Open Journal of Pediatrics
Vol.4 No.1(2014), Article ID:43496,11 pages DOI:10.4236/ojped.2014.41001
Retrospective Surveillance over 11 Years for Intussusception in Children Younger than 14 Years in the State of Qatar
Tariq O. Abbas, Noora AlShahwani, Gaby Jabbour, Mansour Ali
Department of Pediatric Surgery, Hamad General Hospital, Doha, Qatar
Email: tariq2c@hotmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 1 December 2013; revised 2 January 2014; accepted 10 January 2014
ABSTRACT
Background: Accurate estimates of the baseline incidence of childhood intussusception could help safety monitoring after introduction of rotavirus vaccines. Therefore, we studied the incidence of intussusception in the state of Qatar. Methods: We retrospectively reviewed cases of intussusception in children younger than 14 years. Children were treated between 2002 and 2012 at a single hospital in Qatar. We analyzed information according to age, sex, clinical signs, diagnosticand treatment-related characteristics, and length of stay. We calculated the national incidence of intussusception using census data. Results: A total of 106 children were diagnosed and treated for intussusception over 11 years. The male to female ratio of intussusception was 1.4:1. The most common symptom was bleeding per rectum (37%). One hundred and four children with intussusception underwent diagnostic and therapeutic contrast enema, and 31.7% of patients underwent operative management. The length of stay was longer in patients who underwent operative reduction than that in those who did not. Conclusions: This is the first study to estimate the incidence of childhood intussusception prior to introduction of rotavirus vaccination in Qatar. A prospective surveillance system using a standardized case definition would be useful for examining the occurrence of intussusception in the Arabic Gulf area and developing countries.
Keywords:Children; Intussusception; Surgical Reduction; Therapeutic Contrast Enema

1. Background
Intussusception which is invagination of part of the bowel into the next part like a telescope is a common childhood problem that results in serious morbidity and mortality worldwide. In developing nations, the mortality may be as high as 20% [1] . Most intussusceptions are considered to be idiopathic in origin because there is usually no identifiable pathology at the lead point. The underlying cause of intussusception in most infants is unknown, but it is associated with several pathogens, including adenoviruses [2] -[11] . Although the full etiology of intussusceptions remains unclear, adenovirus infection is associated with an increased risk of intussusception. In only approximately 6% of cases (ranging from 1.5% to 12%), intussusception is associated with a pathological lead point, predominantly Meckel’s diverticulum [12] . The vast majority of intussusception episodes, termed “idiopathic”, arise in the ileum because of lymphoid hyperplasia of Peyer’s patches [13] , suggestive of a response to infection. However, the epidemiological features of intussusception recurrence and surgical reduction are not well understood [14] .
In approximately 50% of children with intussusception, an infective etiology is further suggested by the presence of viral shedding in the stools, together with the demonstration of virus particles in pathological specimens [15] . However, natural rotavirus infection is not believed to be linked to this condition, but an association of intussusceptions with an oral rhesus-based tetravalent rotavirus vaccine has been well documented [1] . This vaccine was licensed in the United States in 1998 and administered to approximately 500,000 infants. The subsequent risk of intussusceptions was estimated to be one case per 10,000 vaccine recipients [1] . In 1999, Rotashield (the first-generation rotavirus vaccine licensed in the United States) was withdrawn from the market because of a potential increased risk of intussusception [16] .
In 2004, a new rotavirus vaccine (Rotarix®) was licensed in Mexico. In 2006, another vaccine (Rotateq®, Merck) was approved in the United States. At present, both vaccines are used in routine immunization in several high and low-income countries. Clinical trials of these vaccines and early post-licensure data have shown no major risk of intussusception after vaccination (<1 case in 20,000 vaccines). However, a risk of a lower magnitude cannot be ruled out entirely before further post-licensure data have been accumulated. For this reason, local data on the baseline incidence and epidemiology of intussusception are important for countries that are considering using rotavirus vaccines to help with post introduction safety monitoring [1] .
A recent and extensive review by the World Health Organization (WHO) on intussusception concluded that, in developed countries, the baseline incidence of intussusception is between 0.5 and 4.3 cases per 1000 live births or 0.7 - 1.2 cases per 1000 children aged less than 1 year [17] . Accurate estimates of the incidence of intussusception are not available for most developing countries [18] . Although intussusception remains the most common cause of acute intestinal obstruction in infants and young children, the condition is linked as a side effect to a previous vaccine administered to infants in the United States in 1998 for the prevention of rotavirus gastroenteritis. This resulted in a global need to obtain incidence estimates of intussusception before the introduction of new rotavirus vaccines [19] .
In developed countries, a favorable clinical outcome of intussusception is more likely because of timely diagnosis and early treatment by the less invasive procedure of enema reduction. By contrast, in developing countries, delayed diagnosis with ensuing bowel necrosis, followed by attempted operative reduction, is associated with high case fatality rates (e.g., 18% in Nigeria, 20% in Indonesia, and up to 54% in Ethiopia [20] .
