Open Journal of Ecology
Vol.05 No.08(2015), Article ID:58631,8 pages
10.4236/oje.2015.58028
Silver Carp (Hypophthalmichthys molitrix, Val. 1844) Stocking in Lake Kinneret (Israel)
Moshe Gophen1, Gregory Snovsky2
1Migal-Scientific Research Institute, Kiryat Shmone, Israel
2The Fishery Department, Israeli Ministry of Agriculture and Agricultural Settlements Development, Tiberias, Israel
Email: Gophen@Migal.org.il, Gregorys@moag.gov.il
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 1 July 2015; accepted 1 August 2015; published 6 August 2015
ABSTRACT
Silver Carp (SC) (Hypophthalmichthys molitrix, Valenciennes, 1844) is a worldwide common fish for aquaculture and stocking in lakes and reservoirs. The fish is distributed through rivers, aquaculture and stocking in about 90 countries around the world. SC was first introduced into
Keywords:
Silver Carp, Kinneret, Introduction, Growth, Feeding

1. Introduction
The origin of the Silver Carp (SC) (Hypophthalmichthys molitrix, Valenciennes, 1844) is the Amur River, the world’s tenth longest river forming the border between Far East region of Russia and Northeastern China. SC is a freshwater cyprinid native in China and Eastern Siberia. This fish is a worldwide common organism for aquaculture and stocking in lakes and reservoirs. The fish was introduced into lakes and distributed through rivers over about 90 countries around the world. This successful distribution and usage is due to its high rate of growth, economical factor of food conversion and beneficial protection of water quality. SC has been first introduced into Lake Kinneret in 1969 and continues onwards. The objective of the mass stocking of SC in Lake Kinneret was primarily aimed at water quality improvement by the production of grazing pressure on the annual flourishing of the Pyrrhophyte algae, Peridinium together with enhancing Fishery catches. Total number of stocked fingerlings (1969-2013) was 18.5 × 106, averaged as 441 × 103 per year. The formal annual statistical summaries submitted (in Hebrew) by the Israeli Ministry of Agriculture and Agricultural Settlements Development, Fishery Department, Lake Kinneret Fishery Branch, reported that the total catch of SC during 1972-2013 was 3218 tons with annual average of 75 tons, which was app. 4% of the total landings. The adult life stages of SC are known to be phytoplanktivores whilst young fingerlings mostly prey zooplankton. The ability of SC to consume Cyanophyta is also known. The efficient growth, accompanied by phytoplankton (especially Cyanophytes) consumption, makes this fish suitable for planting in lakes aimed at fishery improvement and water quality protection. The objective of this paper is to promote an insight into practical use of SC in lake fisheries management design.
1.1. Remarks on Taxonomy
A worldwide usage of the title Asian Carp practically include several species: 1) Beaghead Carp, Aristichthys nobilis (Namroon, in Hebrew) and Hypophthalmichthys nobilis (Sass et al., 2014); 2) Silver Carp, Hypophthalmichtys molitrix (Kasif, in Hebrew); 3) Hybridization between Bighead and Silver Carps (Namsif, in Hebrew). These three carps are different in morphology, ecological behavior, (swimming, jumps) reproduction and feeding habits. Our paper refers only to Silver Carp (Hypophthalmychthys molitrix, SC).
