AOPCP enhanced the amplitude of the population spikes by 34% in the hippocampus and by 26.5% in the neostriatum. DPCPX, a selective A1-receptor antagonist, increased the amplitude of the population spikes by 68% in the hippocampus and by 53.5% in the neostriatum. Thus both, release of adenosine from the intracellular space and, extracellular dephosphorylation of adenine nucleotides to a lesser extent, contribute to the effective levels of adenosine in the extracellular space in hippocampus and neostriatum.
Keywords:AOPCP, ecto-5'-nucleotidase, Iodotubercidin, DPCPX, Electrophysiology

1. Introduction
Adenosine is released metabolically from active brain cells and is generated extracellular by degradation of adenosine triphosphate (ATP) [1] , and to provide a scientific data for present research. It needs to explain how adenosine was released from sources in the striatum. Adenosine has long been recognised as an important neuromodulator with profound effects on activity in the central nervous system (CNS). The inhibitory effect on synaptic transmission in hippocampus and neostriatum is mediated by A1 adenosine receptors [2] , which are located both preand postsynaptically. Adenosine acts postsynaptically to induce or to increase potassium conductance’s [3] [4] and presynaptically to decrease the release of neurotransmitters such as glutamate [5] [6] . Adenosine is a normal constituent of all body fluids, including the extracellular fluid in the CNS, where it can regulate nervous activity. It has been proposed to be a sleep propensity factor [7] Adenosine levels are drastically elevated during hypoxia or ischemia [8] for further reference see [9] metabolic stress and seizures [10] , with the consequence of profound inhibition of neuronal excitability and synaptic transmission. Intraand extracellular adenosine concentrations are kept in equilibrium by means of bidirectional nucleoside transporters. Some of these carrier mechanisms can be inhibited by drugs such as dipyridamole, nitrobenzylthioinosine and dilazep [11] [12] . The direction of adenosine transport depends on its concentration gradient between the cytosol and the extracellular space.
The intracellular level of adenosine is regulated by several enzymes. Adenosine kinase phosphorylates intracellular adenosine and keeps the intracellular concentration low. The basal extracellular levels of adenine nucleotides are low. Adenine nucleotide is released in the extracellular space and rapidly hydrolysed by ecto-nucleotidases to form adenosine. The nucleotide catabolizing enzyme ecto-5'-nucleotidase is abundant in the central nervous system [13] -[15] . The relevance of adenosine kinase, adenosine deaminase and SAHH (S-Adenosylhomocysteine hydrolase) activity for adenosine levels has been investigated previously [16] -[18] .
2. Methods
We have now examined the relative importance of the two sources of extracellular adenosine in neural tissue: extracellular adenine nucleotides and adenosine released from the intracellular space. We have determined the effect of ectonucleotidase and adenosine kinase on field potentials from hippocampal and neostriatal slices, in order to estimate the relative contributions of adenosine direct release and nucleotides as sources of extracellular adenosine.
We have used a, b-methylene adenosine 5'-diphosphate (AOPCP), a potent inhibitor of ecto-5'-nucleotidase, 1, 3-dipropyl-8-cyclopentylxanthine (DPCPX), a selective A1-receptor antagonist, and iodotubercidin (Itu), a potent and selective inhibitor of adenosine kinase to study this question electrophysiologically in hippocampal and striatal slices.
Young adult Wistar rats were decapitated, and the brains were rapidly removed and placed in ice-cold artificial cerebrospinal fluid (ACSF) of the following composition (mM): NaCl 124, KCl 3, MgSO4 1.25, CaCl2 2.5, KH2PO4 1.25, NaHCO3 22, Glucose 10. Transverse slices of 400 mm thickness were prepared from the hippocampus using a McIlwain tissue chopper. Oblique slices of 400 mm thickness, about 40 degrees rostral-up to the horizontal plane, were cut from the neostriatum using a vibratome. The slices were kept in ACSF at room temperature and gassed continuously with a mixture of 95% O2 and 5% CO2 to attain a steady-state oxygenation level and maintain a pH of 7.4. Slices were allowed to recover at least 1h before recording began at 33˚C.
