Journal of Behavioral and Brain Science
Vol. 2 No. 1 (2012) , Article ID: 17732 , 4 pages DOI:10.4236/jbbs.2012.21009
Ratanasampil (Tibetan Medicine, RNSP) Reduces β-Amyloid Protein (Aβ) and Pro-Inflammatory Factor Levels and Improves Cognitive Functions in Mild-to-Moderate Alzheimer’s Disease (AD) Patients Living at High Altitude
Institution of Geriatric, Qinghai Provincial Hospital, Xining, China
Email: *zhuaq@hotmail.com
Received August 18, 2011; revised October 13, 2011; accepted November 4, 2011
Keywords: Ratanasampil (Tibetan Medicine, RNSP); Alzheimer’s Disease; β-Amyloid Peptide; Aβ42/Aβ40 Ratio; Pro-Inflammatory Factors; Cognitive Function
ABSTRACT
Ratanasampil (RNSP) is a traditional Tibetan medicine used for the treatment of stroke and cerebrovascular diseases. Previous discoveries that RNSP can reduce β-amyloid protein levels and increase learning and memory in Alzheimer’s mouse models (Tg2576) led us to investigate whether RNSP can improve cognitive functions in Alzheimer’s patients. In this study, 146 AD patients living in Qinghai province received either one gram or 0.33 gram daily of RNSP for 16 weeks. Placebo patients received Piracetam. Serum Aβ40 and Aβ42 levels were measured at the beginning of the study and after 4 and 16 weeks of treatment. Compared to the same group before treatment, MMSE scores, ADAS-cog scores and ADL scores were significantly improved (p < 0.01, p < 0.01 and p < 0.05, respectively) in patients after 16-week treatment with high-dose RNSP. No significant differences were observed in either the low-dose RNSP or placebo groups (p > 0.05, p > 0.05). After 16-week treatment, serum TNF-α, IL-1β, IL-6 and Aβ42 levels were significantly decreased (p < 0. 01) in the high-dose RNSP group, whereas no significant differences were found in the low-dose and placebo groups. The Aβ42/Aβ40 ratio was significantly decreased after 4-week and 16-week treatment in the high-dose RNSP group (p < 0. 05, p < 0.01). Furthermore, serum Aβ42 concentrations had a strong positive correlation with TNF-α, IL-1β and IL-6 levels. There were no observable adverse effects in either treatment or control groups. We conclude that further clinical trials of RNSP in Alzheimer disease are warranted.
1. Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disease commonly seen in the elderly population of developed countries [1,2] as well as in China [3]. The precise mechanisms that cause AD are still unknown. Multiple pathways are known to contribute to the cognitive deficits found in AD by disruption of neuronal signalling involved in learning and memory. These pathways are altered by β-amyloid peptide (Aβ) production and accumulation, aberrant tau phosphorylation, inflamemation, oxidative damage, and dysregulation of neurontransmitters and receptors [4]. Many efforts have been made to develop pathway-specific interventions to block causal events in AD pathogenesis and to improve cognitive function in AD patients.
Various studies have shown that immune-mediated inflammatory responses are closely associated with AD pathogenesis [5]. Overproduction of pro-inflammatory cytokines, such as tumor necrosis factor (TNF-α), inter-leukin-1β (IL-1β) and IL-6, triggers pro-inflammatory responses and promotes neuronal cell damage and excessive Aβ deposition. Both Aβ and pro-inflammatory factors are activators of neurotoxic pathways that lead to brain cell dysfunctions and death [6-8]. Therefore, anti-inflammatory drugs may help to improve brain function in patients with Alzheimer’s disease. Several epidemiological studies have shown that patients on long-term use of non-steroidal anti-inflammatory drugs (NSAIDs) have a lower risk of developing AD than others of the same age group [9].
Traditional Chinese Medicine (TCM) have been used for the treatment of dementia and reportedly improve cognitive function and capabilities of daily living [10]. Ratanasampil (RNSP), which means seventy-taste-pearlballs in the Tibetan language has been used for the treatment of stroke and cerebrovascular diseases. Our previous studies showed that RNSP reduced β-amyloid protein levels and increased learning and memory in Alzheimer’s mouse models (Tg2576) [11,12]. Here, we report that RNSP improved cognitive functions in mild-to-moderate Alzheimer’s patients living at high altitude. Furthermore, we found that RNSP also decreased serum Aβ42 concentration and therefore the Aβ42/Aβ40 ratio, and reduced the level of several pro-inflammatory factors, including TNF-α, IL-1β and IL-6. These data suggest that RNSP could be a new therapeutic drug for the treatment and prevention of AD.
2. Subjects and Methods
2.1. Subjects
146 patients with mild-to-moderate AD living in Xining and its contiguous areas (at altitudes of 2000 - 3200 meters) in the Province of Qinghai, were recruited for this study during the period of January 2005 to December 2007. Out of 146 patients, 5 patients were lost to followup and 1 patient died of pneumonia during the treatment. Therefore 140 patients completed the 16-week of treatment.
The study took place at the Institution of Geriatric of Qinghai Provincial Hospital. These patients were divided into three groups: high-dose RNSP treatment (1 g/day), low-dose RNSP treatment (0.33 g/day) and controls. All three groups were carefully selected to be comparable with respect to age, gender, handedness, level of education, MMSE score, and baseline functioning. Enrolled patients were required to have stable medical conditions for at least 3 months prior to the study and to have an absence of systemic disorders that might confound a diagnosis of AD.
2.2. Eligibility Requirements for Participants
The diagnosis of mild-to-moderate AD was made by the employing “Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV)”criteria and the “National Institute of Neurological Disorders and Stroke/ Alzheimer’s Disease and Related Disorders Association (NINDS/ADRDA)” criteria [13]. Briefly, a mini-mental state examination (MMSE) score between 10 and 24 points; a Hachinski ischemia score ≤ 4 points; and a Hamilton depression scale score (HAMD) < 7 points was used to diagnose mild-to-moderate AD. Brain CT scans and MRIs showed temporal lobe atrophy, a smaller hippocampus, ventricular dilatation, cortical atrophy and white matter lesions.
2.2.1. Exclusion Criteria for This Study Included
1) Vascular dementia or other physical or chemical factors causing dementia such as intracranial lesions.
2) Serious heart, lung, liver, kidney, blood diseases, or other serious mental illness.
3) Any critical condition, because it is difficult to evaluate accurately drug efficacy and safety.
4) Taking mental performance enhancement drugs (smart drugs, central nervous system stimulants), chronic nonsteroidal anti-inflammatory drugs or hormone supplements within 4 weeks of initial treatment.
Patients or their guardians agreed to participate in this study and signed informed consent forms. The protocol was approved by the Qinghai Medical Ethics Committee.
2.3. Study Design
2.3.1. A Randomized, Double-Blind, Controlled Trial of Drug Design
The high-dose treatment group was given a single oral dose of 1 g of (RNSP every day Qinghai Jinke Tibetan Medicine Pharmaceutical Co. LTD, Zhunzi Z63020062). The low-dose treatment group received a single oral dose of 0.33 g/day of RNSP. The control group was treated with a single 24 mg dose of piracetam per day (WuXi KaiXi Pharmaceutical Co., Ltd.).