1.2. Bighead Carp, (Asian Carp), Aristichthys nobilis Invasion in USA
The USFWS (USA-Fish and Wildlife Service) [1] publicized the following on March 21st, 2011: The U.S. Fish and Wildlife Service published a final rule in the Mar. 22 Federal Register officially adding the bighead carp to the federal injurious wildlife list. The final rule codifies the Asian Carp Prevention and Control Act (S. 1421), signed into law by President Obama on Dec. 14, 2010. The injurious wildlife listing means that under the Lacey Act it is illegal to import or to transport live bighead carp, including viable eggs or hybrids of the species, across state lines, except by permit for zoological, education, medical, or scientific purposes. Bighead carp were imported from eastern China to Arkansas in the 1970s to improve water quality in aquaculture ponds and sewage treatment lagoons. The fish, which can grow to 60 or more pounds, have since spread through the Mississippi River basin, Illinois River [2] and have been collected as far north as Lake Pepin in Minnesota. Because of their large size and abundance, Bighead carp routinely out-compete native fish for food. If Bighead carp enter the Great Lakes and become established, they potentially threaten the 1.5 million jobs and $62 billion in wages connected to the Great Lakes [1] . Nevertheless the Bighead invasion is distributed mostly within 13 states in the catchment of the Mississippi. The Bighead carp injurious wildlife listing is just one of many steps the federal government is taking to protect the country’s aquatic ecosystems from Asian carp. On December 16, 2010, the Asian Carp Regional Coordinating Committee (RCC) released an updated version of the Asian Carp Control Strategy Framework. The RCC represents a state and federal partnership dedicated to stopping the spread of all types of injurious Asian carp, including Bighead, into the Great Lakes. Presently, the invasion of Bighead Carp into the Mississippi River system and Northern USA is one of the USFWS major concerns of water quality and fishery protection in Northern USA. Nevertheless the injurious fish is Bighead Carp. Due to the close relation of all Asian Carps fishes a great caution has to be taken when Silver Carp introduction is proposed.
1.3. Silver Carp Studies in East Lake, China [3]
The food-web ecosystem of East Lake in Wuhan City, Hubei Province in China is characterized as phytoplanktivorous dominated food-chain, where >90% of total fish yield is due to Silver and Bighead Carps. The lake is annually stocked by fingerlings of those two carps and prominently affected an increase of landings (670 - 750 Kg /ha) [3] compared to SC yield of 4 - 5 kg/ha in lake Kinneret. Chinese researchers defined SC as phytoplanktivore whilst Bighead carp as an omnivore. The East Lake research project prominently confirmed two major issues: 1) The significant merit of SC stocking to the commercial yield; and 2) The potential impact of SC stocking on water quality through suppressing and reduction of Cyanobacteria (mostly Microcystis) biomass. After the initiation of Carps socking program the commercial landings comprised 80% - 90% Carps of total catch [4] . The feeding habits of the carps [5] indicate that in the presence of high biomass of SC, Microcystis was the main food resource, this Cyanophyte was suppressed and did not become dominant component of algal biomass. Then, the biomass of small chlorophytes and diatoms became dominant. This nano-phytoplankton served as an optimal food for the enhanced grazer zooplankton. Chen [6] estimated in East Lake that in the diet of SC 93% in summer (July-September, the most productive months) was occupied by Microcystis of which 39.2% of the Nitrogen was assimilated. Only 7% of the gut content was due to zooplankton and the efficiency of Nitrogen assimilation was 33.5%. In fall season the portion of phytoplankton component in the gut content was slightly lowered to 80% and that of zooplankton was increased to 20%. [7] [8] documented metabolic parameters (gN per individual fish) of aged 1+, 2+ and 3+ SC during 1983- 1987 in East Lake: the percentage of consumed phytoplankton varied between 83% - 91% (the rest is Zooplankton), the total quantity of consumed food increased with age increment (1+ to 3+ age) whilst assimilation coefficient and specific growth increment declined. This decline was suggested to be resulted by the seasonal change of the algal biomass composition from Microcystis to green algae (Chlorophyta) dominance. The “greens” (small size, nanoplanktonic chlorophytes and Diatoms) are less suitable as food to SC. From the view point of water quality, managers prefer a lake ecosystem with high biomass of the “greens” accompanied by grazer zooplankton which is a better composition than Microcystis dominance and low biomass of cladocerans. Moreover, Opuzsinsky [9] documented an increase of the “greens” and decline of large colonial Cyanobacteria, in the presence of SC in fishponds in Poland. Iwata et al. [10] measured uptake of 12C and 13C (mgC/l/day) by the lake algal communities before Microcystis dominance and after when “green” algae dominated as a result of the introduction of SC in East Lake: 12C uptake increased by 1380% and 13C uptake decline by 63%, i.e. the ratio of 12C /13C was 0.479 and 19.1 before and after SC stocking respectively. Consequently, the SC removed efficiently the “heavy”, better 13C incorporator algae, Microcystis, and the lighter, better 12C up-taker, the “greens” became dominant.
