Open Journal of Inorganic Chemistry
Vol. 3 No. 3 (2013) , Article ID: 33621 , 4 pages DOI:10.4236/ojic.2013.33008
Synthesis and characterization of a ladder-like structure compound formed by cadmium (II) and anionic nitronyl nitroxide
![]()
1College of Chemistry and Chemical Engineering, Anyang Normal University, Anyang, China
2School of Chemical Engineering and Energy, Zhengzhou University, Zhengzhou, China
Email:*chenjinghao2008@163.com
Copyright © 2013 Jing Chen et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 19 March 2013; revised 22 April 2013; accepted 30 April 2013
Keywords: Nitronyl nitroxide; Anionic radical; Ladder-like structure; Cd (II) complex
ABSTRACT
A ladder-like structure compound formed by cadmium (II) and anionic nitronyl nitroxide, [Cd2(NITpBA)4(H2O)4] (where NITpBA = 2-(4-carboxyphenyl)4,4,5,5-tetramethyl-4,5-dihydro-imidazol-1-oxyl-3-oxide), has been synthesized and characterized. X-ray analysis reveals that the compound 1 crystallizes in the monoclinic P21/c (No. 14) space group, and [Cd2(NITpBA)4(H2O)4] units are linked into infinite chains by radical bridging ligands.
1. INTRODUCTION
The field of molecule-based materials has attracted intense interest for decades [1]. The design and synthesis molecular-based magnetic materials by using metalradical approach have become one of the very promising strategies [2]. The organic radicals can act as not only spin carriers but also building blocks. In this field, the increasing attention has been focused on the coordination complexes of paramagnetic metal ions with organic radical ligands in order to exploit new molecular magnetic materials and diverse topological structures [3,4]. The possibility to play with anionic nitronyl nitroxides increases the chances for designing magnetic molecular materials and diverse structures [5,6]. The stable anionic radical NITpBAH, named as 2-(4-carboxyphenyl)-4,4, 5,5-teramethyl-4,5-dihydro1H-imidazol-1-oxyl-3-oxide, has two different types of potentially metal-coordinating site. One is the carboxylate group; the other consists of the two oxygen atoms belonging to the nitronyl nitroxide part. Recently, due to the increased recognition of its role in biological organisms, there has been an increasing interest in the coordination chemistry of cadmium [7].
With the purpose to explore a new polynuclear system derived from nitroxide radicals, we attempted to synthesize transition metal complexes incorporating dianion bridges. In this paper, we report the synthesis and the structural characterization of the first cadmium(II) complex with NITpBA−, of formula [Cd2 (NITpBA)4(H2O)4] (NITpBA = 2-(4-carboxy-phenyl)-4,4,5,5-tetramethyl-4,5- dihydro-imidazol-1-oxyl-3-oxide), in which a ladder-like one dimensional structure formed by cadmium (II) and anionic nitronyl nitroxide ligand.
2. EXPERIMENTAL
2.1. Materials and Measurements
All chemicals used were of reagent grade and used without further purification. The NITPBA was prepared by the literature method [8]. Elemental analysis for carbon, hydrogen and nitrogen were carried out on a PerkinElmer elemental analyzer model 240. The IR spectrum was taken on a Shimadzu 408 IR spectrophotometer in the 4000 - 400 cm−1 region with KBr pellets.
2.2. Synthesis of [Cd2(NITpBA)4(H2O)4]
To a stirred solution (10 mL) of Cd (CH3COO)2·2H2O (0.2 mmol) in methanol was added a methanol solution (10 mL) of NITpBAH (0.4 mmol). The mixture was stirred continually for 1 h and then filtered. The filtrate was kept at room temperature for slow evaporation. After a few days, dark blue single crystals suitable for X-ray analysis appeared. Yield: 38%. Anal. Calcd. for C28 H36 Cd N4 O10 1: C, 47.93; H, 5.18; N, 7.99%. Found: C, 47.89; H, 5.13; N, 8.03%. IR (KBr disk): 1630 cm−1
(νasCOO−), 1380 cm−1 (νsCOO−), 1365 cm−1 (νNO).