RNSP and the control drug were re-packed at the Center into the same size and color capsules. The center randomly assigned numbers to the drugs and distributed them to the patients.
All patients did not take any mental enhancement drugs or central nervous system stimulants during the whole procedure (16 weeks). However, some complications such as hypertension and diabetes were treated accordingly and any drug adverse reactions were recorded.
2.3.2. Efficacy Assessment
All patients were assessed 3 times (before treatment, after 4 weeks of treatment, and at the end of treatment (16 weeks) by accredited psychologists using MMSE, ADAScog and the ADL measurements.
1) The Alzheimer’s Disease Assessment Scale cognitive subscale (ADAS-cog) was used to assess the patient’s cognitive function, including memory, attention, language, orientation, visuo spatial, skill and ability to understand and operate 12 tasks. The lower the score, the better was the cognitive function [14].
2) The Mini-Mental State Examination (MMSE) was also used to assess the patient's cognitive function. This measures 5 factors: orientation in time and space, instanttaneous recall and short term memory, attention and calculation ability, language, and constructional capacities. The higher the MMSE score, the better was the cognitive function [15].
3) The activity of daily living scale (ADL) was used to determine a person’s ability to perform tasks of daily living. This includes 6 items of physical assessment screenings (self-care) and 8 items of functional assessment screening (tool-use ability). The lower the score means the better the ability of daily living [16].
2.3.3. Tolerability and Safety Assessment
Physical examination included vital signs (blood pressure, heart rate, and body temperature), 12-lead electrocardiograph (ECG), and laboratory tests (including serum chemistry, hematology, and urinalysis). They were conducted before drug administration, after 4 weeks of treatment and at the end of the study period after 16 weeks of treatment.
All laboratory tests were performed at an accredited central laboratory of the Qinghai Provincial Hospital. Adverse events were assessed throughout the study period based on direct questioning and spontaneous reports, and were recorded regardless of their suspected relations to the study drugs. All data obtained from the study were monitored by qualified investigators.
2.3.4. Aβ and Pro-Inflammatory Cytokines Measurements
Blood samples (10 ml) were collected from all patients before drug administration, after 4 weeks of treatment and at the end of the study period after 16 weeks of treatment. The samples were centrifuged to separate the serum which was stored in –70˚C until testing Aβ40 and Aβ42 levels were measured using an enzyme linked immunosorbent assay, (ELISA, Adlitteram Diagnostic. Laboratories Co. (USA) Corporation) according to the manufacturer’s protocol.
IL-1β, IL-2, IL-6, IL-8 and TNF-α concentrations were measured by radioimmunoassay (Tianjin Jiuding Medical and Biological Co., Ltd.), following the manufacturer’s recommendations.
2.4. Statistical Analyses
The statistical package SPSS for Windows Ver. 11.5 was used to do statistical analyses. Pearson’s correlation was used for linear correlation analysis. All data are shown as mean ± standard deviation (±SD). Multiple group comparisons were made using analysis of variance (ANOVA). Comparisons between the groups were made by t test. p < 0.05 was considered statistically significant.
3. Results
3.1. The Demographic Characteristics of the Study Population Are Not Significantly Different between the High-Dose RNSP (Single Dose, 1 g/day), Low-Dose RNSP (Single Dose, 0.33 g/day) and Control Groups
The demographic characteristics of the study population are presented in Table 1. All patients were ethnic Han Chinese. Low-dose, high-dose, and placebo groups comprised respectively 40 patients (19 males and 21 females), 50 patients (23 males and 27 females), and 50 patients (25 males and 25 females). The patients ranged in age from 60 to 85 years old (mean age 74.5 ± 8.1 years old) in the low-dose group, from 59 to 84 years old (mean age 75.2 ± 6.9 years old) in the high-dose group and from 61 to 85 years old (mean age 74.7 ± 6.9 years old) in the control group. All three groups had similar education levels. Psychological tests showed that all the patients were in the mild to moderate range of AD. Based upon a review of psychological testing records, the average length that the participants had been diagnosed with AD was 2 years for all 3 groups. The basal MMSE scores prior to our treatment were 17.80 ± 4.60 in the low-dose treatment group, 17.89 ± 5.29 in the high-dose treatment group, and 17.57 ± 5.68 in the control group. Statistical analyzes showed that the demographic characteristics (gender, age, ethnicity, height and weight), education levels, average length of patients diagnosed as AD and basal MMSE scores of all three groups were not significantly different.
Figure 1 displays a flow diagram that describes the
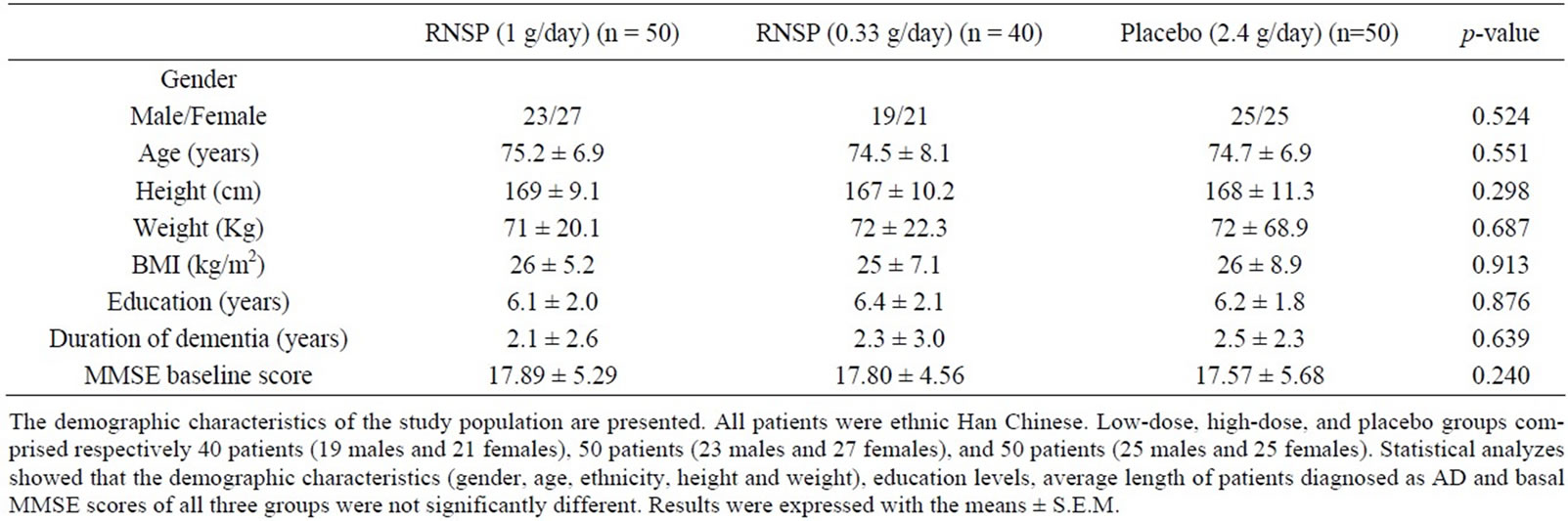
Table 1. Demographic characteristics of the study population by group at baseline.