2. The Silver Carp Research in Lake Kinneret
2.1. Feeding Habits
A study of SC feeding habits with relation to Kinneret native fish, Sarotherodon galilaeus (SG) [11] was carried out aimed at indication if competitive consumption of Lake Kinneret (Israel) plankton exists. A lot of interaction effects were experimentally studied confirming that the effects of the two fishes were not independent and po- tentially competitors. SG suppressed most crustaceans and rotifers while increasing gross and net primary pro- duction and chlorophyll concentrations. SC had less intense effects on zooplankton than SG. Although SC sup- pressed most crustaceans and rotifers (see also [2] ), it had less interaction effects than SG. SC had no statistical- ly significant effects on phytoplankton production or chlorophyll concentration. It is suggested that these expe- riments indicate that although the impacts of SC and SG on phytoplankton production are different, both fishes utilize similar food resources in Lake Kinneret. Fingerlings of SG and SC which are planted in Lake Kinneret annually aim at the improvement of fishermen’ income and prevention of water quality deterioration. SC is known as efficient consumer of Microcystis. It is therefore recommended to introduce SC especially when Microcstis is abundant. It was found [12] that when Peridinium was abundant in Lake Kinneret, 23% - 50% of the gut content of small SC (< 3.0 kg ; n = 67) were due to Peridinium gatunenze and 46% - 70% to zooplankton (mostly Mesocyclops ogunnus and Bosmina longirostris) [13] . During February-August zooplankton and phy- toplankton comprised <50% and >50% of ingested biomass respectively and the opposite in summer (Figure 1 & Figure 2). Indices of electivity (E) [14] ) for zooplankton were negative during winter (0.01 - 0.99) and posi- tive (0.2 - 0.1) in summer and vice versa for phytoplankton: 1 - 0.3 in winter and −0.61 - 0.23 in summer. Con- dition Factor (CF) and Indices of Satiation (IOS) were high during fall, winter and spring seasons. The low val- ues of CF and IOS in summer indicated food limitation. The dominant phytoplankton component in the gut dur- ing the winter was Peridinium gatunenze and in summer Cyanobacteria. The major zooplankton biomass of gut content was due to Mesocyclops ogunnus, Ceriodaphnia reticulata and Bosmina longirostris. It was found that the biomass of the predator Cyclopoid Copepods (M. ogunnus) in gut contents was 4 times higher than that of the nano-Phytop- lankton (“greens”) consumers, Cladocerans, supporting the conclusion of beneficial SC to lake management de- sign of water quality improvement, by intensification of pressure on the “greens”. In the Illinois
Figure 1. Silver carp gut content (%) modified from Spataru & Gophen (1981).
Figure 2. Silver carp gut content (g(ww)/fish) modified from Spataru & Gophen (1981).
River ecosystem, Rotifers abundances increased after the establishment of Bighead and Silver Carp. The biomass of cladocerans and copepods declined in association with the invasion of the two carps accompanied by changes of the food web structure [2] . In the East Lake studies about 13C incorporation compared to 12C , confirmed preference of Microcystis than small “greens” by SC. Similarly SC from Kinneret, sampled during the absence of Microcystis and dominance of Peridinium during March-through early June and small “greens” during the rest of the time, indicated Δ13C in the fish muscles as −28.0‰ which is typical to the small “greens”. Consequently, when Microcystis is absent SC utilize “green” small alga efficiently which is also beneficial for water quality improvement during summer months in Lake Kinneret. Ma et al. [15] investigated the usage of SC for water quality improvement. They documented an effective removal of Microcystis by SC followed by enhancement of small “green” algae. Algal size smaller than 5 µ were not filtered by SC and algal size ranged from 5 ? 20 µ was partly ingested. Large size colonies mainly Microcystis were removed almost completely.
2.2. Age Structure, Growth Rate, Weight: Length Ratio
Previous studies [16] [17] indicated that the age structure of the commercial catch was: 78% of landings varied between 1+ to 4+ year, 16% between 5+ and 9+ year, and 6% younger than 1+. The minimal size in commercial landings was 25 cm TL aged younger than 1 year. The age was determined by scale analysis and samples were collected on deck. Growth rate was calculated by using Von-Bertalanffy equation [18] :
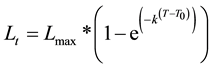
where:
Lt = Predicted Fish Length at age t;
Lmax = Maximum Length;
K = Growth factor;
T = Fish Age (Years);
T0 = Theoretical age when fish length was 0.