The treatment of intussusception has primarily evolved from operative management to the preference for radiological reduction with either air or barium contrast. Radiological reductions of intussusception have been shown to decrease the length of hospitalization, shorten recovery, and reduce the risk of complications associated with major abdominal surgery. The published success rate of radiological reduction averages 80%, but varies widely [21] .
Mortality caused by intussusception in infants and children is now uncommon in developed countries because of better access to healthcare facilities. In contrast, intussusception-associated mortality remains high in some developing countries [15] . Children treated at hospitals with >10,000 pediatric admissions yearly have higher rates of non-operative reduction than children treated at other non-tertiary facilities [22] .
Qatar is a sovereign Arab state, located in Western Asia, occupying the small Qatar Peninsula on the northeasterly coast of the much larger Arabian Peninsula [23] . Out of the total population of approximately 1.5 million (estimated in May 2008), the make-up of ethnic groups is as follows: 20% Qatari (Arab), 20% other Arab, 20% Indian, 10% Filipino, 13% Nepali, 7% Pakistani, 5% Sri Lankan, and 5% others (United States State Department Qatar Page).
Hamad Medical Corporation (HMC), which is affiliated with Cornell University, is the premier non-profit health care provider in Doha, Qatar. The HMC manages five highly specialized hospitals, the biggest of which is Hamad General Hospital.
The incidence of intussusception in the Qatar population is unknown. Therefore, we aimed to determine the prevalence of intussusception and associated epidemiological and environmental risk factors in children younger than 14 years in the State of Qatar. Moreover, we aimed to provide estimates of hospitalization rates for intussusception during 2002-2012. This included patients’ demographics, seasonality, management, and outcomes. We attempted to delineate current outcomes for the treatment of intussusception in children in Qatar. We also examined whether improvement in enema reduction rates has occurred over time and the effect of current reduction techniques and delay in presentation of symptoms on rates of bowel resection, postoperative complications, and length of stay.
2. Methods
This retrospective hospital-based study to review cases of intussusceptions was carried out at Hamad General Hospital (HGH), Doha, Qatar. HGH represents the sole healthcare institution in the state of Qatar. It is 621-bed multispecialty, tertiary-care teaching hospital, which serves a vital, diverse, and multi-national community. The protocol received appropriate institutional ethical review.
All medical records of hospitalized children aged less than 14 years at HGH were reviewed for the period from January 2002 through to December 2012, to identify patients who met the clinical case definition of intussusception as defined by the Brighton Collaboration. Records with intussusceptions were extracted using the International Classification of Diseases, Version 9 (ICD-9) diagnosis code 560.0 from discharge records in the hospital’s medical record database of 2002-2012.
For each identified child, we extracted information on demographics, admission, and discharge dates, clinical signs and symptoms and their duration, and diagnostic and treatment procedures performed. Data on the standardized questionnaire were summarized and entered into an electronic database and checked for accuracy. Data extraction and entry were performed by the same investigator. We analyzed the information according to age, sex, clinical signs, date of hospitalization, and diagnosticand treatment-related characteristics. Because early diagnosis and treatment could provide better outcomes, we compared the length of hospital stay for patients admitted for less than 24 hours and 24 hours or longer after the onset of symptoms. Incidence rates were calculated by using census data.
Retrospective analysis was performed on the medical records of the patients managed with intussusception in the department of pediatric surgery of HGH for 11 years within the previous 10 years (from Jan 2002 to Dec 2012). The database includes data on diagnoses, comorbidities at admission coded, surgical procedures, length of stay, and discharge status, including in-hospital death.
For study inclusion, the diagnosis of intussusception must have been verified by either imaging (ultrasound, enema, or computed tomography) or surgical exploration. Patients were excluded from the study if records could not be validated for the diagnosis of intussusception.
All radiological reduction techniques were performed under fluoroscopy using liquids and numbers of attempts were at the discretion of the radiologist. A rectal catheter was inserted and the buttocks were taped to prevent contrast leakage. Liquid reduction was attempted using either barium or water-soluble diatrizoate meglumine (gastrograffin) prepared with routine methods. A column of contrast was established 1 m above the table. Reduction attempts were followed using real-time fluoroscopic guidance with the patient in the prone position, as well as in the supine and oblige positions. Typically, three consecutive attempts of 3 minutes each were performed. Conscious sedation was not used in the non-operative radiological-guided attempts at intussusception reduction.
Statistical Analysis
Descriptive statistics were used to summarize the demographic characteristics of patients. Means +− standard deviation are reported were appropriate. Ratios with 95% confidence intervals were calculated by using Poisson regression data.
3. Results
3.1. Population
One hundred and six children were diagnosed and treated for intussusception over an 11-year period. A total of 23% of children were less than 6 months old, 56% were 6 months to 1 year old, and 26% were older than 1 year old at the time of diagnosis (Figure 1, Table 1). A total of 35% were Qatari and 65% were non-Qatari (Figure 2, Table 2).