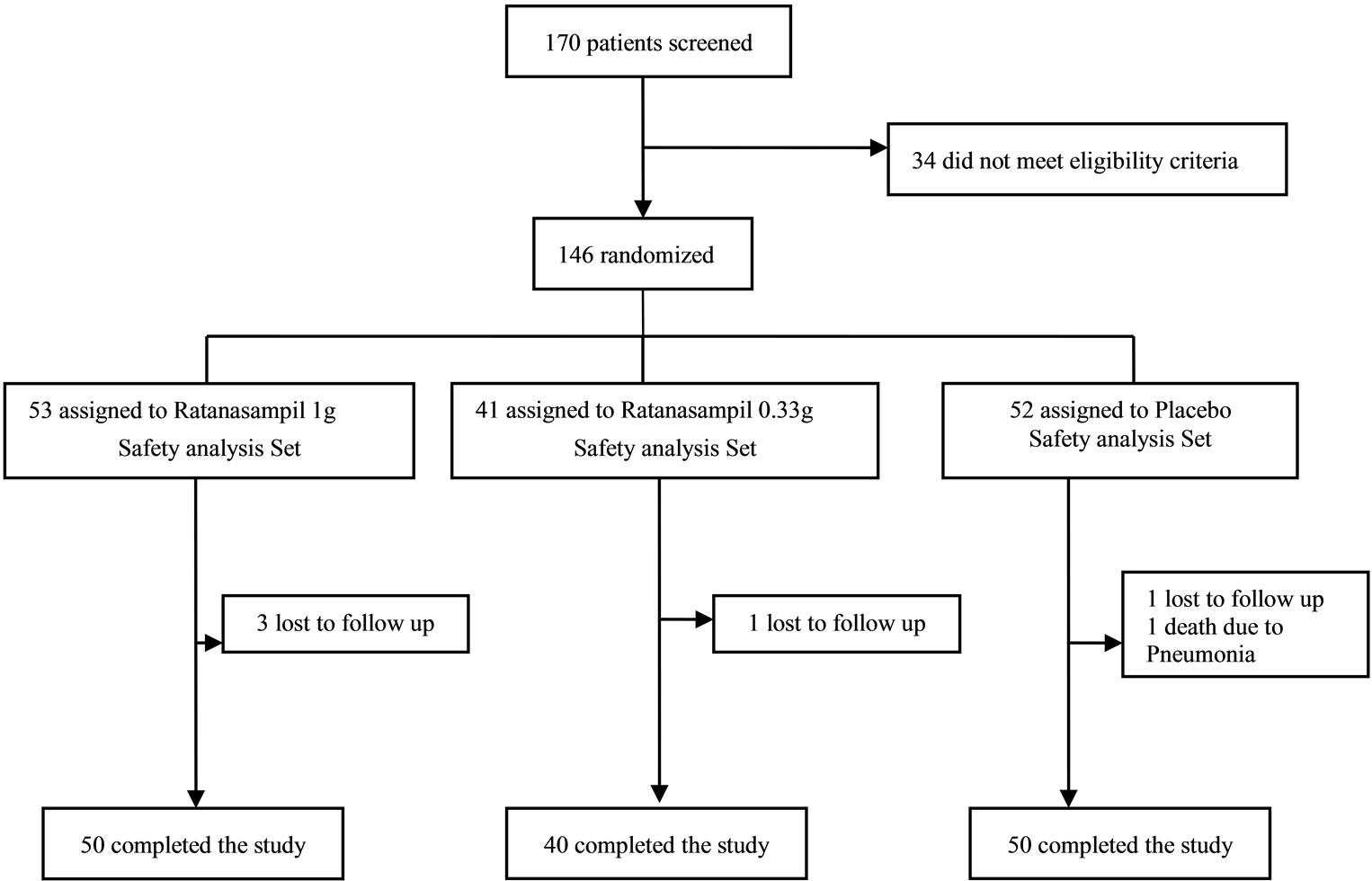
Figure 1. The figure displayed a flow diagram that describes the trial from screening until the end of the study. 170 patients were screened and 34 failed meet the eligibility criteria. 146 patients were enrolled in this study.
trial from screening until the end of the study. 170 patients were screened and 34 failed meet the eligibility criteria. 146 patients were enrolled in this study. 53 patients began the high-dose RNSP treatment (1 g/day), 41 patients began the low-dose RNSP (0.33 g/day) and 52 patients began placebo. In the end, 50 patients (attrition rate 5.7%) in the high-dose RNSP treatment group, 40 patients (attrition rate 2.4%) in the low-dose RNSP treatment group and 50 patients (attrition rate 3.8%) in the placebo group remained in the study after 16-weeks. The reasons for attrition included lost to follow up and death.
3.2. High-Dose RNSP (1 g/day) Significantly Improved Cognitive Functions in Mild-to-Moderate Alzheimer’s Patients after 16-Week Treatment
All 3 groups (high-dose, low-dose, and control) were evaluated at the beginning of treatment, at 4 weeks and at the 16 week termination. MMSE scores, ADAS-cog scores and ADL scores for each group are presented in Table 2 and 3 and Figure 2. The basal MMSE, ADAS-cog and ADL scores were similar among the 3 groups (p > 0. 05). After 4 weeks of treatment, the high-dose RNSP group showed significant improvement in MMSE scores (3 out of 5 sub-scores including orientation in time and space, instantaneous recall and short term memory were significantly improved) and ADAS-cog scores. However, the ADL score was only slightly changed after 4 weeks of treatment (p > 0.05). Interestingly, after treatment for 16 weeks, the high-dose RNSP group showed a further significantly improved MMSE score (all 5 sub-scores were significantly improved), much decreased ADAScog scores (better cognitive functions), and significantly improved ADL scores(both physical and functional assessment screenings were significantly improved). Although there was a slight improvement in the low-dose RNSP treatment group, the difference was not statistically significant (p > 0.05).