Growth rate of SC in Lake Kinneret is higher than in European countries: Hungary: 3.3 kg at 7 years age [19] whilst in Kinneret at the same age the weight was 20 kg . Weight of SC cultured in Germany was 1.5 - 2.0 kg during more than 6 years [20] whilst in Lake Kinneret, 1+ aged SC weighted 2.0 - 2.2 kg .
The relation between Length and Weight was calculated by using equation developed by Sprugel [21] and evaluation of actual measured individual parameters of Length and Weight resulted in as the practical W/L relations of on deck SC (Figure 3):
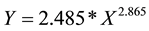 ,
,
where:
Y = Weight (kg);
X = Length (cm).
During 4 years (1988-1992) 361 tons of SC were landed and number of individual fishes was 34,895, consequently 10.3 kg per landed fish. Nevertheless, calculations of survival indicated low percentage (5% - 7%). Total number of introduced fingerlings of SC during 1971-2013 was 18.5 million and the total landing of SC at the same period was 3218 tons. Total number of landed individuals during 1972-2000 was found to be almost 200 × 103. Data of landed individual weights is not available later than 2000. Figure 4 represent the introduction and the annual landings and Figure 5 indicates the positive relation between introduction and annual catch landed three years later. The multiannual average of individual SC in catches was 10.342 Kg (SD ± 0.016 Kg ).
3. Discussion
The stocking policy of SC in Lake Kinneret was recently intensively discussed. The debate was focused on dispute between beneficial and non-beneficial topics. The benefits included introduction, fishermen income and water quality protection. Contradictions were focused on cost versus benefits. Benefits evaluation in this paper was concentrated in water quality and fishery consequences. Significant awareness was recently given to reevaluation of the SC introduction by the discovery of possibly wrong classification of the stocked fingerlings as SC but being indeed Bighead Carp or interbreed between Bighead and SC. The long term recommended SC planting was therefore disputed and intensively debated. Interim reports submitted by local scientists strongly objected SC introduction into Lake Kinneret [22] . Their comments referred to the followings: 1) SC was not a contribution factor for the improvements of water quality in Lake Kinneret; 2) If in future small size (nano- phytoplankton) algal community is enhanced and reaches a level that native components of fish and zooplankton are unable to control, introduction of SC might be reconsidered; 3) As a result of a strong diet overlap it might be possible that SC is competing with S. galilaeus and other native species on food resources; 4) If surplus food resources are available in the Kinneret ecosystem, a thorough consideration has to be done to enhance S. galilaeus planting
Figure 3. Weight/Length relation of commercial sized SC in Lake Kinneret.
Figure 4. Annual Landings (tons) and Number of Stocked Fingerlings (10^3) of SC in Lake Kinneret (1972-2013).
Figure 5. Linear regression (p and r2 are given) between introduced fingerlings and 3 years later Catch of SC (1973- 2000).
instead of SC; 5) Even if there are clear long term indications that SC is not reproducing in Lake Kinneret, the optional possibility of cross hybridization between SC and Bighead might be resulted by reproductive Hybrid in Lake Kinneret. Our studies and those carried out in East Lake, China, indicate an efficient consumption of large colonies of Microcyatis better than S. galilaeus. Our studies also confirmed zooplankton utilization by SC at a lower level than S. galilaeus. The most efficient introduction (introduction vs. catch increment) is done at a level of less than 1 million (Figure 5 & Figure 6) fingerlings a year and their impact on water quality is negligible. The 45 years record of SC introduction in Lake Kinneret confirms that SC is not reproducing in the Lake. Moreover, the length of the major (65% of the total input) inflow water route of the Jordan river is about 60 Km and the annual (1970-2013) average of monthly averages of its discharge is 14 (SD ±10) m3 per second (m3/s) (Max.-Min. range of monthly averages: 1 - 70 m3/s). In nature SC reproduce in long (> 2000 km ) rivers (Mississippi, Yangtze) with discharges of thousands m3 per second (Yangtze-30,166 m3/s). Although SC has low market value its contribution to the Kinneret fishery income is significant during the season of low catches of S. galilaeus. Conclusively, low biomass of SC in Lake Kinneret contributes two major benefits: 1) Fishermen income and 2) removal of unwanted biomass of the Cyanobacterium, Microcystis, when flourished and negligible impact on water quality deterioration when Cyanobacteria are low. It can be definitely concluded that SC stocking improve water quality in ecosystem dominated by large algal colonies. In Lake Kinneret fluctuations of Microcystis biomass is irregular and unpredictable. Therefore the presence of small (200 - 700 tons) standing stock of SC in the lake throughout all seasons might be beneficiary to fishery and to water quality protection. The Linear Prediction (Figure 6) confirmed the existence of optimal conditions in Lake Kinneret for the growth of stocked SC fingerlings as well as reasonable recruitment of planted Young of the Year (YOY) SC to the commercial fishery. Fractional Polynomial plot of stocked fingerlings vs. Commercial landings indicates that the optimal number of introduced specimen is more than 500,000 and below 1000,000 (Figure 7).