3.2. Signs and Symptoms
The most common presenting sign or symptom in our series was bleeding per rectum, occurring in 37% of children. Fifty-three patients presented with the onset of symptoms within 24 h, 47 presented after 1 day of onset, ranging from 1 to 9 days (%), and six were unspecified (Figures 3, 4).
In 106 patients, symptoms on presentation included abdominal pain in 33%, abdominal distension in 2%, vomiting 57%, specifically bilious vomiting in 35%, bleeding per rectum in 37%, diarrhea in 3%, and fever in 5% (Table 2).
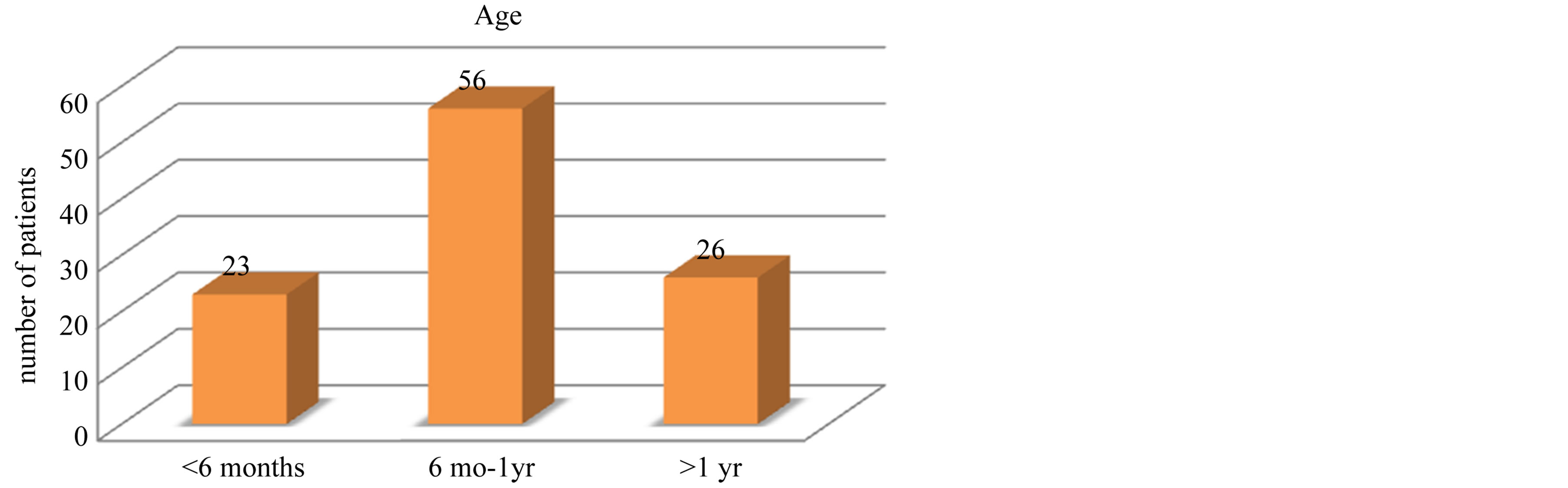
Figure 1. Age distribution of patients with intussusception over the study period.
non-Qatari

Figure 2. Nationality distribution of non-Qatari patients.
Table 1. Proportions of different symptoms at presentation.
Table 2. Gender distribution and average of ages of patients with intussusception over the study period.

Figure 3. Symptoms and signs at presentation.
Duration of Symptoms
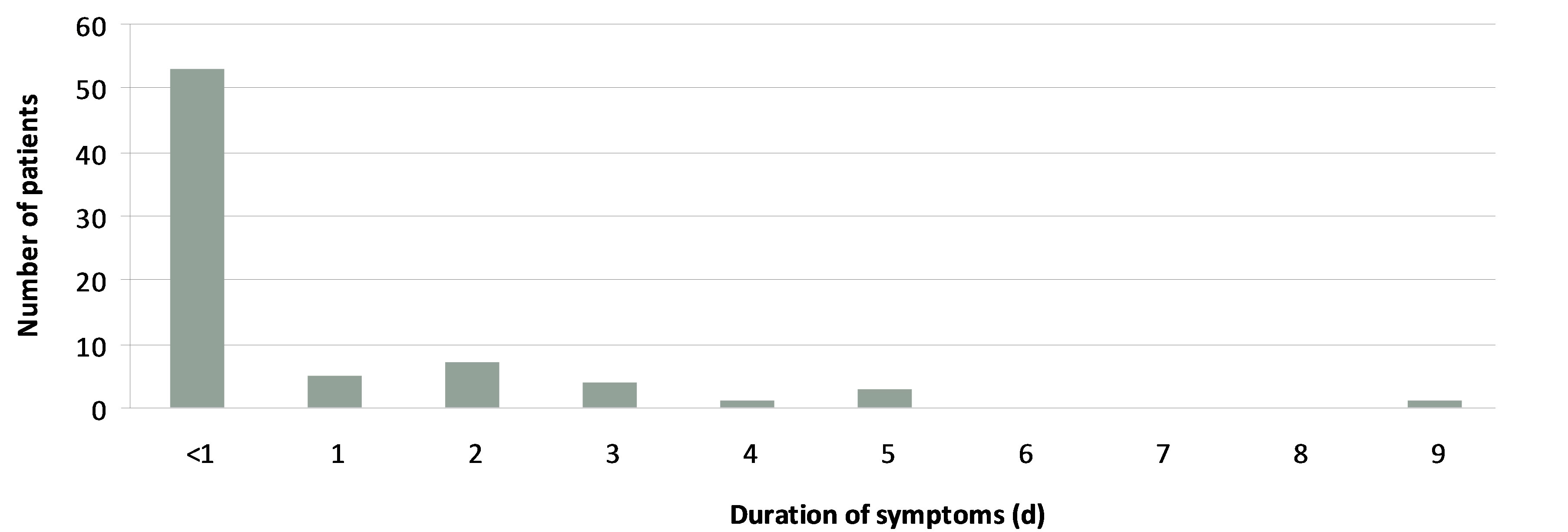
Figure 4. Diagram showing the duration of symptoms in different patients.