3.3. High-Dose RNSP Significantly Decreased the Levels of Aβ42 and Pro-Inflammatory Factors Associated with Alzheimer’s Disease
Blood samples from all 3 groups (high-dose, low-dose and control) were collected 3 times during the 16-week treatment (at the beginning of treatment, at 4 weeks and 16 weeks after treatment) to examine the levels of Aβ and several AD-related pro-inflammatory factors. The results are shown in Table 4. The basal levels of Aβ, TNF-α, IL-1β, IL-6, IL-8 were similar among all 3 groups (p > 0.05). After 4 weeks of treatment, TNF-α, IL-1β, IL-6 and Aβ42 levels were significantly reduced in the highdose RNSP treatment group compared to the control group,
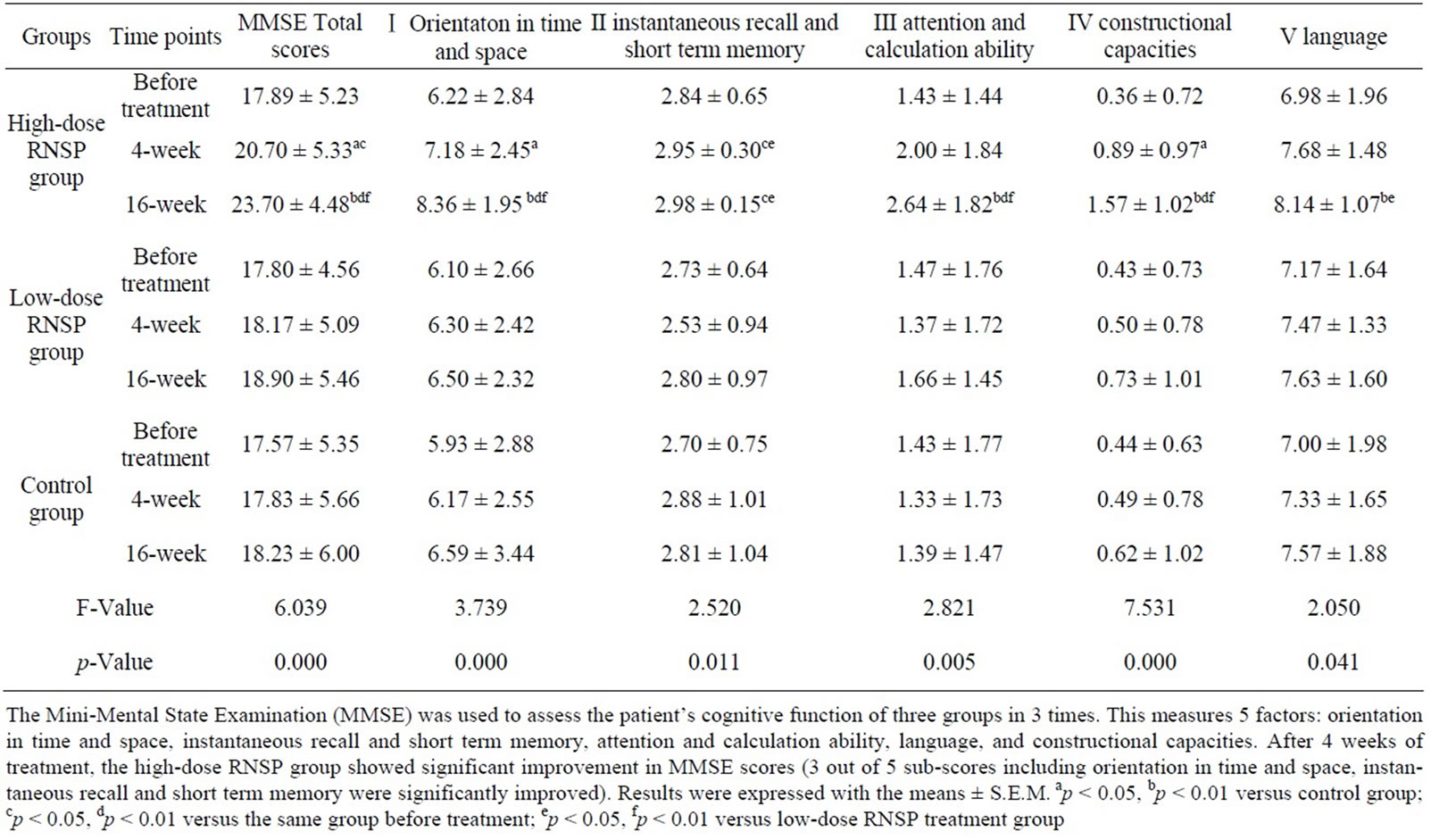
Table 2. MMSE scores (including all 5 sub-scores) before and after treatment (4-weeks and 16-weeks of treatment) in the 3 groups (mean ± SD points).
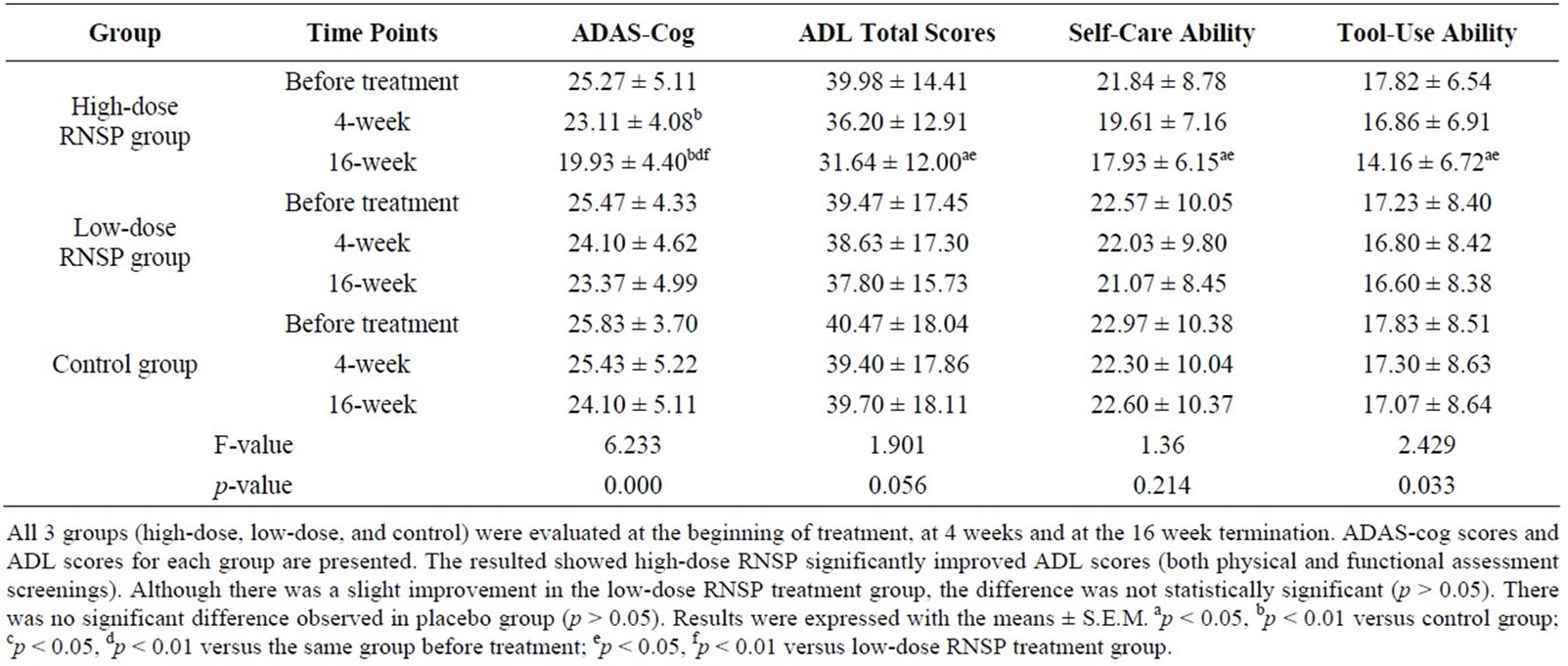
Table 3. ADAS-cog scores and ADL scores (including 2 aspects of screenings) before and after treatment (4-weeks and 16-weeks of treatment) in the 3 groups (mean ± SD points).
(p < 0.05). Interestingly, after treatment for 16 weeks, the concentrations of TNF-α, IL-1β, IL-6 and Aβ42 were further decreased compared to the control group (p < 0.01). The Aβ42/Aβ40 ratio was significantly reduced after both 4-weeks and 16-weeks of treatment (p < 0.05, p < 0.01). However, there were no significant differences in IL-8 and Aβ40 levels between the high-dose RNSP treatment and control groups.