A dark blue crystal with dimensions 0.20 × 0.20 × 0.20 mm3 was mounted on a glass fiber in a random orientation. The determination of the unit cell and data collection was performed on a computer-controlled Bruker APEX-II CCD diffractometer. 3635 independent re- flections [R(int) = 0.0203] in the range 1.63˚ ≤ θ ≤ 28.35˚ with −18≤ h ≤ 18, −12 ≤ k ≤ 11, −12 ≤ l ≤ 16 were collected by using an φ – ω scan technique at 293(2) K using Mo-Kα radiation with a graphite monochromator (λ = 0.71073A). The X-ray structure analysis revealed that the compound belongs to the monoclinic system, space group P21/c. Absorption corrections were applied using SADABS program. The structure of the complex was solved using direct methods by SHELXS-97 program [9]. Cadmium atom was located from an E-MAP. The other non-hydrogen atoms were determined with successive difference Fourier syntheses. The final refinement used full-matrix least-squares method by the SHELXL-97 [10]. Crystallographic data and refinement parameters are listed in Table 1. Atomic coordinates and selected bond distances and bond angles are given in Tables 2 and 3, respectively.

Table 1. Crystal data and structure refinement for the compound 1.
3. RESULTS AND DISCUSSION
Description of the Crystal Structure
Single-crystal X-ray diffraction analysis reveals that complex 1 and the reported compound Mn2(NITpBA)4 (H2O)4 [8] are isostructural. The title complex 1 is the first antimagnetic metal compound involving anionic nitronyl nitroxide ligand. An ORTEP drawing of the asymmetric unit of Cd2(NITpBA)4(H2O)4 is shown in Figure 1. The crystal structure of the complex 1 is shown in figure 2, which reveals a ladder-like one di-
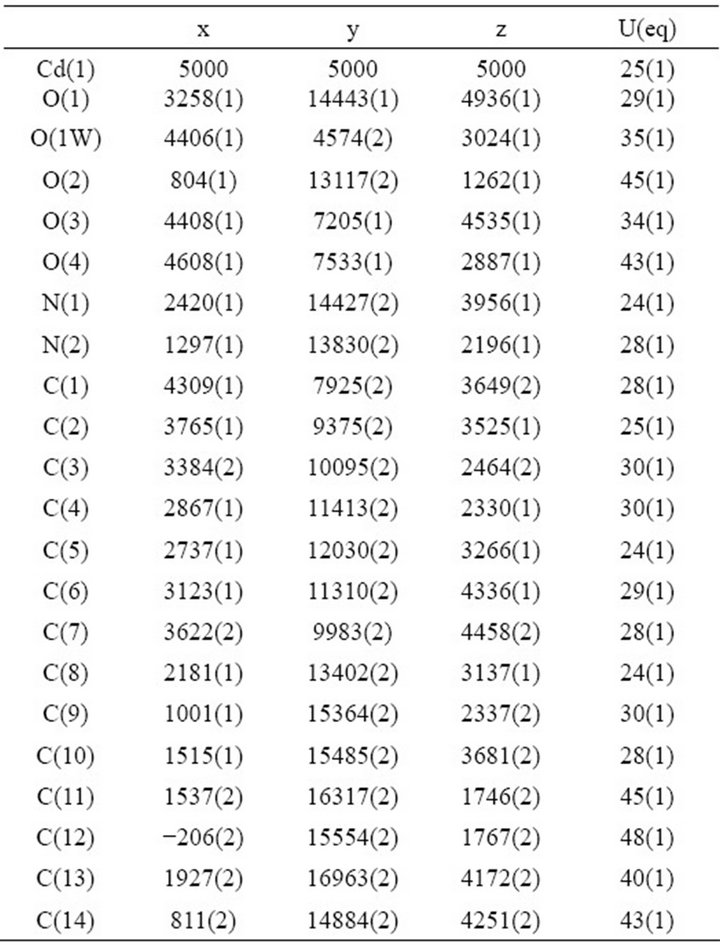
Table 2. Atomic coordinates (×104) and equivalent isotropic displacement parameters (A2 × 103) for 1. U(eq) is defined as one third of the trace of the orthogonalized Uij tensor.