3.3. Enema Reduction
One hundred four children with intussusceptions underwent diagnostic and therapeutic contrast enema. Seventyone (68.3%) of them were successful and 33 (31.7%) failed. The enema reduction failure rate was higher in patients who had symptoms for less than 24 hours (22/53 patients, 41.5%) compared with those who had symptoms for more than 24 h (10/45 patients, 22.2%) (Table 3).
3.4. Operative vs Nonoperative Reduction
A total of 31.7% of patients underwent operative management. Operative intervention was higher in patients who had symptoms for less than 24 h compared with those who had symptoms for more than 24 h (41.5% vs 22.2%) (Table 3).
3.5. Coexisting Conditions
Preceding illnesses and associated congenital anomalies were variable (Table 4).
3.6. Length of Stay
The length of hospital stay ranged between 1 and 7 days (mean, 2.6 days). The length of stay was longer in patients who underwent operative reduction than in those who did not undergo operative reduction in both groups of presentation (symptoms for less than and more than 24 h).
3.7. Seasonality
Intussusception cases were coming through all the year but clear double-peak pattern during April and November. It was noticed also that the average ages of the patients were lowest during these peaks (10 - 11 months) which might be an indicator that idiopathic intussucpetion is seasonal in nature following changes in temperature or vaccination campaigns (Figure 5).
Table 3. Different methods of intervention per duration of symptoms.
Table 4. Preceding illnesses and associated congenital anomalies.
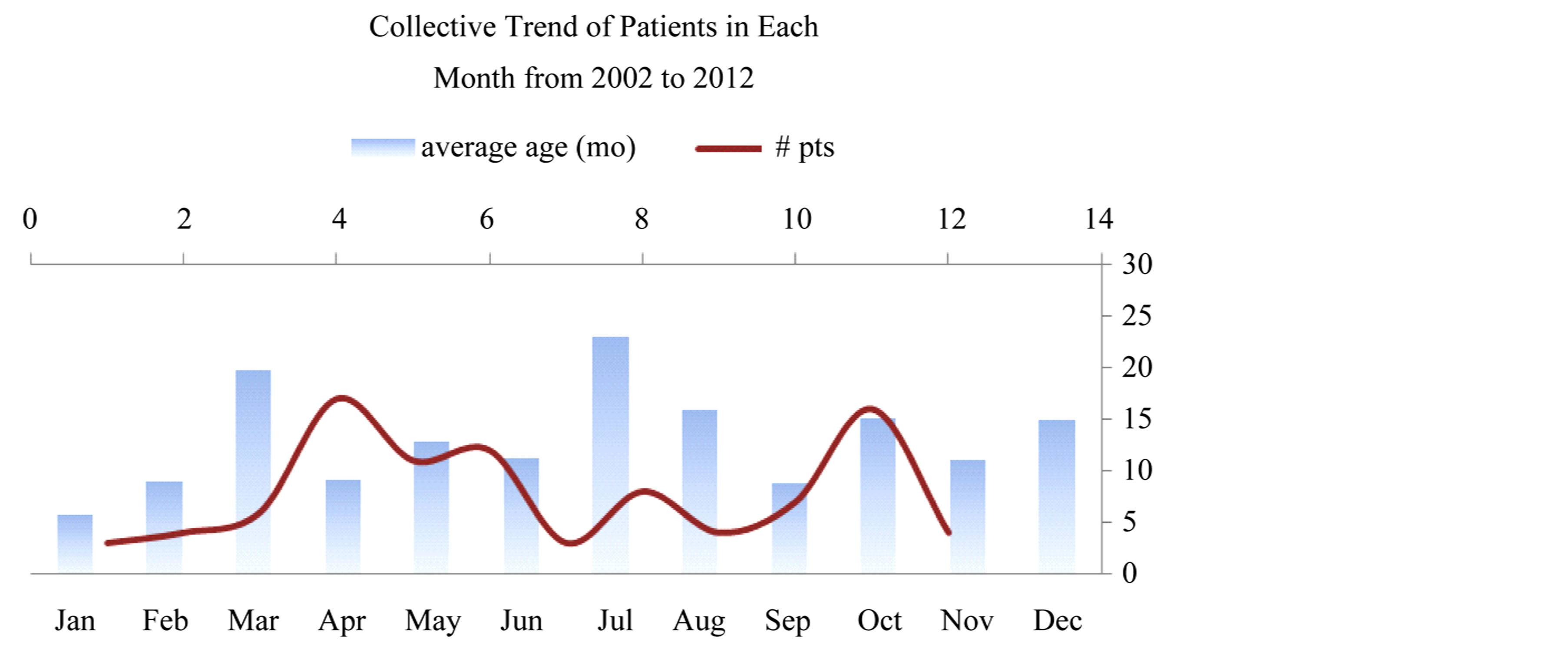
Figure 5. Seasonality pattern in patients with intussusception over the study period.