Compared to the control group, the levels of IL-6 in the low-dose RNSP treatment group were also decreased after 16 weeks of treatment, while no significant differences were observed in the levels of other pro-inflammatory factors. There was also no difference in the control group before and after treatment.
Linear correlation analyses showed that serum Aβ42 levels had a positive correlation with the levels of TNF-α (r = 0.564), IL-6 (r = 0.271) and IL-1β (r = 0.205) (p < 0.01, p < 0.05 and p < 0.05). However, we found no correlation between the Aβ42 levels and IL-8 levels (p > 0.05).
3.4. Toxicological Measurements
Clinical adverse events Patients from all 3 groups reported occasional nauseaanorexia, dizziness, mild diarrhea and dry mouth (See Table 5). These symptoms usually eased or disappeared without any special treatment. One patient from the control group died of pneumonia.
Laboratory Tests The heart rate, PR interval, QRS wave and QTC of ECG showed no significant changes before and after treatment. Serum chemistry, hematology, and urinalysis (liver and kidney function, glucose, lipids, electrolytes) before and after treatment showed no significant changes in all 3 groups.
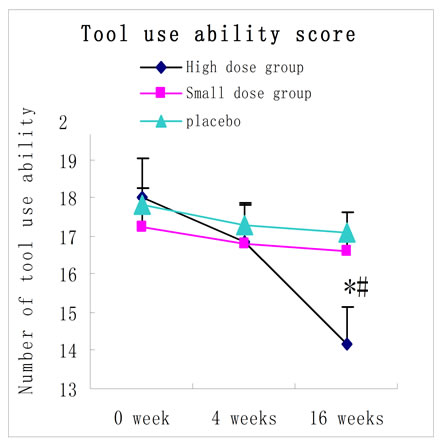
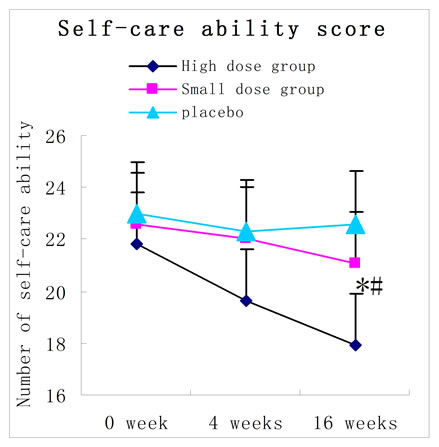
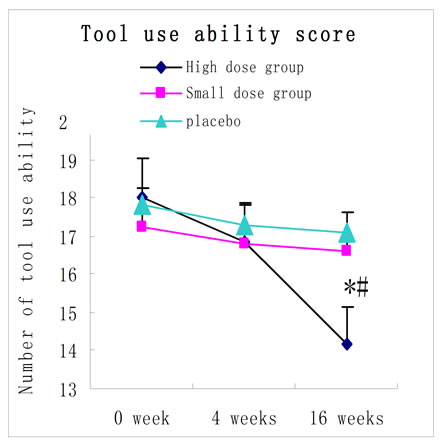
Figure 2. ADL total scores and its two sub-scores before and after treatment (4-weeks and 16-weeks treatment) in the 3 groups (mean ± SD points).
The basal ADL scores were similar among the 3 groups (p > 0. 05). However, the ADL score was only slightly changed after 4 weeks of treatment (p > 0.05). After treatment for 16 weeks, the high-dose RNSP group showed a further significantly improved ADL scores (both physical and functional assessment screenings were significantly improved). Results were expressed with the means ± S.E.M. *p < 0.05 versus control group; #p < 0.05 versus the same group before treatment.
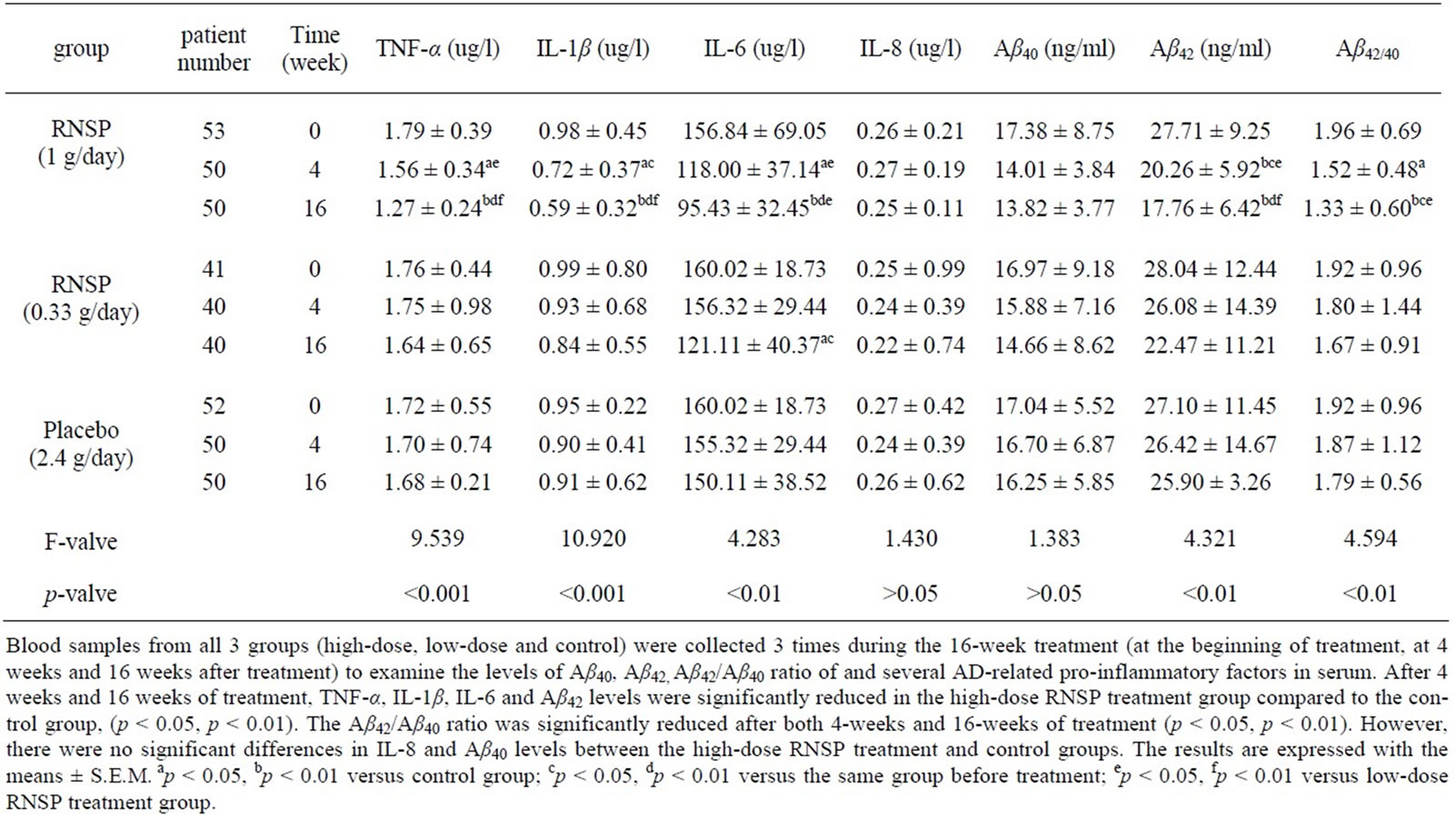
Table 4. Serum levels of TNF-α, IL-1β, IL-6, IL-8, Aβ40 and Aβ42 before and after treatment (4-weeks and 16-weeks of treatment) in the 3 groups (mean ± SD).