Field potentials were recorded in the stratum pyramidal of area CA1 following electrical stimulation of the Schaffer collateral pathway in the hippocampus. In the striatum preparation, stimulation took place on the corpus callosum or the external capsule to activate the cortical inputs (0.1 Hz). Recording electrodes were glass micropipettes filled with 2M NaCl, resistance ranged between 6 and 10 MW. The stimulus strength was adjusted to evoke half-maximal field potentials which were averaged over 8 to 10 sweeps.
Data are presented as mean ± standard error of the mean (S.E.M.). For statistical analyses of the responses before, during and after drug application were compared using students’ test. All drugs were added to the perfusion fluid. Adenosine, AOPCP was obtained from Sigma (Deisenhofen, Germany) and DPCPX, iodotubercidin from Research Biochemicals Inc. (Natick, MA, USA).
3. Results
AOPCP (50 mM) significantly (p < 0.01) enhanced the amplitude of population spikes by 34% ± 17.5% (n = 11) in the hippocampus and by 26.5% ± 14% (n = 7) in the neostriatum. The effect of AOPCP was reversible within 10 to 20 minutes (Figure 1). This indicates that a portion of extracellular adenosine is derived from adenine nucleotides. The population spike enhancement by AOPCP was maximal at these concentrations.
Exogenously applied adenosine, at 40 µM, caused a larger decrease in amplitude of field potentials in both preparations: to 47.5% ± 12.5% of control (n = 5) in the hippocampus and 60% ± 15% of control (n = 5) in the neostriatum (Figure 2).
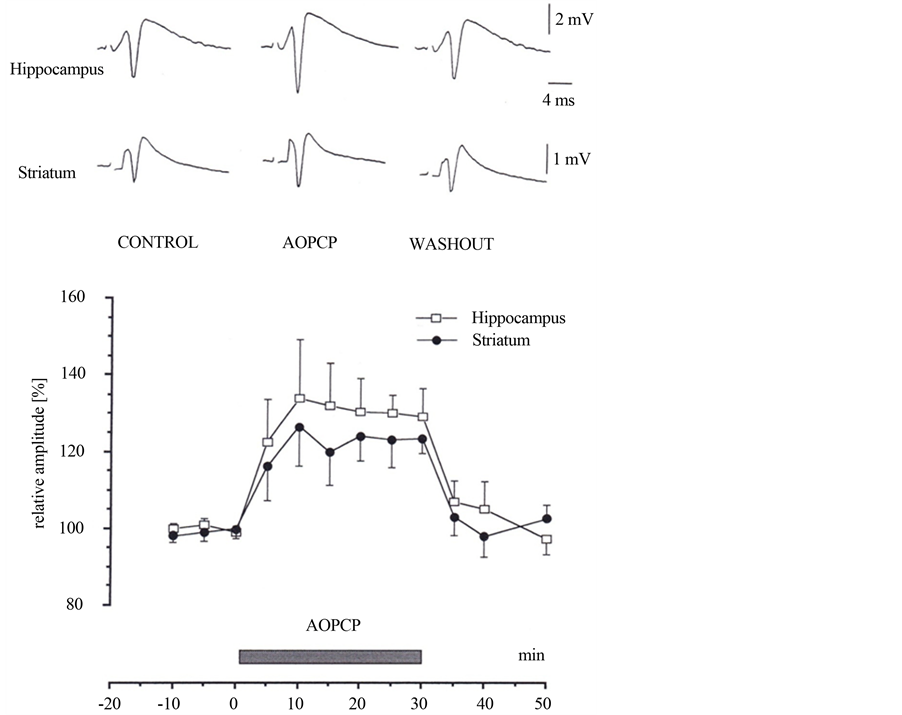
Figure 1. The Effects of a, b methylene adenosine 5'-diphosphate (AOPCP) on field potentials. Averaged field potentials recorded in the absence (control and washout) and during the application of AOPCP (40 mM). AOPCP enhanced the amplitude of the population spikes (mean ± S.E.M., n = 11 hippocampus and n = 7 striatum) in field potentials of hippocampus and neostriatum. The effect of AOPCP was reversible; the amplitude of population spikes was almost at the control level after a washout period of 10 to 20 minutes. Hippocampus: open circles, n = 11 and striatum: filled circles, n = 7.