4. Discussion
Our study examined the nature of childhood intussusception in Qatar. To the best of our knowledge, this is the first study to investigate the incidence of intussusception among the Qatar population using a nationwide database combined with the advantage of a single tertiary hospital being the referral center for all suspected patients having the disease.
4.1. Rotavirus
Considering the global introduction of the rotavirus vaccine, the risk of intussusception attributable to rotavirus vaccine needs to be addressed by accurate data on the background rate of intussusception. The number of intussusceptions that occur by chance alone will increase in the week after administration of rotavirus vaccine, while the incidence in intussusception is larger in the month after the vaccine is given in countries where intussusception rates are high, or if the coexistence between both is valid.
4.2. Imaging
Although abdominal X-ray has a low sensitivity (29% - 50%), a significant number of children in the present study were exposed to unnecessary radiation for little likely clinical benefit. However, abdominal ultrasonography has high sensitivity (98%). Abdominal ultrasonography should be the first-line investigation in children with suspected intussusception [24] . Little is known about the effect of subspecialty training and hospital volume on outcomes for children with intussusception [25] . A recent study using discharge data from the state of Washington suggested that low-volume hospitals, with less than 10,000 pediatric admissions per year, performed more operative reductions and bowel resections for children with intussusception than those cared for in a high-volume specialty setting [26] .
4.3. Success of Enema Reduction
Nonoperative reduction of intussusception is performed by a radiologist using hydrostatic or pneumatic pressure under fluoroscopic or sonographic visualization, and is successful in most cases. Nonoperative reduction of intussusception has been shown to reduce the length of stay, shorten recovery, decrease hospital costs, and decrease the risk of complications related to abdominal surgery [27] . The success rate of enema reduction varies widely in the published literature. Reported rates of successful enema reduction average 75% [28] [29] .
4.4. Surgical Intervention
The need for operative reduction is important for a number of reasons. First, surgical management is associated with a longer length of stay, especially when bowel resection is performed. This results in higher hospital costs, as well as inefficient use of health care resources. In addition, the need for laparotomy affects morbidity, painand the long-term risk of adhesive bowel obstruction. Finally, a delay in the diagnosis of intussusception in rare cases can lead to bowel necrosis and overwhelming sepsis, which is a potentially lethal complication [25] . In one study, surgical reduction was still required in 6% to 53% of children, including patients with failed enema reduction, with a pathological lead point, or with contraindications to attempted enema reduction [30] -[34] .
4.5. Epidemiology
The epidemiology of intussusception in Qatar was similar to that described in other parts of the world. Previous reports have shown that intussusception is more frequent in males, with our study yielding a male to female ratio of 1.4:1. The ratio is reported to be vary widely across different regions of the world, but all reports have indicated a predominance of males. For example, this ratio was reported as 1.3:1 in Singapore [35] and 9:1 in India [36] [37] . A predominance of intussusception among males has been noted with male-to-female ratios ranging from 1.3 to 2.0 [38] -[41] . An interesting finding in a study from Taiwan was that the incidence rate ratio between two sexes was dynamic, increasing from 1.31 in children less than 1 year old to 2.52 in children 9 years old [14] . The gradual increase in male-to-female incidence rate ratio by age may be related to the differential onset time between the two sexes, because this previous study found that the mean age of the initial onset of intussusception was significantly older in males than in females (27.3 months vs. 24.9 months, p < 0.001). The male-to-female incidence rate ratio decreased after 9 years of age, but the limited number of cases did not permit any strong inference among older children or adolescents [14] .
Most cases of intussusception occur in infants between 3 - 6 months, which is the age when rotavirus vaccination is administered. This may challenge surveillance of intussusceptions following vaccination because a possible increase in cases due to vaccination may coincide with its natural peak. In Qatar, distinct seasonality was detected, which is different to reports from countries such as the United States [42] [43] and Europe [44] [45] . Seasonality of the occurrence of intussusceptions is controversial. Some studies have reported no seasonal variation [39] [46] -[50] , while other studies have favored seasonality [47] [51] .