4. Summary
The planting of an exotic fish like SC in Lake Kinneret indicated a reasonable implementation of the proposed objectives without predicted risks. The SC preferably consumed phytoplankton during 8 months (1 - 8) by selection of large cells Peridinium or big size colonies of Microcystis (Figure 1 & Figure 2; [3] [9] ). The Index of Satiation and Body Condition Factor (Figure 8) were only at low level in summer indicating food limitation during this season and high in winter and fall when the Peridinium or Microcystis were alternately dominant. SC is an exotic species in Lake Kinneret but its growth rate is higher (Figure 3) than those observed in temperate zone countries [19] [20] . The major contribution of SC to commercial landings is given at the fingerlings age of 3+ - 4+ (Figure 4) with maximal stocking of 1.8 × 106 fingerling in 1977 and 187 tons in 1980. The positive relation between introduced fingerlings and annual landing 3 years later was statistically confirmed (p = 0.0005 and r2 = 0.295) (Figure 5). Data on the weight/length relation during 2001-2000 indicated a high rate of increment to marketable size. SC did not present an indication of reproduction in Lake Kinneret and the low (but not absent) competition with native species. SC contributed to the improvement of water quality in Lake Kinneret by Microcystis or Peridinium removal when they are dominant. After 45 years of SC introduction in Lake Kinneret, no indications of water quality deterioration attributed to SC were confirmed. If Microcystis remains dominant
Figure 6. Linear Prediction between 10^3 stocked fingerlings (independent value) and annual landings (tons) (fitted value) of SC in Lake Kinneret (1972-2023).
Figure 7. Fractional polynomial relation between number of stocked fingerlings (10^3) (SC) and Predicted annual landing (tons) In lake Kinneret (1972-2013).
Figure 8. SC: Indices of Satiation (IOS) and Condition Factor (CF) In four seasons: 1-Winter; 2-Spring; 3-Summer; 4-Fall. Modified from Spataru & Gophen (1981).
within the algal community of Lake Kinneret, the beneficiary of SC is obvious. If Peridinium reappears and becomes dominant, SC might also contribute to the decline of algal biomass beside being a factor of fishery enhancement.
Cite this paper
MosheGophen,GregorySnovsky, (2015) Silver Carp (Hypophthalmichthys molitrix, Val. 1844) Stocking in Lake Kinneret (Israel). Open Journal of Ecology,05,343-351. doi: 10.4236/oje.2015.58028
References
- 1. USFWS (2011) Final Rule Federal Register Asian Carp Prevention and Control Act (S. 1421), Signed into Law by President Obama on Dec 14, 2010. USA Fish and Wildlife Service, Washington DC.
- 2. Sass, G.G., Hinz, C., Erickson, A.C., McClelland, N.N., McClelland, M.A. and Epifanlo, J.M. (2014) Invasive Bighead and Silver Carp Effects on Zooplankton Communities in the Illinois River, Illinois, USA. Journal of Great Lakes Research, 40, 911-921. http://dx.doi.org/10.1016/j.jglr.2014.08.010
- 3. Miura, T., Ed. (1989) East Lake: A Phytoplanktivorous Fishes Dominated Lake Ecosystem. Contribution of the Otsu Hydrobiological Station Kyoto University, No. 334 (Foreign Language Ser.), 142 p.
- 4. Tatsukawa, K., Zhu, Z. and Miura T. (1989) Chapter: Acoustic Estimation of Fish Population, In: Miura, T., Ed., East Lake, Otsu Hydrobiological Station, Kyoto University, Kyoto, 13-30.
