Pharmacology & Pharmacy
Vol.5 No.7(2014), Article
ID:46882,10
pages
DOI:10.4236/pp.2014.57078
Differential Effect on Two Immobility Responses by Chronic Administration of 1,3-di-o-Tolyl-Guanidine (Sigma Receptor Agonist) in Rats with Neonatal Ventral Hippocampal Lesion
Blanca Estela Jaramillo1,2, Fidel De la Cruz1, Genaro Vargas3, Oscar O. Morales1, Gonzalo Flores4, Sergio R. Zamudio1, Linda Garcés-Ramírez1
1Departamento de Fisiología, Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional, México D. F., México
2Programa Educativo de la Licenciatura en Terapia Física, Universidad Politécnica de Pachuca, Zempoala, Hidalgo, México
3Programa Educativo de la Ingeniería en Biotecnología, Universidad Politécnica de Pachuca, Zempoala, Hidalgo, México
4Instituto de Fisiología, Benemérita Universidad Autónoma de Puebla, Puebla, México
Email: adnil_gr@yahoo.com.mx, flacruz90@hotmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 30 April 2014; revised 30 May 2014; accepted 12 June 2014
ABSTRACT
The effect of chronic administration of the sigma agonist: 1,3-di-o-tolyl-guanidine (DTG), in neonatal ventral hippocampal lesioned (nVHL) rats, on the immobility by clamping and dorsal immobility, were investigated. The nVHL increases the duration of immobility by clamping, but does not affect the duration of the dorsal immobility. We found that DTG augments the duration of the dorsal immobility in the unlesioned rats, but does not modify the duration of immobility induced by clamping the neck. DTG also counteracts the increase in the duration of the immobility by clamping produced by nVHL. However, the increase in the duration of the dorsal immobility produced by DTG is counteracted by nVHL. These results are discussed with respect to the differential effect on the two immobility responses tested, suggesting that they are different forms of immobility mediated by different mechanisms although they behaviorally share common characteristics.
Keywords:Immobility Response, Sigma Receptor, DTG, Schizophrenia, Neonatal Ventral Hippocampal Lesion

1. Introduction
Immobility response is expressed in a wide variety of circumstances, which share some neural mechanisms and may occur in response to natural stimuli, by the action of some drugs or in certain diseases of the nervous system. It is a state of complete absence of movements and relative unresponsiveness, which can be triggered in a variety of species by several different kinds of stimuli. Various names have been used to describe this phenomenon: animal hypnosis, death feigning, tonic immobility, akinesia, catalepsy, cataplexy, catatonia etc. Death feigning is a form of immobility described by centuries in which the animal remains completely still in the face of a threat. “Play dead” is one of the oldest defense mechanisms of many species, including our own, and it occurs in practically all the phylogenetic scale. It is characterized by restraint and reduced capacity to respond to external stimuli, with a variable duration depending on the species. The animal can fight or try to escape, but can also opt for passive defense, adopting a motionless posture, which may persist from a few seconds to several hours. Some features include temporary immobility response [1] -[3] , defecation, muscle rigidity and tremors of the extremities, abnormal EEG patterns, changes in heart rate, breathing and core temperature [4] . This behavior can also be induced by pharmacological means. Morphine produces a form of immobility known as catatonia, in which the righting reflexes and stiffness are inhibited [2] . Another form of immobility is catalepsy, where righting reflex (used as an indicator of some antipsychotic drugs such as haloperidol) is preserved [2] . These neuroleptic action is related to the capacity of the drug to block dopamine receptors. It has been stated that such drugs act in the striatum and nucleus accumbens [5] . Also the electrolytic lesion of the caudate-putamen and accumbens reduced haloperidol-induced catalepsy. It has been proposed that the basal nuclei may interact through cholinergic pathways with systems such as the prefrontal system. Many experiments have produced immobility responses by damaging certain regions of the brain that control postural reflexes and locomotion, such as the lateral hypothalamus [6] . As well, mesencephalic lesioned rats have also been tested and shown an increase in different ways of immobility [7] . The so-called catatonia of schizophrenics is related to some hyperactivity in the dopaminergic system, since it is highly susceptible to neuroleptic drug treatment. An increase in dopaminergic activity has also been implicated in opiate-induced catalepsy. In addition, certain forms of immobility are presented in some psychiatric disorders such as catatonic schizophrenia. This disorder is characterized by physical immobility experienced by the person when he is in catatonic seizure and is unable to respond to external stimuli. Other forms of immobility appear in certain diseases of the central nervous system that cause people to disconnect from the world.
Schizophrenia is one of the major psychiatric disorders that is characterized by positive symptoms (e.g., hallucinations and delusions), negative symptoms (e.g. affective flattening, alogia, or abolition) and cognitive impairments (e.g. severe deterioration of concentration and memory). It has been reported that approximately 1.1% of the population aged 18 years and older develops schizophrenia (DSM-IV classification) [8] . Although schizophrenia itself has been recognized for centuries, pharmaceutical treatment only began in the 1950s, classical antipsychotics are still widely active used in the treatment of schizophrenia. Nevertheless, they induce severe extrapyramidal side-effects. That is why many researchers have been focused on the development of new agents with low extrapyramidal side-effects. The neonatal ventral hippocampal lesion (nVHL) has been proposed as a heuristic neurodevelopmental model of schizophrenia-related behaviors in the rat. These behaviors include hyperresponsiveness to novel environment and psychostimulant administration [9] -[11] , deficits in sensory gating [12] and working memory impairments [13] , which appear only after puberty. Additionally, nVHL induces neuronal rearrangements [14] [15] , disrupted cholinergic and dopaminergic (DA) transmission [16] [17] , as observed in schizophrenic patients.