Although intussusception is a common surgical condition in children, there are no current guidelines in the state of Qatar regarding treatment of children with this condition. However, over the last 2 years, there has been some movement toward standardization of the referral system of children having this condition in the form of sending patients suspected from the Pediatric Emergency Centre from different regions of the state to perform ultrasound. At the same time, the on call pediatric surgeon is contacted to evaluate the patient, admit him/her in case of confirmation by ultrasound, and arrangement of hydrostatic reduction should be performed as soon as possible by the same radiologist who performed the ultrasound. Because it is impractical to refer all children with abdominal pain and vomiting to the radiology department of the main hospital and initiate this protocol, the results of this study should stimulate efforts toward improved education about the diagnosis and management of intussusception for those working in the community.
4.6. Limitations
The retrospective nature of this study resulted in some limitations. First, there was a lack of complete immunization data, which made it difficult to reliably count the number of immunizations given prior to hospitalization for intussusception. However, this was not a main objective of the study. Second, it was difficult to obtain accurate birth-cohort data for the catchment population. Nevertheless, the strength of this retrospective study is that it analyzed data from the only tertiary care facility in the country with complete investigations and reliable documentation. The specificity of case ascertainment is therefore likely to be high, and the description of the clinical presentation, treatment outcomes, and epidemiology is consequently an accurate representation of intussusception in Qatar.
5. Conclusion
In conclusion, we report our 11-year experience of intussusception in Qatar, which provides accurate baseline data. We found an increasing overall incidence in children less than 14 years, with a clear upward trend between 2002 and 2011, and an absence of seasonality in Qatar.
6. Conflict of Interests
All of the authors do not have a direct financial relation with the commercial identities mentioned in the paper that might lead to a conflict of interests.
7. Authors Contributions
TA carried out the bulk of proposal writing, data analysis and manuscript building. NA assisted in data collection and analysis. GJ participated in data collection. MA participated in manuscript structuring.
Acknowledgements
The authors thank Medical Research Centre at Hamad Medical Corporation for funding this study.
References
- Blakelock, R.V. and Beasley, S.W. (1998) The Clinical Implications of Non-Idiopathic Intussusception. Pediatric Surgery International, 14, 163-167. http://dx.doi.org/10.1007/s003830050475
- Ross, J.G., Potter, C.W. and Zachary, R.B. (1962) Adenovirus Infection in Association with Intussusception in Infancy. Lancet, 2, 221-223. http://dx.doi.org/10.1016/S0140-6736(62)92315-2
- Bell, T.M. and Steyn, J.H. (1962) Viruses in Lymph Nodes of Children with Mesenteric Adenitis and Intussusception. British Medical Journal, 2, 700-702. http://dx.doi.org/10.1136/bmj.2.5306.700
- Gardner, P.S., Knox, E.G., Court, S.D., et al. (1962) Virus Infection and Intussusception in Childhood. British Medical Journal, 2, 697-700. http://dx.doi.org/10.1136/bmj.2.5306.697
- Clarke, E.J. Jr., Phillips, I.A. and Alexander, E.R. (1969) Adenovirus Infection in Intussusception in Children in Taiwan. JAMA, 208, 1671-1674. http://dx.doi.org/10.1001/jama.1969.03160090031007
- Nicolas, J.C., Ingrand, D., Fortier, B., et al. (1982) A One-Year Virological Survey of Acute Intussusception in Childhood. Journal of Medical Virology, 9, 267-271. http://dx.doi.org/10.1002/jmv.1890090404
- Montgomery, E.A. and Popek, E.J. (1994) Intussusception, Adenovirus, and Children: A Brief Reaffirmation. Human Pathology, 25, 169-174. http://dx.doi.org/10.1016/0046-8177(94)90274-7
- Hsu, H.Y., Kao, C.L., Huang, L.M., et al. (1998) Viral Etiology of Intussusception in Taiwanese Childhood. The Pediatric Infectious Disease Journal, 17, 893-898. http://dx.doi.org/10.1097/00006454-199810000-00009
- Bode, C.O. and Omilabu, S.A. (2002) Viral Isolates of Intussusception in Nigerian Infants. South African Journal of Surgery, 40, 57-58.
- Guarner, J., de Leon-Bojorge, B., Lopez-Corella, E., et al. (2003) Intestinal Intussusception Associated with Adenovirus Infection in Mexican Children. American Journal of Clinical, 120, 845-850. http://dx.doi.org/10.1309/LBRNGF9MJW2MHT97
- Selvaraj, G., Kirkwood, C., Bines, J., et al. (2006) Molecular Epidemiology of Adenovirus Isolates from Patients Diagnosed with Intussusception in Melbourne, Australia. Journal of Clinical Microbiology, 44, 3371-3373. http://dx.doi.org/10.1128/JCM.01289-06
- Daneman, A. and Navarro, O. (2003) Intussusception Part 1: A Review of Diagnostic Approaches. Pediatric Radiology, 33, 79-85.