DPCPX (5 mM), a selective adenosine A1-receptor antagonist [2] , increased the amplitude of population spikes by 68% ± 10.5% (n = 7) in hippocampus and 53.5% ± 7.5% (n = 5) in striatum (Figure 3). Comparison of the effect of the adenosine receptor antagonist DPCPX with the inhibitor of ectonucleotidase AOPCP shows in both structures statistically significant difference (paired t test, t = 4.61, p = 0.0003 for hippocampus, t = 3.89, p = 0.003 for striatum).
4. Discussion
In a previous study [17] we have demonstrated an adenosine mediated inhibitory effect of iodotubercidin, a potent inhibitor of adenosine kinase in the hippocampus. We report now a similar reversible effect by iodotubercidin (5 mM) in the neostriatum, by 72.5% ± 16.5% (n = 5) (Figure 4).
Inhibition of ecto-5'-nucleotidase with AOPCP enhances field potentials in superfused hippocampal slices by 35% and by 25% in neostriatal slices. Inhibition of the catabolizing enzyme prevents the degradation of the nucleotides to adenine nucleoside, thus leading to enhanced excitability of cells in the CA1 region of hippocampus and in neostriatum. AOPCP increased the amplitude of the population spikes in both preparations lesser that observed with an A1-receptor antagonist (Figure 1, Figure 3).
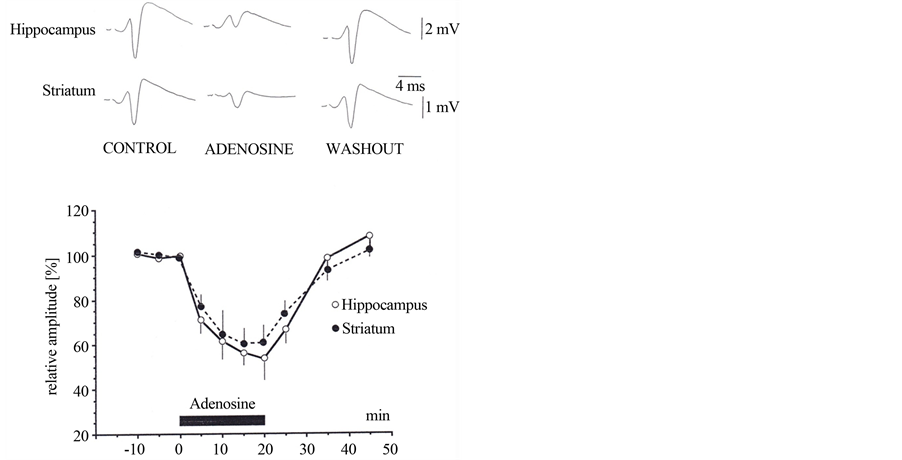
Figure 2. The effect of exogenous adenosine on field potentials recorded from the CA1 pyramidal cell layer in the hippocampus following stimulation of the stratum radiatum and from the striatum following stimulation of the cortico-striatal pathway. Normalised amplitude of averaged field potentials recorded in the absence (control and washout) and during the application of adenosine (40 mM) are displayed. The amplitudes are means of 5 population spikes in each case. Hippocampus: open circles, n = 5 and striatum: filled circles, n = 5.
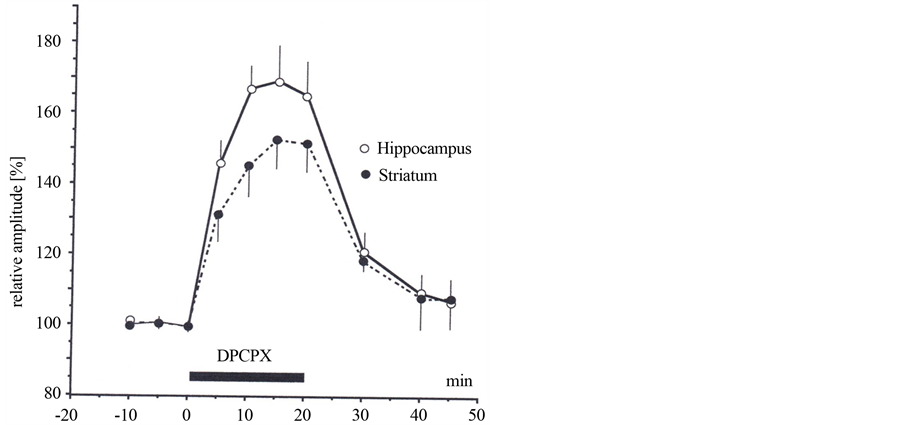
Figure 3. Effects of 1, 3-dipropyl-8-cyclopentylxanthine (DPCPX) on field potentials recorded from the CA1 pyramidal cell layer in the hippocampus following stimulation of the striatum radiatum and from the striatum following stimulation of the cortico-striatal pathway. Normalised amplitude of averaged field potentials recorded in the absence (control and washout) and during the application of DPCPX (5 mM) are displayed. Hippocampus: open circles, n = 7 and striatum: filled circles, n = 5.