The possible involvement of sigma receptors (σ receptors) in schizophrenia has been suggested, based on the psychotomimetic actions of σ receptor agonists [18] , the density of σ receptors detected in various mesencephalic and limbic areas [19] [20] , and the action of several receptor ligands on dopaminergic neurons [21] -[24] . In fact, there are reports showing a reduction of sigma-1 receptors in the brains of schizophrenic patients postmortem [19] [20] [25] and other link between sigma receptors and schizophrenia, is that the typical and extensively used antipsychotic haloperidol has a very high affinity for sigma-1 receptors [26] [27] . Interestingly, it is also reported that sigma-1 receptors are engaged in modulating NMDA-type glutamate receptors, [28] [29] which might be involved in the pathophysiology of schizophrenia [30] -[33] .
The sigma receptors are chaperon proteins that were originally proposed as a subtype of opioid receptors (σ opioid receptors) and were suggested to contribute to delusion and psychosis induced by benzomorphans such as SKF-10047 and pentazocine [34] . However, later studies confirmed that σ receptors are nonopioid receptors, and they are now called σ receptors instead of σ opioid receptors to distinguish the protein from opioid receptors [26] [35] . Sigma receptors consist of at least two subtypes, σ-1 and σ-2 receptors [28] [36] .
Sigma-1 receptors have been cloned [37] and their biological and physiological roles have been more intensively examined; σ-2 receptors neither have been well studied or cloned. The σ-1 receptor gene encodes 25 kDa membrane proteins that have no homology to any mammalian protein [37] . Sigma-1 receptors are intracellular receptors mainly localized on the endoplasmic reticulum, and they dynamically translocate inside cells [38] [39] . Sigma-1 receptors are expressed in specific regions of the brain such as layers of the cortex, hippocampus, hypothalamic nuclei, substantia nigra and Purkinje cells in the cerebellum [28] [40] . Occupancy of sigma-1 receptors by agonists causes translocation of the receptor protein from the endoplasmatic reticulum to the cell membrane where the receptor can regulate ion channels and neurotransmitter release [38] [41] . The sigma-1 receptor is implicated in cellular differentiation [42] [43] , neuroplasticity [44] [45] , neuroprotection [46] [47] , and cognitive functioning of the brain [48] . Such neuroprotection and neurite outgrowth are thought to contribute to the improvement of various neuropsychiatric diseases like the schizophrenia.
To our knowledge, there are no studies trying to correlate the immobility, schizophrenia and sigma agonists. Therefore, we evaluated the effect of sub chronic administration of sigma receptor agonist 1,3-di-o-tolyl-guanidine (DTG) on two immobility responses in neonatal ventral hippocampal lesion rats as a neurodevelopmental model of schizophrenia
2. Materials and Methods
2.1. Animals
Thirty-two Sprague-Dawley male rats were provided by Institute of Healthy Science (ICSA) of Autonomous University of Hidalgo, México All animal procedures were done in agreement with the “Guide for Care and Use of Laboratory Animals” of the Mexican Council for Animal Care (Norma Oficial Mexicana NOM-062-ZOO- 1999) and the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Rats were housed in a temperatureand humidity-controlled room maintained on a 12-hour light/dark cycle (lights on: 07:00 - 19:00 h) and had free access to food and water. Experiments were conducted during the light phase (between 09:00 and 12:00 h).
2.2. Neonatal Ventral Hippocampal Lesion
On postnatal day 7 (PD7), male pups (weighing 15 - 18 g) were randomly assigned to either lesion or false lesion (sham) group. Pups were anesthetized by hypothermia and were placed on ice for 18 - 20 min and immobilized on a modified platform [49] fixed to a stereotaxic frame. An incision in the skin overlaying the skull was made and two 1 mm holes were drilled. A needle connected to an infusion pump through a Hamilton syringe was lowered into each ventral hippocampal coordinate: AP −3.0 mm, ML ±3.5 relative to bregma and VD −4.9 relative to dura. Ibotenic acid (0.3 µL [10 mg/mL]; Sigma, St Louis, MO, USA) in 0.15 M phosphate buffer saline (PBS) pH 7.4 or vehicle was infused bilaterally at a flow rate of 0.15 µL/min. After the procedure, the pups were placed under a heating lamp and then returned to their mothers (the pups were handled with gloves to avoid odors). On PD21, the animals were weaned and similar number of sham and lesioned rats was placed in cages (four animals per cage).
2.3. Drug Administration
Four groups were formed (8 animals per group): 1) vehicle-treated sham, 2) DTG-treated sham, 3) vehicle-treated nVHL, and 4) DTG-treated nVHL. The rats were administered with one dose diary during 27 days, either of the sigma agonist 1,3-di-o-tolyl-guanidine (DTG; Tocris Biosciences) 10 mg/kg (i.p.), [50] or phosphate buffer saline (vehicle).