- Robinson, C.G., Hernanz-Schulman, M., Zhu, Y., et al. (2004) Evaluation of Anatomic Changes in Young Children with Natural Rotavirus Infection: Is Intussusception Biologically Plausible? The Journal of Infectious Diseases, 189, 1382-1387. http://dx.doi.org/10.1086/382655
- Parasher, V.D., Holman, R.C. and Cummings, K.C. (2000) Trends in Intussusceptions Associated Hospitalizations and Deaths among US Infants. Pediatrics, 106, 1413-1421. http://dx.doi.org/10.1542/peds.106.6.1413
- Bhowmick, K., Kang, G., Bose, A., et al. (2009) Retrospective Surveillance for Intussusception in Children Aged Less Than Five Years in a South Indian Tertiary-Care Hospital. Journal of Health, Population and Nutrition, 27, 660-665.
- Murphy, T.V., Gargiullo, P.M., Massoudi, M.S., et al. (2001) Intussusception among Infants Given an Oral Rotavirus Vaccine. The New England Journal of Medicine, 344, 564-572. http://dx.doi.org/10.1056/NEJM200102223440804
- Nakagomi, T., Takahashi, Y., Arisawa, K., et al. (2006) A High Incidence of Intussusception in Japan as Studied in a Sentinel Hospital over a 25-Year Period (1978-2002). Epidemiology & Infection, 134, 57-61. http://dx.doi.org/10.1017/S0950268805004644
- WHO (2002) Vaccines and Biological Acute Intussusceptions in Infants and Children. Incidence, Clinical Presentation and Management: A Global Perspective. World Health Organization, Geneva, 1-98.
- Samad, L., Marven, S., El Bashir, H., et al. (2012) Prospective Surveillance Study of the Management of Intussusception in UK and Irish Infants. British Journal of Surgery, 99, 411-415. http://dx.doi.org/10.1002/bjs.7821
- Boudville, I.C., Phua, K.B., Quak, S.H., et al. (2006) The Epidemiology of Paediatric Intussusception in Singapore: 1997 to 2004. ANNALS Academy of Medicine Singapore, 35, 674-679.
- Jen, H.C. and Shew, S.B. (2009) The Impact of Hospital Type and Experience on the Operative Utilization in Pediatric Intussusception: A Nationwide Study. Journal of Pediatric Surgery, 44, 241-246. http://dx.doi.org/10.1016/j.jpedsurg.2008.10.050
- Bratton, S.L., Haberkern, C.M., Waldhausen, J.H.T., et al. (2001) Intussusception: Hospital Size and Risk of Surgery. Pediatrics, 107, 299-303. http://dx.doi.org/10.1542/peds.107.2.299
- Carneiro, P.M. and Kisusi, D.M. (2004) Intussusception in Children Seen at Muhimbili National Hospital, Dar es Salaam. East African Medical Journal, 81, 439-442. http://dx.doi.org/10.4314/eamj.v81i9.9217
- Yoon, C.H., Kim, H.J. and Goo, H.W. (2001) Intussusception in Children: US-Guided Pneumatic Reduction—Initial Experience. Radiology, 218, 85-88. http://dx.doi.org/10.1148/radiology.218.1.r01ja1085
- Tate, J.E., Simonsen, L., Viboud, C., et al. (2008) Trends in Intussusception Hospitalizations among US Infants, 1993-2004: Implications for Monitoring the Safety of the New Rotavirus Vaccination Program. Pediatrics, 121, e1125.
- Somme, S., To, T. and Langer, J.C. (2006) Factors Determining the Need for Operative Reduction in Children with Intussusception: A Population-Based Study. Journal of Pediatric Surgery, 41, 1014-1019. http://dx.doi.org/10.1016/j.jpedsurg.2005.12.047
- Le Masne, A., Lortat-Jacob, S. and Sayegh, N. (1999) Intussusception in Infants and Children: Feasibility of Ambulatory Management. European Journal of Pediatrics, 158, 707-710. http://dx.doi.org/10.1007/s004310051184
- Daneman, A. and Navarro, O. (2004) Intussusception Part 2: An Update on the Evolution of Management. Pediatric Radiology, 34, 97-108. http://dx.doi.org/10.1007/s00247-003-1082-7
- Applegate, K.E. (2006) Intussusception in Children: Diagnostic Imaging and Treatment, In: Blackmore, C.C. and Medina, S., Eds., Evidence-Based Imaging, Springer, New York, 475-492. http://dx.doi.org/10.1007/0-387-31216-1_26
- Lai, A.H.M., Phua, K.B., Teo, E.L.H.J., et al. (2002) Intussusception: A Three-Year Review. ANNALS Academy of Medicine Singapore, 31, 81-85.