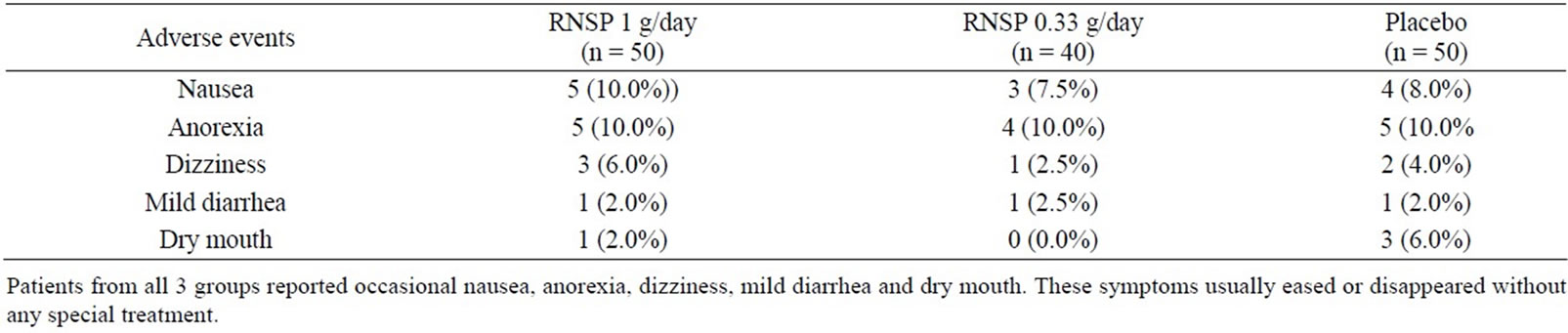
Table 5. Adverse events reports for three groups.
4. Discussion
Patients with AD suffer progressive memory loss, cognitive dysfunction, impairment of language skills, impairment in activities of daily living and behavioral and psychological abnormalities. AD ranks fourth among the leading causes of death, after cardiovascular diseases, cancer and stroke. In 2004, there were an estimated 18 million people suffering from AD worldwide according to the World Health Organization (WHO), with approximately 5 million AD patients in China. The prevalence rate for the population aged 60 years and over was about 1.26% [17]. According to the WHO, the AD population was expected to double to 34 million around 2025 [18]. While the rate of dementia is expected to double between 2001 and 2040 in developed nations, it is predicted it will increase by more than 300 percent in India, China and South Asia [19].
Qinghai is located on the Qinghai-Tibet Plateau. The climate in Qinghai is that of a typical continental plateau, which is dry, windy, and cold, with huge temperature differences between each region, day and night. People living in these areas experience many environmental stresses, including chronic hypoxia, strong ultraviolet radiation, high altitude and relative isolation from the outside world. Besides the relatively backward economic and medical situations, a high number of the elderly population suffer declines in memory and cognitive function [20-22]. Exploring methods of treatment of AD has thus become a significant focus for current medical research in this area.
Traditional Chinese medicine (TCM) has a time-honored history of several thousand years and there is a very rich record of Chinese herbal medicine used to treat dementia, a significant feature of AD. However, Tibetan medicine has not yet been systematically tested in AD clinical research. About 1450, two contradictory schools of thought (northern and southern) arose in Tibet. Each school had its own views concerning prescription methods and the Four Medical Classics. Sukar, a representative of the southern school, and his disciples conducted research on diseases in southern Tibet and developed seventy-taste-pearl-balls (RNSP), which has been used as an anti-aging and anti-hypoxic drug ever since. It is regarded as one of the most important Tibetan medicine treasures [23]. In the eighteenth century, RNSP was included in the well-known “One-thousand prescriptions”, in which RNSP is described as able to treat various acute and chronic cerebrovascular diseases such as cerebral hemorrhage, cerebral infarction, epilepsy, and brain concussion. RNSP is composed of more than seventy components, including Zuotai, natural pearls, antelope horn, saffron, natural bezoar, benzoin, incense, and nine-eye stone. Through generations of the continuous practice of Tibetan medicine and recent clinical observations, it has been found that RNSP has sedative and anticonvulsant effects, improved learning and memory, improved circulation, reduced platelet aggregation, and antithrombotic properties [24,25].
In this study, we used comprehensive clinical and neurological rating systems to evaluate whether the Tibetan medicine RNSP could improve the cognitive function and activities of daily living in AD patients living at high altitude. We chose piracetam as the control medicine because it is a mild anti-inflammatory agent which has been tested and reported in numerous studies [26]. After high-dose RNSP (1 g/day) treatment for 4 weeks, the patients showed a significant improvement in the MMSE total score and the ADAS-cog score. Although compared to the control group there was an improvement in the ADL score as well, the difference was not statistically significant. After treatment for 16 weeks, the MMSE total scores (and its 5 sub-scores) and the ADAS-cog scores were further improved compared to the control group. ADL scores (both self-care ability and tool-use ability sub-scores) also improved significantly. However, there were no significant differences between the lowdose RNSP treatment group (0.33 g/day) and the control group in MMSE, ADAS-cog and ADL scores. These data suggested that high-dose RNSP treatment effectively improved clinical measures of cognitive functions in mild-to-moderate Alzheimer’s patients living at high altitude. Cognitive processes including perception, attention, memory, calculating, thinking, language and understanding, are one of the basic psychological processes. Cognitive functions not only affect emotional, behavioral and other psychological processes, but also have direct impact on the ability to perform activities of daily living. Therefore RNSP had positive effects on the quality of daily life in AD patients.
Neuroinflammation is now recognized as a crucial player influencing and linking Aβ deposition and neuronal damage in Alzheimer’s pathogenesis. Anti-inflammatory treatments are considered beneficial to AD patients. The possible mechanisms that inflammatory responses are involved in AD pathogenesis include the pro-inflammatory cytokine IL-1β as a key regulator of acute inflammatory processes in the CNS. Over-expression of IL-1β stimulates and accelerates the deposition of Aβ, which is an early event in AD pathogenesis [27]. IL-6 over-expression begins before the formation of senile plaques, suggesting that IL-6 plays an important role in their early development [28]. The IL-6 –174˚C allele in the promoter region of the IL-6 gene was over-represented in AD patients. Increased brain and serum IL-6 levels were associated with a higher risk of AD [29]. It has been found that there is a large amount of TNF-α around the senile plaques in the brains of AD patients. Aβ and other proteins found in the senile plaques are potent activators that induce inflammatory responses, accompanied by leukocyte activation and inflammatory cytokine production. The activation of microglia increases the expression of TNF-α, suggesting that abnormal expression of high levels of TNF-α could be closely related to the pathogenesis of AD [30,31]. Genetic studies suggest a significant anti-inflammatory role of IL-4, IL-8, IL-10 and IL- 13 in AD pathogenesis, although the mechanisms are not known [32].