2.4. Behavioral Tests
The behavioral studies were performed 1 day after finishing the DTG or vehicle administration. The animals were tested and videotaped; the behavioral responses were analyzed by a person blind to the experimental groups to avoid an unconscious predisposition of the results.
2.4.1. Immobility Induced by Clamping
It was induced by clamping the neck (IC). A clamp was applied (the clamp was 3.2 cm long (IDL, No. 10050, Carlstide, NJ)) between the base of the skull and the back of the ears so that the pressure was sufficient to lift the animal by the clamp. With this pressure, the animals did not vocalize, attempt to scratch at, or pull off the clamps. The rat was then placed on its back on a flat surface [3] . The behavioral sequence starting from the moment the animal was place on its back until it regained the full prone position with the palm of all 4 limbs contacting the ground. The duration of immobility was measured and the observations were terminated at 240 s even if the whole sequence had not been completed by that time [2] .
2.4.2. Dorsal Immobility
The animal was gently grasped by the skin at the back of the neck and lifted off its feet with no part of the animal’s body touching any other surface. The duration of this immobility response was measured from the onset of the response until the animal made escape-like movements or directed at the experimenter’s hand [3] .
2.5. nVHL Lesion Assessment
For assessment of lesion size, the brains fixed in formaldehyde were sectioned into 15 µm thick slices on the coronal plane using a vibrotome (Campden Instrument, MA752, Leicester, UK). Sections at the level of the ventral hippocampus were collected on cleaned, gelatin-coated microscope slides (four sections/slides). Sections were stained with 0.5% cresyl violet and examined under a microscope where the lesions and probe placement could be seen.
2.6. Data Analysis
The immobility data were analyzed by two-way ANOVA test followed by the Newman-Keuls test for post hoc comparisons, with lesion and treatment as independent factors (P < 0.05 as statistically significant). Sigma Stat: Systat Software, Inc.
3. Results
3.1. Verification of the Lesion
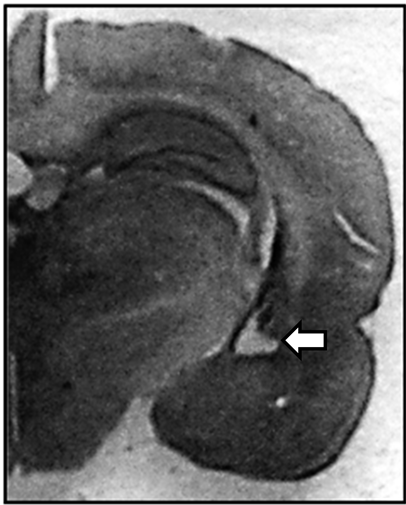
Cresyl violet-stained sections obtained from adult nVHL-lesioned animals revealed significant bilateral damage of the ventral hippocampus with neuronal loss, atrophy and apparent retraction of the hippocampal formation (Figure 1) as reported in earlier studies [9] [15] [51] [52] . A cavity resulting from the lesion was frequently observed. The brains of the sham animals did not show any morphological alterations.
3.2. Immobility Responses
The effects of repeated DTG administration in nVH lesioned rats on the immobility response (IR) are illustrated in Figure 2. Both groups, sham and nVHL animals exhibited the IR. The duration time of immobility caused by clamping the neck (IC) was significant increased by the nVH lesion (Figure 2(a)) (Two way ANOVA interaction between lesion and treatment F(1,28) = 17.279, P < 0.001), this behavior was reversed by repeated DTG administration (P < 0.01), whereas no differences in the IC response were seen between sham vehicle or DTG groups (F(1,28) = 1.49; P > 0.05, two-way ANOVA, Student-Newman-Keuls post-test). The duration of the IR by gently grasping the skin at the back of the neck, dorsal immobility (ID), is illustrated in Figure 2(b). A twoway ANOVA showed that the ID was not significantly affected by lesion, the post-hoc tests did not shown a difference between sham rats and those with lesion. However, nVHL rats treated with DTG shown a diminution ID duration than nVHL rats with vehicle (interaction between lesion and treatment F(1,28) = 9.960, P < 0.01 two way ANOVA, Student-Newman-Keuls post-test) while sham rats treated with DTG increase the duration of the ID (comparisons for factor: lesion within DTG treatment, P < 0.01).