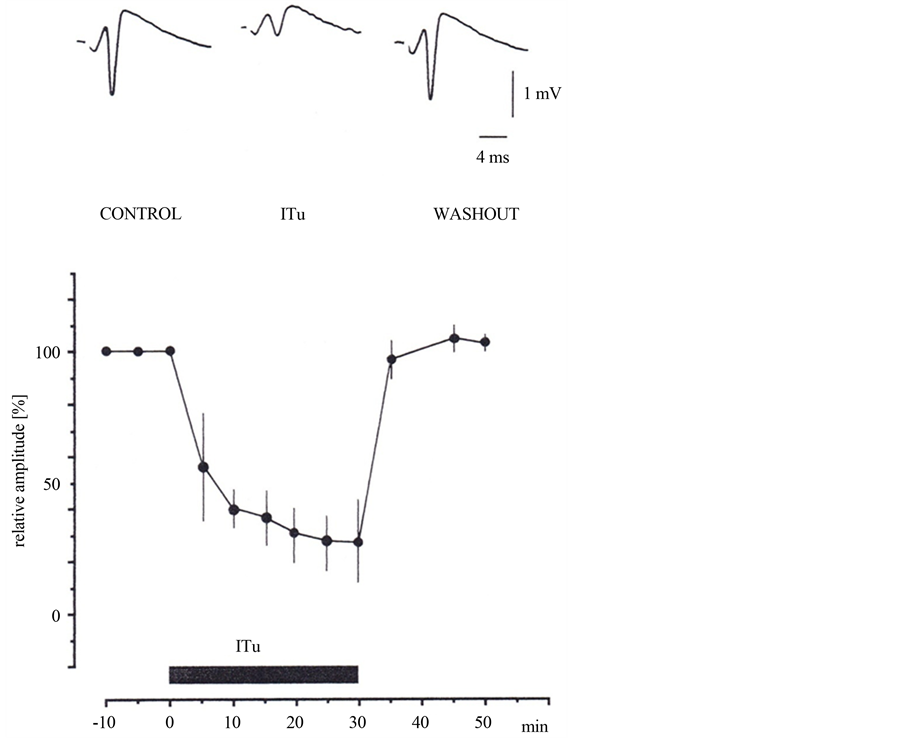
Figure 4. Effects of iodotubercidin (ITu) on field potentials in neostriatum. Averaged field potentials recorded in neostriatum in the absence (control and washout) and during the application of ITu (5 mM) following stimulation of the cortico-striatal pathway. The effect of ITu was reversible; the amplitude of population spikes was almost at the control level after a washout period of 10 to 20 minutes. The amplitudes are means of 5 population spikes.
The ecto-5'-nucleotidase is abundant on the surface of neuronal membranes [14] [15] [19] [20] There are at least two types of ectonucleotidases which are specific for different substrates: ecto-ATPase is specific for ATP, ecto-5'-nucleotidase hydrolyses both 5'-nucleoside and AMP. They are bound to the cell membrane and directed towards the extracellular space [21] [22] have shown that AOPCP blocks responses to the nucleotides but has no significant effects on responses to adenosine itself.
Intracellular adenosine is produced either by hydrolysis of AMP (5'-AMP) via cytosolic 5'-nucleotidase or by hydrolysis of SAH via SAH-hydrolase. Adenosine kinase, which phosphorylates adenosine to AMP, and adenosine deaminase, which deaminates adenosine to inosine, are both thought to be predominantly cytoplasmic enzymes [23] . Adenosine can cross cell membranes either by active transport or by facilitated diffusion, a carrier mediated process in which adenosine follows its concentration gradient [24] . The transporters operate bi-directionally, depending on the adenosine concentrations in the cytosol and the extracellular space.