Recent studies revealed that non-steroidal anti-inflammatory drugs reduced Aβ production and deposition in AD animal models [33,34]. Aβ is generated by the consecutive cleavage of the amyloid precursor protein (APP) by two proteases, β-secretase and γ-secretase [35]. Aβ is a protein consisting of two major forms, Aβ40 and Aβ42. Aβ42 is thought to be the major form that drives Aβ aggregation and plaque formation. Elevated levels of Aβ42 and an increased Aβ42/Aβ40 ratio are early events in AD pathology and the ratio may predict preclinical AD in cognitively normal patients [36]. Reports also showed that elevated serum Aβ42 levels increased the risk of AD [37]. These data suggested that Aβ42 could be a biomarker for AD diagnosis and the evaluation of treatment efficacy. The link between Aβ deposition, Aβ induced inflammation and AD provides a new therapeutic strategy for the treatment of patients suffering from the disease [38, 39].
Here we showed that high-dose RNSP reduced serum TNF-α, IL-1β, IL-6 and Aβ42 levels after treatment for 16 weeks. The Aβ42/Aβ40 ratio was decreased after 4 weeks of high-dose RNSP treatment and further decreased after 16 weeks of treatment. Statistical analyses showed a positive correlation between serum Aβ42 levels and TNFα, IL-1β and IL-6 levels. These results suggest that RNSP could inhibit inflammatory responses, reduce the serum levels of Aβ42, decrease the Aβ42/Aβ40 ratio and slow down the progression of AD. Furthermore, there was no significant adverse medication reaction observed in either RNSP treatment or control groups during the whole procedure, which suggested that RNSP (even at a high dose) is safe and well tolerated. All these results indicated that Tibetan Medicine RNSP could have great potentials for the treatment of AD. In this study, we found a positive relationship between the levels of pro-inflammatory cytokines and Aβ42 peptides and cognitive function scores. We aim to investigate further whether the changed levels of these cytokines and Aβ42 predict the progression of AD on long-term follow-up. At the same time, we plan to increase our sample size and observe whether RNSP can further improve AD patients’ cognitive function with an even longer period of treatment time, such as 6 months or 12 months. Additionally, we will investigate whether low-dose RNSP treatment for a longer period of time (6-12 months) can enhance AD patients’ cognitive scores. Our future experiments will provide more information on the efficacy and safety of RNSP. The further clinical trials of RNSP in AD will study at different altitudes.
5. Acknowledgements
We would like to thank Professors Edith and Patrick McGeer at the Kinsmen Laboratory of Neurological Research, University of British Columbia, for their editorial assistance and many useful suggestions and feedback to our manuscript. We also thank Dr. Jing Zhang for her valuable discussions and translating assistance. This work was supported by “Key Grant for Returnees on Science and Technology” from the Ministry of Personnel, China. (Ministry of Personnel [2005] Number: 129). The authors would like to thank the Institution of Geriatric for its support and all members in the Centre laboratory of Qinghai Provincial Hospital for helpful comments and feedback throughout this study. We would like to thank all the patients or their guardians for participating in this study.
REFERENCES
- Alzheimer’s Association, “2009 Alzheimer’s Disease Facts and Figures,” Alzheimers & Dementia, Vol. 5, No. 3, 2009, pp. 234-270. doi:10.1016/j.jalz.2009.03.001
- H. Brodaty, M. M. Breteler, S. T. Dekosky, et al., “The World of Dementia beyond 2020,” Journal of the American Geriatrics Society, Vol. 59, No. 5, 2011, pp. 923-927. doi:10.1111/j.1532-5415.2011.03365.x
- M. J. Dong, B. Peng, X. T. Lin, et al., “The Prevalence of Dementia in the People’s Republic of China: A Systematic Analysis of 1998-2004 Studies,” Age and Ageing, Vol. 36, No. 6, 2007, pp. 619-624. doi:10.1093/ageing/afm128
- D. Galimberti and E. Scarpini, “Treatment of Alzheimer’s Disease: Symptomatic and Disease-Modifying Approaches,” Current Aging Science, Vol. 3, No. 1, 2010, pp. 46-56. doi:10.2174/1874609811003010046
- E. G. McGeer and P. L. McGeer, “Inflammatory Processes in Alzheimer’s Disease,” Progress in Neuro-Psychopharmacology and Biological Psychiatry, Vol. 27, No. 5, 2003, pp. 741-749. doi:10.1016/S0278-5846(03)00124-6
- B. Ray and D. K. Lahiri, “Neuroinflammation in Alzheimer’s Disease: Different Molecular Targets and Potential Therapeutic Agents Including Curcumin,” Current Opinion in Pharmacology, Vol. 9, No. 4, 2009, pp. 434-444. doi:10.1016/j.coph.2009.06.012
- D. Ajit, M. L. Udan, G. Paranjape, et al., “Amyloid-Beta (1-42) Fibrillar Precursors Are Optimal for Inducing Tumor Necrosis Factor-Alpha Production in the THP-1 Human Monocytic Cell Line,” Biochemistry, Vol. 48, No. 38, 2009, pp. 9011-9021. doi:10.1021/bi9003777
- L. Ruan, Z. Kang, G. Pei, et al., “Amyloid Deposition and Inflammation in APPswe/PS1dE9 Mouse Model of Alzheimer’s Disease,” Current Alzheimer Research, Vol. 6, No. 6, 2009, pp. 531-540. doi:10.2174/156720509790147070
- ADAPT Research Group, “Alzheimer’s Disease Anti-Inflammatory Prevention Trial: Design, Methods, and Baseline Results,” Alzheimer’s and Dementia, Vol. 5, No. 2, 2009, pp. 93-104. doi:10.1016/j.jalz.2008.09.004
- R. Jesky and C. Hailong, “Are Herbal Compounds the Next Frontier for Alleviating Learning and Memory Impairments? An Integrative Look at Memory, Dementia and the Promising Therapeutics of Traditional Chinese Medicines,” Phytotherapy Research, Vol. 25, No. 8, 2011, pp. 1105-1118. doi:10.1002/ptr.3388
- A. Q. Zhu, Y. D. Chu, Q. X. Li, et al., “Tibet-medicine Effects on β-amyloid Pathology in a Transgenic Mouse Model of Alzheimer’s Disease,” Chinese Pharmacological Bulletin, Vol. 6, No. 25, 2009, pp. 720-724.
- A. Q. Zhu, C. L. Masters and Q. X. Li, “Tibet-Medicine Ratanasampil Modulates Amyloid Precursor Protein Cleavage and C-terminal Fragments (CTFS) in Tg2576 Transgenic Mice Brain of Alzheimer’s Disease,” Chinese Journal of Geriatrics, Vol. 11, No. 28, 2009, pp. 950-954.
- American Psychiatric Association, “Diagnostic and Statistical Manual of Mental Disorders,” 4th Edition, American Psychiatric Association Press, Washington DC, 1994.