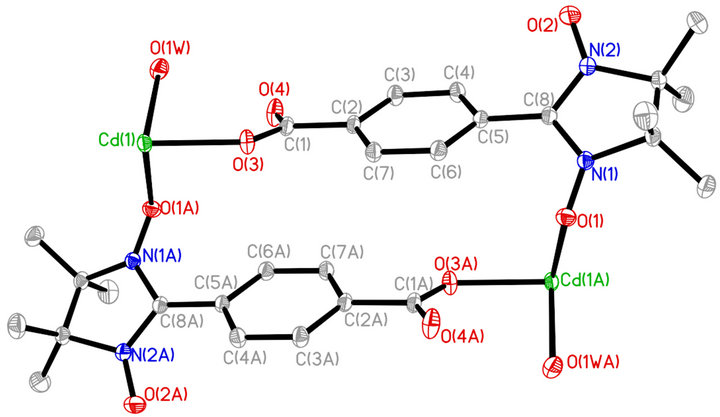
Figure 1. ORTEP of Cd2(NITpBA)4(H2O)4 drawn at the 30% probability level. All hydrogen atoms and disordered fluorine atoms are omitted for clarity.
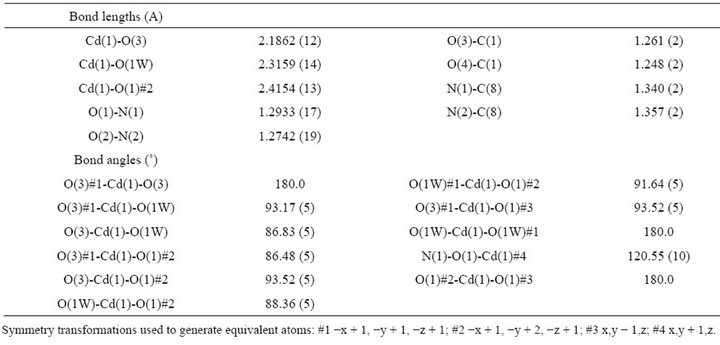
Table 3. Bond lengths (A) and angles (˚) for 1.
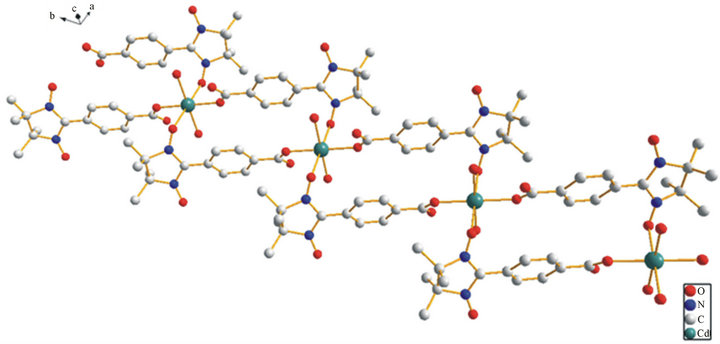
Figure 2. Draw of the ladder-like structure of the title compound 1.
mensional chain of repeating Cd2(NITpBA)4(H2O)4 units.
The Cd (II) atom exhibits a slightly distorted octahedral coordination sphere and lies on an inversion center. The Cd (II) ion is coordinated by two pairs radical which trans to each other. One pair binds through one oxygen atom of the carboxylic anion, while the other coordinates through one of the NO group. Four oxygen atoms from nitronyl nitroxide ligand form the equatorial plane. Each nitronyl nitroxide coordinats the adjacent Cd (II) atom in the opposite fashion thus forming the chains. The Cd (II) ion is coordinatively saturated by two H2O molecules in axial positions, thus completing the CdO6 core. The Cd-O distances involving both types of radical oxygen atoms are markedly different to each other. The Cd-O3 (O from carboxy group) is 2.1862 A, which is shorter than that Cd-O1 (O from nitroxide, 2.4154 A). The axial Cd-O 1W bond is equal to 2.3159 A. The N-O distances are slightly different from each other, and they are shorter than those found in NITpBAH, which are in the range of the typical distances of this kind of nitronyl nitroxide derivatives [10-12]. The band distance of the coordinated N-O in the radical is 1.2933 A, slightly longer than that of the uncoordinated N-O (1.2742 A). The bond of coordinated C1-O3 of the carboxyl group is 1.261 A, longer than that of the uncoordinated C1-O4 (1.2482 A). The fragment O1-N1-C8-N2-O2 is nearly planar, and forms a dihedral angle of 39.5(1)˚ with the plane of the phenyl ring. The dihedral angle between the phenyl ring and the carboxylic group is 14.6(3)˚. In the linear chain, the distance between successive Cd (II) ions is 9.2637 A. Interchain contacts are found between the water molecule oxygen O and the non-coordinated carboxylic oxygen O separated by 2.768 (2) A, indicative of hydrogen bonding.