4. Discussion
The results show that the nVHL increases the duration of immobility by clamping, but does not affect the duration of the dorsal immobility. DTG in the unlesioned rats augments the duration of the dorsal immobility but
 (a)
(a)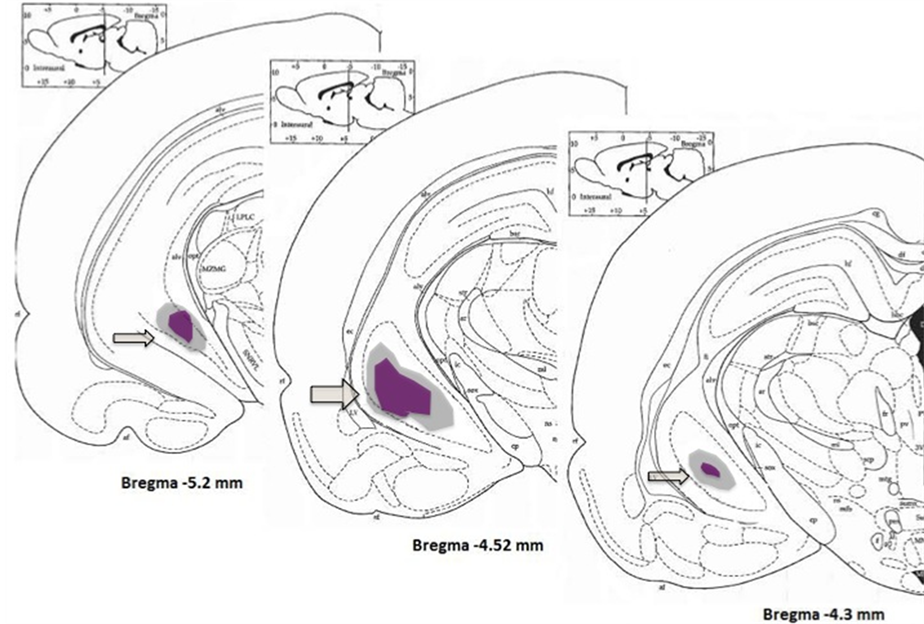 (b)
(b)
Figure 1. Photograph of a coronal section of the ventral hippocampus. (a) Slices were stained with cresyl violet showing retraction of the tissue and neural loss from nVHL rats at postpubertal age. The arrow indicates the zone of the lesion; (b) Schematic drawing of coronal sections illustrating areas of least and greatest VH lesion as determined by Nissl-stained sections of the hippocampus of animals with nVHL at a postpubertal age. Gray, reconstruction of the neuronal loss and gliosis in the hippocampus of the rat with the most widespread lesion. Purple, extent of the lesion in the rat with the minimal lesion considered significant. Numbers indicate distance (mm) posterior from bregma according to [53] .
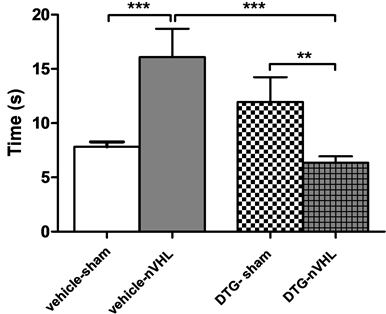 (a)
(a) (b)
(b)
Figure 2. Effects of DTG administration on the duration of the IRs. (a) Immobility by clamping; (b) The dorsal immobility. Values are means + SEM in seconds (n = 8 animals per group), by two-way ANOVA, StudentNewman-Keuls post-tests *P < 0.05, **P < 0.01, ***P < 0.001.
does not modify the duration of immobility by clamping. We also found that DTG counteracts the increase in the duration of the immobility by clamping produced by nVHL. In addition, the increase in the duration of the dorsal immobility produced by DTG is counteracted by nVHL. This differential effect on the two immobility responses tested, suggests that they are mediated by different mechanisms. The differential effect on the two immobility responses in the rat, obtained by the neonatal hippocampal lesion as well as by the sigma agonist DTG, raises the question: are there multiple immobility responses, or is there one system that can be activated in multiple ways? We think that it is most likely to be multiple immobility responses; one clue supporting this view is that an immobility state can be elicited with several kinds of stimuli. In rodents, immobility states can be induced in pups [1] ; during pinch-induced catalepsy in mice [54] ; and during the suppression of wet dogs by tail and scruff pinch [55] . In all these behaviors, the neck is the region that receives the greatest amount of stimulation. It is not surprising then to find that for neck region there is a somatotopic mapping in the dorsal central gray linked to analgesia [56] . So it was not surprising to find that in the mouse, analgesia is linked to the immobility by clamping [57] . In a previous study [1] , we found that either clamping or bandaging the neck region can induce immobility in neonate rats, whereas in adult rats, only clamping can induce it. An immobility state can be elicited in a variety of species by several kinds of stimuli and by several kinds of drugs. Morphine and haloperidol produce in the rat two distinct and contrasting immobility states: the former produces a state characterized by inhibition of the postural support, while the latter produces a state in which tonic reactions subserving the maintenance of stable static equilibrium prevail at the expense of phasic-locomotor reactions [58] . The immobility induced by morphine is characterized by inhibition of righting reactions [4] . Endorphinergic system has been found to interact with dopaminergic systems [59] . The fact that states of experimentally dopaminergic deficiency are accompanied by catalepsy, which is a form of immobility somewhat different than opiate-induced catalepsy, suggested that these two states of immobility might be linked to a change of dopaminergic activity; some findings have supported this assumption. Thus, the destruction of the neoor palio-striatal dopaminergic systems largely abolished the immobility induced by neuroleptics, but potentiated the induced by morphine [59] . The so-called catatonia of schizophrenics is probably related to some hyperactivity in the dopaminergic system, since it is highly susceptible to neuroleptic drug treatment.
It was demonstrated that haloperidol which is a D2 antagonist and the main sigma 1 receptor antagonist [2] increases the duration of immobility response elicited by clamping the neck. Haloperidol is still widely used for the treatment of schizophrenia. Interestingly, in this study, we observed that DTG reverts the increment in duration of IC due to nVHL. These findings suggest that dopaminergic and sigma receptor pathways could modulate immobility response elicited by clamping. However, in dorsal immobility, the sham rats treated with DTG increased significantly their time of immobility and DTG reduces duration of this immobility response, it is known that these two immobility responses have differences and they cannot be following the same modulation [60] . These results show that DTG by itself has an effect on dorsal immobility, suggesting that this behavior is more sensitive to sigma receptor agonist. So far, there had been no a study that correlates neither the immobility response elicited by clamping the neck nor dorsal immobility with sigma receptors. Nevertheless, there are various studies about sigma receptors and depression where they have seen that sigma receptor agonists reduce the time of immobility in forced swimming test; it demonstrates that sigma receptors are involved in different forms of immobility [61] .