Adenosine has a role as a modulator of synaptic activity in the CNS [25] . The modulation of the excitatory synaptic transmission is occurred by adenosine released from hippocampal pyramidal neurons [26] . Data from this and other studies [17] [18] [27] suggest that adenosine kinase plays a key role in the regulation of extracellular concentration of adenosine. Intracellular formation of adenosine is the most important source of adenosine in the extracellular space, but a significant amount comes from dephosphorylation of extracellular nucleotides.
References
- Kase, H., Richardsonand, P.J. and Jenner, P. (2000) Adenosin Receptors and Parkinson Disease. Academic Press, London.
- Bruns, R.F., Fergus, J.H., Badger, E.V., Bristol, J.A., Santay, L.A., Hartman, J.D., Hays, S.J. and Huang, C.C. (1987) Binding of the A1 Selective Adenosine Antagonist 8-Cyclopentyl-1,3-dipropylxanthine to Rat Brain Membranes. NaunynSchmiedeberg’s Archives of Pharmacology, 335, 59-63.
- Gerber, U., Greene, R.W., Haas, H.L. and Stevens, D.R. (1989) Characterization of Inhibition Mediated by Adenosine in the Hippocampus of the Rat in Vitro. The Journal of Physiology, 417, 567-578.
- Thompson, S.M., Haas, H.L. and Gahwiler, B.H. (1992) Comparison of the Actions of Adenosine at Preand Postsynaptic Receptors in the Rat Hippocampus in Vitro. The Journal of Physiology, 451, 347-363.
- Fastbom, J. and Fredholm, B.B. (1985) Inhibition of [3H] Glutamate Release from Rat Hippocampal Slices by Lphenylisopropyladenosine. Acta Physiologica, 125, 121-123. http://dx.doi.org/10.1111/j.1748-1716.1985.tb07698.x
- Dunwiddie, T.V. (1985) The Physiological Role of Adenosine in the Central Nervous System. International Review of Neurobiology, 27, 63-139. http://dx.doi.org/10.1016/S0074-7742(08)60556-5
- Huston, J.P., Haas, H.L., Boix, F., Pfister, M., Decking, U., Schrader, J. and Schwarting, R.K. (1996) Extracellular Adenosine Levels in Neostriatum and Hippocampus during Rest and Activity Periods of Rats. Neuroscience, 73, 99- 107. http://dx.doi.org/10.1016/0306-4522(96)00021-8
- Winn, H.R., Rubio, R. and Berne, R.M. (1981) Brain Adenosine Concentration during Hypoxia in Rats. American Journal of Physiology, 241, 235-242.
- Chao, D. and Xia, Y. (2010) Ionic Storm in Hypoxic/Ischemic Stress: Can Opioid Receptors Subside It? Progress in Neurobiology, 90, 439-470. http://dx.doi.org/10.1016/j.pneurobio.2009.12.007
- During, M.J. and Spencer, D.D. (1992) Adenosine: A Potential Mediator of Seizure Arrest and Postictal Refractoriness. Annals of Neurology, 32, 618-624. http://dx.doi.org/10.1002/ana.410320504
- Deckert, J., Morgan, P.F. and Marangos, P.J. (1988) Adenosine Uptake Site Heterogeneity in the Mammalian CNS? Uptake Inhibitors as Probes and Potential Neuropharmaceuticals. Life Sciences, 42, 1331-1345. http://dx.doi.org/10.1016/0024-3205(88)90162-2
- Lee, C.W. and Jarvis, S.M. (1988) Nucleoside Transport in Rat Cerebral-Cortical Synaptosomes. Evidence for Two Types of Nucleoside Transporters. Biochemical Journal, 249, 557-564.