- B. Dubois, H. H. Feldman, C. Jacova, et al., ”Research Criteria for the Diagnosis of Alzheimer s Disease: Revising the NINCDS-ADRDA Criteria,” Lancet Neurology, Vol. 6, No. 8, 2007, pp. 734-746. doi:10.1016/S1474-4422(07)70178-3
- W. G. Rosen, R. C. Mohs and K. L. Davis, “A New Rating Scale for Alzheimer’s Disease,” The American Journal of Psychiatry, Vol. 141, No. 11, 1984, pp. 1356-1364.
- M. F. Folstein, S. E. Folstein and P. R. McHugh, “‘Mini Mental State’: A Practical Method for Grading the Cognitive State of Patients for the Clinician,” Journal of Psychiatric Research, Vol. 12, No. 3, 1975.pp. 189-198. doi:10.1016/0022-3956(75)90026-6
- M. P. Lawton and E. M. Brody, “Assessment of Older People: Self-Maintaining and Instrumental Activities of Daily Living,” The Gerontologist, Vol. 9, No. 3, 1969, pp. 179-186. doi:10.1093/geront/9.3_Part_1.179
- M. J. Dong, B. Peng, X. T. Lin, et al., “The Prevalence of Dementia in the People’s Republic of China: A Systematic Analysis of 1980-2004 Studies,” Age and Ageing, Vol. 36, No. 6, 2007, pp. 619-624. doi:10.1093/ageing/afm128
- C. P. Ferri, M. Prince, C. Brayne, et al., “Alzheimer’s Disease International Global Prevalence of Dementia: A Delphi Consensus Study,” The Lancet, Vol. 366, No. 9503, 2005, pp. 2112-2117. doi:10.1016/S0140-6736(05)67889-0
- L. X. Yang, Y. D. Chu, A. Q. Zhu, et al., ”Sex Hormone and Blood Lipid Changes in the Female Alzheimer’s Patients Living at High Altitude,” Chinese Journal of Gerontology, Vol. 6, No. 21, 2001, pp. 459-460.
- A. Q. Zhu, C. Y. Song, L. X. Yang, et al., “EEG and CT Changes in Alzheimer’s Patients Living at High Altitude,” Clinical Focus, Vol. 22, No. 17, 2002, pp. 1313-1314.
- G. Gudmundsson and T. Gudbjartsson, “High Altitude Sickness—Review,” Laeknabladid, Vol. 95, No. 6, 2009, pp. 441-447.
- Q. X. Wu, L. Ji and J. L. Ku, “Analysis of Trace Elements and Macro Elements in 13 Anti-Anoxic Traditional Tibetan Medicine,” Spectroscopy and Spectral Analysis, Vol. 28, No. 8, 2008, pp. 1938-1941.
- E. J. An and Y. R. Suo, “Pharmacology Research on Tibetan Medicine Seventy-Taste-Pearl-Balls,” Journal of Medicine & Pharmacy of Chinese Minorities, Vol. 4, No. 2, 2004, pp. 33-35.
- S. H. L. Bayi and H. Hu, “Research Advances in Tibetan Medicine Seventy-Taste-Pearl-Balls,” Xinjiang Journal of Traditional Chinese Medicine, Vol. 5, No. 23, 2005, pp. 82-83.
- Y. X. Ling, Y. Q. Zheng and Z. H. Wang, “Effect of Huperzine a Combined with Ginaton on Alzheimer’s Disease,” Journal of Guangdong Medical College, Vol. 25, 2007, pp. 140-141.
- S. S. Shaftel, S. Kyrkanides and J. A. Olschowka, ”Sustained Hippocampal IL-1 Beta Overexpression Mediates Chronic Neuroinflammation and Ameliorates Alzheimer Plaque Pathology,” The Journal of Clinical Investigation, Vol. 117, No. 6, 2007, pp. 1595-1604. doi:10.1172/JCI31450
- Y. J. Lee, S. B. Han and S. Y. Nam, “Inflammation and Alzheimer’s Disease,” Archives of Pharmacal Research, Vol. 33, No. 10, 2010, pp. 1539-1556. doi:10.1007/s12272-010-1006-7
- P. Vural, S. Değirmencioğlu, H. Parildar-Karpuzoğlu, et al., “The Combinations of TNF Alpha-308 and IL-6-174 or IL-10-1082 Genes Polymorphisms Suggest an Association with Susceptibility to Sporadic Late-Onset Alzheimer’s Disease,” Acta Neurological Scandinavica, Vol. 120, No. 6, 2009, pp. 396-401. doi:10.1111/j.1600-0404.2009.01230.x
- E. Tobinick, “Tumour Necrosis Factor Modulation for Treatment of Alzheimer’s Disease: Rationale and Current Evidence,” CNS Drugs, Vol. 23, No. 9, 2009, pp. 713-725. doi:10.2165/11310810-000000000-00000
- Y. Shen, S. Q. Chen and Y. P. Wang, “Study on the Plasma Level and TNF-α-308G/A Polymorphism in Patients with Alzheimer’s Disease,” Journal of Tropical Medicine, Vol. 7, No. 2, 2007, pp. 133-136.
- D. Weisman, E. Hakimian and G. J. Ho, “Interleukins, Inflammation and Mechanisms of Alzheimer’s Disease,” Vitamins & Hormones, Vol. 74, 2006, pp. 505-530. doi:10.1016/S0083-6729(06)74020-1
- M. Morita, K. Osoda, M. Yamazaki, F. Shirai, et al., “Effects of Non-Steroidal Anti-Inflammatory Drugs on Abeta Deposition in Aβ1-42 Transgenic C. elegans,” Brain Research, Vol. 1295, No. 21, 2009, pp. 186-191. doi:10.1016/j.brainres.2009.08.002
- M. Hirohata, K. Ono and M. Yamada, “Non-Steroidal AntiInflammatory Drugs as Anti-Amyloidogenic Compounds,” Current Pharmaceutical Design, Vol. 14, No. 30, 2008, pp. 3280-3294. doi:10.2174/138161208786404173
- S. Funamoto, M. Morishima-Kawashima and Y. Tanimura, “Truncated Carboxyl-Terminal Fragments of β-Amyloid Precursor Protein are Processed to Amyloid Beta-Protein 40 and 42,” Biochemistry, Vol. 43, No. 42, 2004, pp. 13532-13540. doi:10.1021/bi049399k
- N. Schupf, M. X. Tang, H. Fukuyama, et al., “Peripheral Aβ Subspecies as Risk Biomarkers of Alzheimer’s Disease,” Proceedings of the National Acadmemy Science of USA, Vol. 105, No. 37, 2008, pp. 14052-14057. doi:10.1073/pnas.0805902105
- M. A. Findeis, “The Role of Amyloid Peptide 42 in Alzheimei’s Disease,” Pharmacology & Therapy, Vol. 116, No. 2, 2007, pp. 226-286.
- D. M. Barten and C. F. Albright, “Therapeutic Strategies for Alzheimer’s Disease,” Molecular and Neurobiology, Vol. 37, No. 2-3, 2008, pp. 171-186. doi:10.1007/s12035-008-8031-2
- P. A. Adlard, S. A. James, A. I. Bush, et al., “Beta-Amyloid as a Molecular Therapeutic Target in Alzheimer’s Disease,” Drugs Today, Vol. 45, No. 4, 2009, pp. 293- 304. doi:10.1358/dot.2009.045.004.1353853
NOTES
*Corresponding author.

