 J. Biomedical Science and Engineering, 2009, 2, 564-573 doi: 10.4236/jbise.2009.27082 Published Online November 2009 (http://www.SciRP.org/journal/jbise/ JBiSE ). Published Online November 2009 in SciRes. http://www.scirp.org/journal/jbise Signal averaging for noise reduction in anesthesia monitoring and contr ol with communication channels Zhi-Bin Tan1, Le-Y i Wang2*, Hong Wang3** 1,2Department of Electrical and Computer Engineering, Wayne State University, Detroit, USA; 3Department of Anesthesiology, Wayne State University, Detroit, USA. Email: 1au6063@wayne.edu; 2lywang@wayne.edu; 3howang@med.wayne.edu Received 29 May 2009; revised 14 July 2009; accepted 14 July 2009. ABSTRACT This paper investigates impact of noise and signal averaging on patient control in anesthesia applica- tions, especially in networked control system settings such as wireless connected systems, sensor networks, local area networks, or tele-medicine over a wide area network. Such systems involve communication channels which introduce noises due to quantization, channel noises, and have limited communication bandwidth resources. Usually signal averaging can be used effectively in reducing noise effe cts when remote monitoring and diagnosis are involved. However, when feedback is intended, we show that signal av- eraging will lose its utility substantially. To explain this phenomenon, we analyze stability margins under signal averaging and derive some optimal strategies for selecting window sizes. A typical case of anesthe- sia depth control problems is used in this develop- ment. Keywords: Anesthesia Depth; Anesthesia Monitoring; Anesthesia Control; Signal Averaging; Noise Reduction; Open and Closed Loop Systems; Communications; Net- worked Systems 1. INTRODUCTION To maintain an adequate depth of anesthesia without compromising patient’s health, an anesthesiologist usu- ally works as a multi-task feedback controller to roughly regulate the drugs titration while observing a variety of patient outcomes. Automatic anesthesia controller design aims to automatically regulate anesthesia levels by tak- ing account on several physiological measurements and then frees up anesthesiologists for more important tasks in operation. Closed-loop control of anesthesia has been a goal of many researchers since the middle of 20th century. With the emergence of BIS monitor in late 1990s, the interests in closed-loop control of depth of hypnosis is renewed, the most notable works are seen in [1,2,3,4]. In an operation room, a wide range of medical devices are connected together or connected to patient through cables for measuring, monitoring and diagnosis. The cable clutter interferes with patient care, creats haz- ards for clinical staff and delays transport and position- ing. To improve the clinical room efficiency and safty, it has been suggested to replace those cables with wireless connections [5]. While anesthesia patient vital signs such as anesthesia depth index, blood pressure, heart rate etc. are transmit- ted through a noisy wireless channel in a wide area, those transmitted signals will be corrupted by the trans- mission noise. It is well understood that within most algorithms that reduce effects of random noises on sig- nals and systems, some types of signal averaging are used [6,7]. This is mainly because the laws of large numbers and central limit theorems provide a foundation for noise reduction. The rationale is that when averaging is applied, noises diminish in an appropriate sense. This fundamental understanding leads to algorithms in filter- ing, signal reconstruction, state estimation, parameter estimation, system identification, and stochastic control. The signal averaging can be used effectively when re- mote monitoring and diagnosis are involved. On the other hand, signal averaging introduces dynamic delays. Such delays will have detrimental effects on closed-loop systems, even destabilizing the system. Consequently, signal averaging encounters a fundamental performance limitation in feedback systems. To explain this phe- nomenon, we analyze stability margins under signal av- eraging and derive some optimal strategies for selecting window sizes. A typical case of anesthesia depth control problems is used in this development. *Research of this author was supported in part by the National Science Foundation under ECS-0329597 and DMS-0624849, and Michigan Economic Development Council. **Research of this author was supported in part by Michigan Economic Development Council and Wayne State University’s Research En- hancement Program. This paper is organized as follows. Section 2 dis- cusses patient modeling and control in anesthesia appli-  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 565 cations. A typical case is presented with a detailed pa- tient model derived from clinical data. A feedback con- trol is designed to achieve closed-loop system stability, on the basis of state observers and pole placement con- trol. Signal averaging and its effectiveness on open-loop and closed-loop applications are demonstrated in