- Richardson, P.J., Brown, S.J., Bailyes, E.M. and Luzio, J.P. (1987) Ectoenzymes Control Adenosine Modulation of Immunoisolated Cholinergic Synapses. Nature, 327, 232-234. http://dx.doi.org/10.1038/327232a0
- Zimmermann, H., Vogel, M. and Laube, U. (1993) Hippocampal Localization of 5'-Nucleotidase as Revealed by Immunocytochemistry. Neuroscience, 55, 105-112. http://dx.doi.org/10.1016/0306-4522(93)90458-R
- Magalhães-Cardoso, M.T., Pereira, M.F., Oliveira, L., Ribeiro, J.A., Cunha, R.A. and Correia-de-Sá, P. (2003) EctoAMP Deaminase Blunts the ATP-Derived Adenosine A2A Receptor Facilitation of Acetylcholine Release at Rat Motor Nerve Endings. The Journal of Physiology, 549, 399-408. http://dx.doi.org/10.1113/jphysiol.2003.040410
- Lloyd, H.G., Lindstrom, K. and Fredholm, B.B. (1993) Intracellular Formation and Release of Adenosine from Rat Hippocampal Slices Evoked by Electrical Stimulation or Energy Depletion. Neurochemistry International, 23, 173-185. http://dx.doi.org/10.1016/0197-0186(93)90095-M
- Pak, M.A., Haas, H.L., Decking, U.K. and Schrader, J. (1994) Inhibition of adenosine Kinase Increases Endogenous Adenosine and Depresses Neuronal Activity in Hippocampal Slices. Neuropharmacology, 33, 1049-1053. http://dx.doi.org/10.1016/0028-3908(94)90142-2
- Lloyd, H.G. and Fredholm, B.B. (1995) Involvement of Adenosine Deaminase and Adenosine Kinase in Regulating Extracellular Adenosine Concentration in Rat Hippocampal Slices. Neurochemistry International, 26, 387-395. http://dx.doi.org/10.1016/0197-0186(94)00144-J
- Fastbom, J., Pazos, A. and Palacios, J.M. (1987) The Distribution of Adenosine A1 Receptors and 5'-Nucleotidase in the Brain of Some Commonly Used Experimental Animals. Neuroscience, 22, 813-826. http://dx.doi.org/10.1016/0306-4522(87)92961-7
- Latini, S. and Pedata, F. (2001) Adenosine in the Central Nervous System: Release Mechanisms and Extracellular Concentrations. Journal of Neurochemistry, 79, 463-484.
- Zimmermann, H. (1996) Biochemistry, Localization and Functional Roles of Ecto-Nucleotidases in the Nervous System. Progress in Neurobiology, 49, 589-618. http://dx.doi.org/10.1016/0301-0082(96)00026-3
- Dunwiddie, T.V., Diao, L. and Proctor, W.R. (1997) Adenine Nucleotides Undergo Rapid, Quantitative Conversion to Adenosine in the Extracellular Space in Rat Hippocampus. Journal of Neuroscience, 17, 7673-7682.
- Geiger, J.D. and Fyda, D.M. (1991) Adenosine Transport in Nervous Tissues. In: Stone, T.W., Ed., Adenosine in the Nervous System, Academic Press, London, 1-23. http://dx.doi.org/10.1016/B978-0-12-672640-4.50007-8
- Bender, A.S., Wu, P.H. and Phillis, J.W. (1981) The Rapid Uptake and Release of [3H] Adenosine by Rat Cerebral Cortical Synaptosomes. Journal of Neurochemistry, 36, 651-660. http://dx.doi.org/10.1111/j.1471-4159.1981.tb01638.x
- Brundege, J.M. and Dunweddie, T.V. (1997) Role of Adenosine as a Modulator of Synaptic Activity in the Central Nervous System. Advances in Pharmacology, 39, 353-391. http://dx.doi.org/10.1016/S1054-3589(08)60076-9
- Brundege, J.M. and Dunweddie, T.V. (1996) Modulation of the Excitatory Synaptic Transmission by Adenosine Relased from Single Hippocampal Pyramidal Neurons. Journal of Neuroscience, 16, 5603-5612.
- Sperlagh, B. and Vizi, E.S. (2011) The Role of Extracellular Adenosine in Chemical Neurotransmission in the Hippocampus and Basal Ganglia: Pharmacological and Clinical Aspects. Current Topics in Medicinal Chemistry, 11, 1034- 1046. http://dx.doi.org/10.2174/156802611795347564
NOTES

*Corresponding author.