5. Conclusion
Since the sigma agonist (DTG)) and the neonatal ventral hippocampal lesion are involved in a differential and opposite way on the two forms of immobility that we tested (immobility by clamping the neck and dorsal immobility), it suggests that they are mediated by different mechanisms, although behaviorally share common characteristics. So, we believe that this evidence could help us to link, two known phenomena—schizophrenia and immobility response, which may be related or somehow present in catatonic schizophrenia.
Acknowledgements
The authors would like to thank to Dr. Gianluigi Tanda and Dr. Jonathan L. Katz for giving us the sigma receptor agonist (DTG) and the staff of bioterio ICSA-UAEH for animal housing facilities, QFB Alvaro Ceron, M. V. Z. Héctor Hernández and M. V. Z. Alejandro Manzano.
References
- De La Cruz, F., Junquera, J. and Russek, M. (1987) Ontogeny of Immobility Reactions Elicited by Clamping, Bandaging and Maternal Transports in Rats. Experimental Neurology, 97, 315-326.http://dx.doi.org/10.1016/0014-4886(87)90092-6
- De La Cruz, F., Russek, M. and Junquera, J. (1990) Ontogeny of the Endorphinergic and Dopaminergic Modulation on the Immobility Reflex Elicited by Clamping in Rats. International Journal of Psychophysiology, 9, 171-177.http://dx.doi.org/10.1016/0167-8760(90)90071-K
- De La Cruz, F. and Uriostegui, T. (1996) The Differential Haloperidol Effect on the Immobility Response Elicited by Clamping, Grasping, Bandaging and Inversión in Guinea Pig, Hamster and Rat. Behavioural Brain Research, 78, 195-199. http://dx.doi.org/10.1016/0166-4328(95)00249-9
- Klemm, W.R. (1971) Neurophysiological Studies of the Immobility Reflex (Animal Hypnosis). In: Ehrenpreis, S. and Stalnisky, O.C., Eds., Neurosciences Research, Academic, New York, 165-210.http://dx.doi.org/10.1016/B978-0-12-512504-8.50011-1
- Rowlet, J.K., Pedigo, N.W. and Bardo, M.T. (1991) Catalepsy Produced by Striatal Microinjections of the D1 Dopamine Receptor Agonist SCH 23390 in Neonatal Rats. Pharmacology Biochemistry and Behavior, 40, 829-834.http://dx.doi.org/10.1016/0091-3057(91)90093-H
- Teitelbaum, P., Wolgin, D.L., De Ryck, M. and Marin, O.S.M. (1976) Bandage Backfall Reaction: Occurs in Infancy, Hypothalamic Damage, and Catalepsy. Proceeding of the National Academic of Sciences of the USA, 73, 3311-3314.http://dx.doi.org/10.1073/pnas.73.9.3311
- De la Cruz, F., Uriostegui, T., Zamudio, S., Pacheco, J., Quevedo, L. and Chuc, E. (1995) Potentiation of the Imobility Response Elicited by Bandaging and Clamping in Mesencephalic Rats. Physiology & Behavior, 58, 737-742.http://dx.doi.org/10.1016/0031-9384(95)00123-Z
- Regier, D.A., Narrow, W.E., Rae, D.S., Manderscheid, R.W., Locke, B.Z. and Goodwin, F.K. (1993) The de Facto US Mental and Addictive Disorders Service System. Epidemiologic Catchment Area Prospective 1-Year Prevalence Rates of Disorders and Services. Archives of General Psychiatry, 50, 85-94.http://dx.doi.org/10.1001/archpsyc.1993.01820140007001
- Flores, G., Barbeau, D., Quirion, R. and Srivastava, L.K. (1996) Decreased Binding of Dopamine D3 Receptors in Limbic Subregions after Neonatal Bilateral Lesion of Rat Hippocampus. The Journal of Neuroscience, 16, 2020-2026.