- 5. Miura, T., Zhu, Z. and Tatsukawa, K. (1989) Growth of Silver Carp and Bighead Carp. In: Miura, T., Ed., East Lake, Otsu Hydrobiological Station, Kyoto University, Kyoto, 31-38.
- 6. Chen, L.S. (1982) Analysis of Food Composition in the Silver and Bighead Carps Stocked in East Lake (China). Reservoir Fishery Report, 3, 21-26 (in Chinese, English Summary).
- 7. Miura, T., Iwata, K. and Zhu, Z. (1989) Production and Food Consumption of the Silver and Bighead Carp Populations. In: Miura, T., Ed., East Lake, Otsu Hydrobiological Station, Kyoto University, Kyoto, 106-114.
- 8. Miura, T. and Chen, S. (1989) Nitrogen Flow in a Subsystem Formed by Planktivorous Fishes, Phytoplankton and Zooplankton In: Miura, T., Ed., East Lake, Otsu Hydrobiological Station, Kyoto University, Kyoto, 115-126.
- 9. Opuszynski, K. (1980) The Role of Fishery Management in Counteracting Rutrophication Processes. Hydrobiologia, 2, 263-269.
- 10. Iwata, K., Chen, S. and Liu, X. (1989) Utilization of Blue-Green Algae (Microcystis aeruginosa) by the Cyprinid Phytoplankton Feeders, Hypophthalmichthys molitrix and Aristichthys nobilis. In: Miura, T., Ed., East Lake, Otsu Hydrobiological Station, Kyoto University, Kyoto, 81-103.
- 11. Gophen, M. (2014) Competitive Consumption of the Lake Kinneret (Israel) Plankton by Hypophthalmichthys molitrix and Sarotherodon galilaeus. Open Journal of Ecology, 4, 532-542.
http://dx.doi.org/10.4236/oje.2014.49043 - 12. Shapiro, J. (1985) Food and Intestinal Content of the Silver Carp Hypophthalmichthys molitrix (Val), in Lake Kinneret during 1982-1984. Bamidgeh, 37, 3-18.
- 13. Spataru, P. and Gophen, M. (1981) Feeding Behaviour of Silver Carp Hypophthalmichthys molitrix Val. and Its Impact on the Food Web in Lake Kinneret, Israel. Hydrobiologia, 120, 53-61.
- 14. Ivlev, V.S. (1961) Experimental Ecology of the Feeding of Fish. Yale University Press, New York, 302.
- 15. Ma, H., Cui, F., Liu, Z., Fan, Z., He, W. and Yin, P. (2010) Effect of Filter Feeding Fish Silver Carp on Phytoplankton Specoies and Size Distribution in Surface Water: A Field Study in Water Works. Journal of Environmental Sciences, 22, 161-167. http://dx.doi.org/10.1016/S1001-0742(09)60088-7
- 16. Sanovsky, Z. (2000) Silver Carp in Lake Kinneret during 1989-1991: Biological and Fishery Perspectives. Fishery and Aquaculture, 3, 108-117.
- 17. Sanovsky, Z. and Pizanty, S. (2002) Silver Carp in Lake Kinneret-Fishery and Introduction Efficiency. Fishery and Aquaculture, 33, 349-355.
- 18. Bertalanffy, V. (1983) A Quantitative Theory of Organic Growth. Human Biology, 10, 181-213.
- 19. Schmidt, K. and Ziebarth, G. (1984) Ungarische Erfahrungen beider Reproduction von Silbercarpfen, Hypophthalmichthis molitrix (Val.). Binnenfischerei DDR, 31, 49-53.
- 20. Nagel, L. (1988) Erzeugung von Speisefischen der Fishcharten pflanzenfressender Fische in Teichen. Z. Binnenfischerei DDR, 35, 256-257.
- 21. Sprugel, D.B. (1983) Correcting for Bias in Long-Transformed Allometric Equations. Ecology, 64, 209-210. http://dx.doi.org/10.2307/1937343
- 22. Goren, M. (2014) Silver Carp Introduction into Lake Kinneret. Background Document Submitted to the Israeli Water Authority and the Fishery Department of the Israeli Ministry of Agriculture and Agricultural Settlements Development. 2 p (in Hebrew).