- Bratton, S.L., Haberkern, C.M., Waldhausen, J.H.T., et al. (2001) Intussusception: Hospital Size and Risk of Surgery. Pediatrics, 107, 299-303. http://dx.doi.org/10.1542/peds.107.2.299
- Okuyama, H., Nakai, H. and Okada, A. (1999) Is Barium Enema Reduction Safe and Effective in Patients with a Long Duration of Intussusception? Pediatric Surgery International, 15, 105-107. http://dx.doi.org/10.1007/s003830050526
- Hadidi, A.T. and Shal, N.E (1999) Childhood Intussusception: A Comparative Study of Nonsurgical Management. Journal of Pediatric Surgery, 34, 304-307. http://dx.doi.org/10.1016/S0022-3468(99)90196-3
- Guo, J.Z., Ma, X.Y. and Zhu, Q.H. (1986) Results of Air Pressure Enema Reduction of Intussusception: 6,396 Cases in 13 Years. Journal of Pediatric Surgery, 21, 1201-1203. http://dx.doi.org/10.1016/0022-3468(86)90040-0
- Samad, L., Marven, S., El Bashir, H., et al. (2012) Prospective Surveillance Study of the Management of Intussusception in UK and Irish Infants. British Journal of Surgery, 99, 411-415. http://dx.doi.org/10.1002/bjs.7821
- Boudville, I.C., Phua, K.B., Quak, S.H., et al. (2006) The Epidemiology of Paediatric Intussusception in Singapore: 1997 to 2004. Annals of the Academy of Medicine, Singapore, 35, 674-679.
- Gopi, V.K., Joseph, T.P. and Varma, K.K. (1989) Acute Intestinal Obstruction. Indian Pediatrics, 26, 525-530.
- Chen, S.C., Wang, J.D., Hsu, H.Y., et al. (2010) Epidemiology of Childhood Intussusception and Determinants of Recurrence and Operation: Analysis of National Health Insurance Data between 1998 and 2007 in Taiwan. Pediatrics & Neonatology, 51, 285-291. http://dx.doi.org/10.1016/S1875-9572(10)60055-1
- Archibong, A.E., Usoro, I.N., Ikpi, E., et al. (2001) Paediatric Intussusceptions in Calabar, Nigeria. East African Medical Journal, 78, 19-21. http://dx.doi.org/10.4314/eamj.v78i1.9106
- Boudville, I.C., Phua, K.B., Quak, S.H., et al. (2006) The Epidemiology of Paediatric Intussusception in Singapore: 1997 to 2004. Annals of the Academy of Medicine, Singapore, 35, 674-679.
- Tate, J.E., Simonsen, L., Viboud, C., et al. (2008) Trends in Intussusceptions Hospitalizations among US Infants, 1993-2004: Implications for Monitoring the Safety of the New Rotavirus Vaccination Program. Pediatrics, 121, e1125-e1132. http://dx.doi.org/10.1542/peds.2007-1590
- Rao, P.L., Prasad, C.N., Mitra, S.K., et al. (1979) Intussusception in Infancy and Childhood. Indian Journal of Pediatrics, 46, 126-134. http://dx.doi.org/10.1007/BF02896746
- WHO (2000) Report of the Meeting on Future Directions for Rotavirus Vaccine Research in Developing Countries. World Health Organization, Geneva, 9-11 February 2000.
- Anatol, T. (1985) The Pattern of Gastro-Intestinal Obstruction in Trinidadian Children. West Indian Medical Journal, 34, 238-243.
- Rennels, M.B., Parasher, U.D. and Holman, R.C. (1998) Lack of Apparent Association between Intussusception and Wild or Vaccine Rotavirus Infection. The Pediatric Infectious Disease Journal, 17, 924-925. http://dx.doi.org/10.1097/00006454-199810000-00018
- Blanch, A.J., Perel, S.B. and Acworth, J.P. (2007) Paediatric intussusception: epidemiology and outcome. Emerg Med Australas; 19:45-50. http://dx.doi.org/10.1111/j.1742-6723.2007.00923.x
- Ho, W.L., Yang, T.W., Chi, W.C., et al. (2005) Intussusception in Taiwanese Children: Analysis of Incidence, Length of Hospitalization and Hospital Costs in Different Age Groups. Journal of the Formosan Medical Association, 104, 398-401.
- Buettcher, M., Baer, G., Bonhoeffer, J., et al. (2007) Three-Year Surveillance of Intussusception in Children in Switzerland. Pediatrics, 120, 473-480. http://dx.doi.org/10.1542/peds.2007-0035
- Awasthi, S., Agarwal, G.G., Mishra, V., et al. (2009) Four-Country Surveillance of Intestinal Intussusception and Diarrhoea in Children. Journal of Paediatrics and Child Health, 45, 82-86. http://dx.doi.org/10.1111/j.1440-1754.2008.01434.x
- Chen, Y.E., Beasley, S. and Grimwood, K. (2005) Intussusception and Rotavirus Associated Hospitalisation in New Zealand. Archives of Disease in Childhood, 90, 1077-1081. http://dx.doi.org/10.1136/adc.2005.074104
- Parashar, U.D., Holman, R.C., Cummings, K.C., et al. (2000) Trends in Intussusception-Associated Hospitalizations and Deaths among US Infants. Pediatrics, 106, 1413-1421. http://dx.doi.org/10.1542/peds.106.6.1413