- Lipska, B.K. and Weinberger, D.R. (2000) To Model a Psychiatric Disorder in Animals: Schizophrenia as a Reality Test. Neuropsychopharmacology, 23, 223-239. http://dx.doi.org/10.1016/S0893-133X(00)00137-8
- Lipska, B.K., Jaskiw, G.E. and Weinberger, D.R. (1993) Postpubertal Emergence of Hyperresponsiveness to Stress and to Amphetamine after Neonatal Excitotoxichippocampal Damage: A Potential Animal Model of Schizophrenia. Neuropsychopharmacology, 9, 67-75. http://dx.doi.org/10.1038/npp.1993.44
- Sams-Dodd, F., Lipska, B.K. and Weinberger, D.R. (1997) Neonatal Lesions of the Rat Ventral Hippocampus Result in Hyperlocomotion and Deficits in Social Behavior in Adulthood. Psychopharmacology (Berl), 132, 303-310.http://dx.doi.org/10.1007/s002130050349
- Chambers, R.A., Moore, J., McEvoy, J.P. and Levin, E.D. (1996) Cognitive Effects of Neonatal Hippocampal Lesions in a Rat Model of Schizophrenia. Neuropsychopharmacology, 15, 587-594.http://dx.doi.org/10.1016/S0893-133X(96)00132-7
- Alquicer, G., Morales-Medina, J.C., Quirion, R. and Flores, G. (2008) Postweaning Social Isolation Enhances Morphological Changes in the Neonatal Ventral Hippocampal Lesion Rat Model of Psychosis. Journal of Chemical Neuroanatomy, 35, 179-187. http://dx.doi.org/10.1016/j.jchemneu.2007.10.001
- Flores, G., Silva-Gómez, A.B., Barbeau, D., Srivastava, L.K., Zamudio, S.R. and De La Cruz, F. (2005) Effect of Excitotoxic Lesions of the Neonatal Ventral Hippocampus on the Immobility Response in Rats. Life Sciences, 76, 2339-2348. http://dx.doi.org/10.1016/j.lfs.2004.11.007
- Laplante, F., Stevenson, C.W., Gratton, A., Srivastava, L.K. and Quirion, R. (2004) Effects of Neonatal Ventral Hippocampal Lesion in Rats on Stress-Induced Acetylcholine Release in the Prefrontal Cortex. Journal of Neurochemistry, 91, 1473-1482. http://dx.doi.org/10.1111/j.1471-4159.2004.02831.x
- Laplante, F., Nakagawasai, O., Srivastava, L.K. and Quirion, R. (2005) Alterations in Behavioral Responses to a Cholinergic Agonist in Post-Pubertal Rats with Neonatal Ventral Hippocampal Lesions: Relationship to Changes in Muscarinic Receptor Levels. Neuropsychopharmacology, 30, 1076-1087. http://dx.doi.org/10.1038/sj.npp.1300640
- Tam, S.W. (1983) Naloxone-Inaccessible Sigma Receptor in Rat Central Nervous System. Proceedings of the National Academy of Sciences of the United States of America, 80, 6703-6707. http://dx.doi.org/10.1073/pnas.80.21.6703
- Weissman, A.D., Casanove, M.F., Kleinman, J.E., London, E.D. and de Souza, E.B. (1991) Selective Loss of Cerebral Cortical Sigma, but Not PCP Binding Sites in Schizophrenia. Biological Psychiatry, 29, 41-54. http://dx.doi.org/10.1016/0006-3223(91)90209-5
- Weissman, A.D., Su, T.P., Hedreen, J.C. and London, E.D. (1988) Sigma Receptors in Post-Mortem Human Brains. Journal of Pharmacology and Experimental Therapeutics, 247, 29-33.
- Steinfels, G.F. and Tam, S.W. (1989) Selective Sigma Receptor agonist and Antagonist Affect Dopamine Neuronal Activity. European Journal of Pharmacology, 163, 167-170. http://dx.doi.org/10.1016/0014-2999(89)90413-5
- Zhang, J., Chiodo, L.A., Wettstein, J.G., Junien, J.L. and Freeman, A.S. (1993) Repeated Administration of Sigma Ligands Alters the Population Activity of Rat Midbrain Dopaminergic Neurons. Synapse, 13, 223-230. http://dx.doi.org/10.1002/syn.890130305
- Moison, D., De Deurwaerdère, P., Cagnotto, A., Marrazzo, A., Prezzavento, O., Ronsisvalle, G., et al. (2003) Intrastriatal Administration of Sigma Ligands Inhibits Basal Dopamine Release in Vivo. Neuropharmacology, 45, 945-953. http://dx.doi.org/10.1016/S0028-3908(03)00253-3
- Garcés-Ramírez, L., Green, J.L., Hiranita, T., Kopajtic, T.A., Mereu, M., Thomas, A.M., Mesangeau, C., Narayanan, S., McCurdy, C.R., Katz, J.L. and Tanda, G. (2011) Sigma Receptor Agonists: Receptor Binding and Effects on Mesolimbic Dopamine Neurotransmission Assessed by Microdialysis. Biological Psychiatry, 69, 208-217.
- Shibuya, H., Mori, H. and Toru, M. (1992) Sigma Receptors in Schizophrenic Cerebral Cortices. Neurochemical Research, 17, 983-990. http://dx.doi.org/10.1007/BF00966825
- Su, T.P. (1982) Evidence for Sigma Opioid Receptor: Binding of [3H]SKF-10047 to Etorphine-Inaccesible Sites in Guinea-Pig Brain. Journal of Pharmacology and Experimental Therapeutics, 223, 284-290.
- Tam, S.W. and Cook, L. (1984) Sigma Opiates and Certain Antipsychotic Drugs Mutually Inhibit (+)-[3H] SKF 10,047 and [3H]Haloperidol Binding in Guinea Pig Brain Membranes. Proceedings of the National Academy of Sciences of the United States of America, 81, 5618-5621. http://dx.doi.org/10.1073/pnas.81.17.5618
- Kitaichi, K., Chabot, J.G., Moebius, F.F., Flandorfer, A., Glossmann, H. and Quirion, R. (2000) Expression of the Purported Sigma1 (1) Receptor in the Mammalian Brain and Its Possible Relevance in Deficits Induced by Antagonism of the NMDA Receptor Complex as Revealed Using an Antisense Strategy. Journal of Chemical Neuroanatomy, 20, 375-387.