In summary, we have successfully obtained the first ladder-like structure compound formed by cadmium (II) and anionic nitronyl nitroxide ligand, of formula [Cd2 (NITpBA)4(H2O)4] (NITpBA = 2-(4-carboxy-phenyl)-4,4, 5,5-tetramethyl-4,5-dihydro-imidazol-1-oxyl-3-oxide anion).
4. SUPPLEMENTARY MATERIAL
Detailed crystallographic data in CIF format for the title complex is available from Cambridge Crystallo-graphic Data Center (CCDC reference number: No. 898048).
5. ACKNOWLEDGEMENTS
The work was supported by the National Natural Science Foundation of China (No. 21071006), the Natural Science Foundation of Henan Province (No. 102102210457) and the Natural Science Foundation of the Henan Higher Education Institutions of China (No. 2010B150001).
![]()
![]()
REFERENCES
- Kahn, O. (1993) Molecular magnetism. Vancouver Coastal Health, New York.
- Caneschi, A. Gatteschi, D. Sessoli, R. and Rey, P. (1989) Toward molecular magnets: The metal-radical approach. Accounts of Chemical Research, 21, 392-398. doi:10.1021/ar00167a004
- Numata, Y. Inoue, K. Baranov, N. Kurmoo, M. and Kikuchi, K. (2007) Field-induced ferrimagnetic state in a molecule-based magnet consisting of a Co (II) ion and a chiral triplet bis(nitroxide) radical. Journal of American Chemical Society, 129, 9902-9909. doi:10.1021/ja064828i
- Maspoch, D. Domingo, N. Ruiz-Molina, D. Wurst, K. Vaughan, G. Tejada, J. Rovira, C. and Veciana, J. (2004) A robust purely organic nanoporous magnet. Angewandte Chemie International Edition, 43, 1828-1832.
- Fegy, K. Sanz, N. Luneau, D. Belorizky, E. and Rey, P. (1998) Proximate nitroxide ligands in the coordination spheres of manganese (II) and nickel (II) ions. Precursors for high-dimensional molecular magnetic materials. Inorganic Chemistry, 37, 4518-4523. doi:10.1021/ic971618l
- Laget, V. Hornick, C. Rabu, P. Drillon, M. and Ziessel, R. (1998) Molecular magnets hybrid organic-inorganic layered compounds with very long-range ferromagnetism. Coordination Chemistry Reviews, 178-180, 1533-1553. doi:10.1016/S0010-8545(98)00166-0
- Huang, C.F. Wei, H.H. Lee, G.H. and Wang, Y. (1998) Structure and magnetic properties of a novel chloro-bridged polymeric cadmium (II) complex with pyridyl-subsituted nitrony nitroxide. Inorganica Chimica Acta, 279, 233-237. doi:10.1016/S0020-1693(98)00105-4
- Schiødt, N.C. Biani, F.F. Caneschi, A. and Gatteschi, D. (1996) Structure andmagnetism of nickel (II) and manganese (II) complexes of a nitronyl nitroxide carboxylic acid. Inorganica Chimica Acta, 248, 139-146. doi:10.1016/0020-1693(95)04996-7
- Sheldrick, G.M. (1990) SHELXS-97, program for X-ray crystal structure solution. University of Gottingen, Germany.
- Sheldrick, G.M. (1997) SHELXL-97, program for X-ray crystal structure refinement. University of Gottingen, Germany.
- Hirel, C. Li, L.C. Brough, P. Vostrikova, K. Pécaut, J. Mehdaoui, B. Bernard, M. Turek, P. and Rey, P. (2007) New spin-transition-like-nitroxide species. Inorganic Chemistry, 46, 7545-7552 doi:10.1021/ic700851x
- Chen, Y.W. Wei, H.X. Wang, X.G. Gao, D.Z. Sun, Y.Q. Zhang G.Y. and Xu, Y.Y. (2012) Syntheses, crystal structures, and magnetic properties of three 1D chain metal—Nitroxide complexes bridged by flexible dicerrboxylate anions. Zeitschrift für anorganische und allgemeine Chemie, 638, 1328-1334 doi:10.1002/zaac.201200056
- Chen, J. Liao, D.Z. Jiang, Z.H. and Yan, S.P. (2005) A new heterospin complex from oxamide-bridged Cu (II) binuclear units and carboxy-phenyl-substituted nitronyl nitroxide. Inorganic Chemistry Communications, 8, 564- 567. doi:10.1016/j.inoche.2005.02.015
NOTES
*Corresponding author.