- Shimazu, S., Katsuki, H., Takenaka, C., Tomita, M., Kume, T., Kaneko, S., et al. (2000) Sigma Receptor Ligands Attenuate N-Methyl-D-Aspartate Cytotoxicity in Dopaminergic Neurons of Mesencephalic Slice Cultures. European Journal of Pharmacology, 388, 139-146. http://dx.doi.org/10.1016/S0014-2999(99)00852-3
- Goff, D.C. and Coyle, J.T. (2001) The Emerging Role of Glutamate in the Pathophysiology and Treatment of Schizophrenia. The American Journal of Psychiatry, 158, 1367-1377. http://dx.doi.org/10.1176/appi.ajp.158.9.1367
- Hashimoto, K., Fukushima, T., Shimizu, E., Komatsu, N., Watanabe, H., Shinoda, N., et al. (2003) Decreased Serum Levels of D-Serine in Patients with Schizophrenia: Evidence in Support of the N-Methyl-D-Aspartate Receptor Hypofunction Hypothesis of Schizophrenia. Archives of General Psychiatry, 60, 572-576. http://dx.doi.org/10.1001/archpsyc.60.6.572
- Hashimoto, K., Okamura, N., Shimizu, E. and Iyo, M. (2004) Glutamate Hypothesis of Schizophrenia and Approach for Possible Therapeutic Drugs. Current Medicinal Chemistry, Central Nervous System Agents, 4, 147-154.
- Hashimoto, K. (2006) The NMDA Receptor Hypofunction Hypothesis for Schizophrenia and Glycinemodulatory Sites on the NMDA Receptors as Potential Therapeutic Drugs. Clinical Psychopharmacology and Neuroscience, 4, 3-10.
- Martín, W.R., Eades, C.G. and Thompson, J.A. (1976) The Effects of Morphine and Nalorphine-Like Drugs in Nondependent and Morphine Dependent Chronic Spinal Dog. Journal of Pharmacology and Experimental Therapeutics, 197, 517-535.
- Quirion, R., Bowen, W.D., Itzhak, Y., Junien, J.L., Musacchio, J.M., Rothman, R.B., et al. (1992) A Proposal for the Classification of Sigma Binding Sites. Trends in Pharmacological Sciences, 13, 85-86. http://dx.doi.org/10.1016/0165-6147(92)90030-A
- Bowen, W.D., Hellewell, S.B. and McGarry, K.A. (1989) Evidence for a Multi-Site Model of the Rat Brain Sigma Receptor. European Journal of Pharmacology, 163, 309-318. http://dx.doi.org/10.1016/0014-2999(89)90200-8
- Hanner, M., Moebius, F., Flandorfer, A., Knaus, H.G., Striessnig, J., Kempner, E., et al. (1996) Purification, Molecular Cloning and Expression of the Mammalian sigma1-Binding Site. Proceedings of the National Academy of Sciences of the United States of America, 93, 8072-8077. http://dx.doi.org/10.1073/pnas.93.15.8072
- Hayashi, T. and Su, T.P. (2003) Intracellular Dynamics of sigma-1 Receptors (sigma1 Binding Sites) in NG108-15 Cells. Journal of Pharmacology and Experimental Therapeutics, 306, 726-733. http://dx.doi.org/10.1124/jpet.103.051292
- Hayashi, T. and Su, T.P. (2003) Sigma-1 Receptors (sigma1 Binding Sites) Form Raft-Like Microdomains and Target Lipid Droplets on the Endoplasmic Reticulum: Roles in Endoplasmic Reticulum Lipid Compartmentalization and Export. Journal of Pharmacology and Experimental Therapeutics, 306, 718-725. http://dx.doi.org/10.1124/jpet.103.051284
- Heroux, J.A., Tam, S.W. and De Souza, E.B. (1992) Autoradiographic Identification and Characterization of Sigma Receptors in Guinea Pig Brain Using [3H]1(Cyclopropylmethyl)-4-(2’-(4≈-Fluorophenyl)-2’-Oxoethyl) Piperidine ([3H]DuP 734), a Novel Sigma Receptor Ligand. Brain Research, 598, 76-86. http://dx.doi.org/10.1016/0006-8993(92)90170-E
- Hayashi, T. and Su, T.P. (2007) Sigma-1 Receptor Chaperones at the ER-Mitochondrion Interface Regulate Ca2+ Signaling and Cell Survival. Cell, 131, 596-610. http://dx.doi.org/10.1016/j.cell.2007.08.036
- Hayashi, T. and Su, T.P. (2004) Sigma-1 Receptors at Galactosylceramide-Enriched Lipid Microdomains Regulatesoligodendrocyte Differentiation. Proceedings of the National Academy of Sciences of the United States of America, 101, 14949-14954. http://dx.doi.org/10.1073/pnas.0402890101
- Hayashi, T. and Su, T.P. (2008) An Update on the Development of Drugs for Neuropsychiatric Disorders: Focusing on the Sigma(1) Receptor Ligand. Expert Opinion on Therapeutic Targets, 12, 45-58. http://dx.doi.org/10.1517/14728222.12.1.45
- Takebayashi, M., Hayashi, T. and Su, T.P. (2004) A Perspective on the New Mechanism of Antidepressants: Neuritogenesis through Sigma-1 Receptors. Pharmacopsychiatry, 37, 208-213. http://dx.doi.org/10.1055/s-2004-832679
- Tsai, Y.K., Hayashi, T., Harvey, B.K., Wang, Y., Su, P.T., et al. (2009) Sigma-1 Receptors Regulate Hippocampaldendritic Spine Formation via a Free Radical-Sensitive Mechanism Involving Rac1 GTP Pathway. Proceedings of the National Academy of Sciences of the United States of America, 106, 22468-22473. http://dx.doi.org/10.1073/pnas.0909089106
- Maurice, T. and Lockhart, B.P. (1997) Neuroprotective and Anti-Amnesic Potentials of Sigma Receptor Ligands. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 21, 69-102. http://dx.doi.org/10.1016/S0278-5846(96)00160-1
- Marrazzo, A., Caraci, F., Salinaro, E.T., Su, T.P., Copani, A. and Ronsisvalle, G. (2005) Neuroprotective Effects of sigma-1 Receptor Agonists against Beta-Amyloid-Induced Toxicity. Neuroreport, 16, 1223-1226. http://dx.doi.org/10.1097/00001756-200508010-00018
- Maurice, T. (2007) Cognitive Effects of Sigma-Receptor Ligands. In: Matsumoto, R.R., Bowen, W.D. and Su, T.P., Eds., Sigma Receptors: Chemistry, Cell Biology and Clinical Implications, Chap. 12, Springer, New York, 237-271. http://dx.doi.org/10.1007/978-0-387-36514-5_12
- Sierra, A., Camacho-Abrego, I., Escamilla, C., Negrete-Diaz, J.V., Rodriguez-Sosa, L. and Flores, G. (2009) Economical Body Platform for Neonatal Rats Stereotaxic Surgery. Revista de Neurología, 48, 141-146.
- Weber, E., Sonders, M., Quarum, M., McLean, S., Pou, S. and Keana, J.F. (1986) 1,3-Di(2-tolyl)guanidine: A Selective Ligand That Labels Sigma-Type Receptors for Psychotomimetic Opiates and Antipsychotic Drugs. Proceedings of the National Academy of Sciences of the United States of America, 83, 8784-8788. http://dx.doi.org/10.1073/pnas.83.22.8784
- Brake, W.G., Sullivan, R.M., Flores, G., Srivastava, L.K. and Gratton, A. (1999) Neonatal Ventral Hippocampal Lesions Attenuate the Nucleus Accumbens Dopamine Response to Stress: An Electrochemical Study in the Adult Rat. Brain Research, 831, 25-32. http://dx.doi.org/10.1016/S0006-8993(99)01477-8
- Silva-Gomez, A.B., Bermudez, M., Quirion, R., Srivastava, L.K., Picazo, O. and Flores, G. (2003) Comparative Behavioral Changes between Male and Female Postpubertal Rats Following Neonatal Excitotoxic Lesions of the Ventral Hippocampus. Brain Research, 973, 285-292. http://dx.doi.org/10.1016/S0006-8993(03)02537-X
- Paxinos, G. and Watson, C. (1986) The Rat Brain in Stereotaxic Coordinates. Academic Press, New York.
- Ornstein, K. and Amir, G. (1981) Pinch Induced Catalepsy in Mice. Journal of Comparative and Physiological, 95, 827-835.
- Ornstein, K. (1981) Suppression of Wet-Dog Shakes by Tail and Scruff Pinch. Physiology & Behavior, 27, 13-17. http://dx.doi.org/10.1016/0031-9384(81)90293-6
- Soper, W. and Melzack, R. (1981) Stimulation Produced Analgesia: Evidence for Somatotopic Organization in the Midbrain. Brain Research, 251, 301-313. http://dx.doi.org/10.1016/0006-8993(82)90747-8
- Miranda, A., De La Cruz, F. and Zamudio, S.R. (2006) Immobility Response Elicited by Clamping the Neck Induces Antinociception in a “Tonic Pain” Test in Mice. Life Science, 79, 1108-1113. http://dx.doi.org/10.1016/j.lfs.2006.03.018
- De Ryck, M., Schallert, T. and Teitelbaum, P. (1980) Morphine versus Haloperidol Catalepsy in the Rat: A Behavioral Analysis of Postural Mechanisms. Brain Research, 201, 143-172. http://dx.doi.org/10.1016/0006-8993(80)90781-7
- Blasing, J. (1978) On the Role of Brain Catecholamines in Acute and Chronic Opiate Action. In: Herz, A., Ed., Developments in Opiate Research, Dekker, New York, 279-356.
- Fregoso-Aguilar, T., Urióstegui, T., Zamudio, S. and De la Cruz, F. (2002) The Differential Effect of Haloperidol and Repetitive Induction on Four Immobility Responses in Mouse and Guinea Pig. Behavioural Pharmacology, 13, 253-260. http://dx.doi.org/10.1097/00008877-200207000-00001
- Dhir, A. and Kulkarni, S.K. (2008) Possible Involvement of Sigma-1 Receptors in the Anti-Immobility Action of Bupropion, a Dopamine Reuptake Inhibitor. Fundamental & Clinical Pharmacology, 22, 387-394.