Section 3. We show that while extending filter windows can im- prove noise attenuation in open-loop systems, it can de-stabilize a closed-loop system, implying a fundamen- tal performance limitation. The idea of using fast sam- pling is discussed. Theoretical foundation of our performance analysis is presented in Section 4. It is shown that this can be trans- formed into a calculation of gain margin of a modified system. Performance limitation is analyzed that leads to an optimal selection of filter windows. It is shown that for a given sampling rate, even optimally designed av- eraging filters can only have very limited benefits in reducing noise effects. It is shown that noise reduction ratio is proportional to the sampling interval, providing a means of obtaining noise reduction with communication resources. These findings are applied to anesthesia con- trol problems in Section 5. Finally, Section 6 summa- rizes some issues that are related but not resolved in this paper. 2. PATIENT MODELS AND FEEDBACK CONTROL Real-time anesthesia decisions are exemplified by gen- eral anesthesia for attaining an adequate anesthetic depth (consciousness level of a patient), ventilation control, etc [3,4,8,9]. One of the most critical requirements in this decision process is to predict the impact of the inputs (drug infusion rates, fluid flow rates, ventilator mode, etc.) on the outcomes (consciousness levels, blood pres- sures, heart rates, airway pressures, and oxygen satura- tion, etc.). This prediction capability can be used for control, display, warning, predictive diagnosis, decision analysis, outcome comparison, etc. 2.1. Patient Models The core function of this prediction capability is em- bedded in establishing a reliable model that relates the drug or procedure inputs to the outcomes in real-time and in individual patients. Due to significant deviations in physical conditions, ages, metabolism, pre-existing medical conditions, and surgical procedures, patient dy- namics demonstrate nonlinearity and large variations in their responses to drug infusion. A basic information- oriented model structure for patient responses to drug infusion was introduced in [10,11,12]. Propofol (a com- mon anesthesia drug) titration is administered by an in- fusion pump. The patient’s anesthesia depth is measured by a BIS (Bi-Spectrum) monitor [13,14]. The monitor provides continuously an index in the range of [0,100] such that the lower the index value, the deeper the anes- thesia state. Hence, an index value 0 will indicate “brain dead” and 100 will be “awake”. To establish patient models for monitoring and control, clinical data were collected. One of these data sets is used in this paper. The anesthesia process lasted about 76 minutes, starting from the initial drug administration and continuing until last dose of administration. Propofol was used in both titration and bolus. Fentanal was in- jected in small bolus amount three times, two at the ini- tial surgical preparation and one near incision. Analysis shows that the impact of Fentanal on the BIS values is minimal. As a result, it is treated as a disturbance and not explicitly modeled in this example. The drug infusion was controlled manually by an experienced anesthesi- ologist. The trajectories of titration (in μg/sec) and bolus injection (converted to μg /sec) during the entire surgical procedure were recorded, which are shown together with the corresponding BIS values in Figure 1. The patient was given bolus injection twice to induce anesthesia, first at minute with 20 mg and then at minute with 20 mg. They are shown in the figure as 10000 μg /sec for two seconds, to be consistent with the titration units. The surgical procedures were manu- ally recorded. Three major types of stimulation were identified: 1) During the initial drug administration (the first 6 minutes), due to set-up stimulation and patient nervousness. 2) Incision at minute for about 5 minutes duration. 3) Closing near the end of the surgery at minute. 3=t 5=t =t 45=t 60 The data from the first 30 minutes are used to deter- mine model parameters and functional forms. For esti- mating the parameters in the patient block, the data in the interval where the bolus and stimulation impact is minimal (between to minutes) are used. The patient model parameters were identified through Least-Squares estimation method [15]. 10=t30=t Under a sampling interval 1= second, which is the standard data transfer interval for the BIS monitor, the combined linear dynamics was estimated. The patient model with propofal infusion rate as the input and BIS measurement as the output was identified as 0.26780.29840.59890.75011.159 0.090160.088130.01872 = )( 2345 2 zzzzz zz zP (1) The actual BIS response is then compared to the model response over the entire surgical procedure. Comparison results are demonstrated in Figure 2. Here, the inputs of titration and bolus are the recorded real-time data. The model output represents the patient response very well. In particular, the model captures the SciRes Copyright © 2009 JBiSE 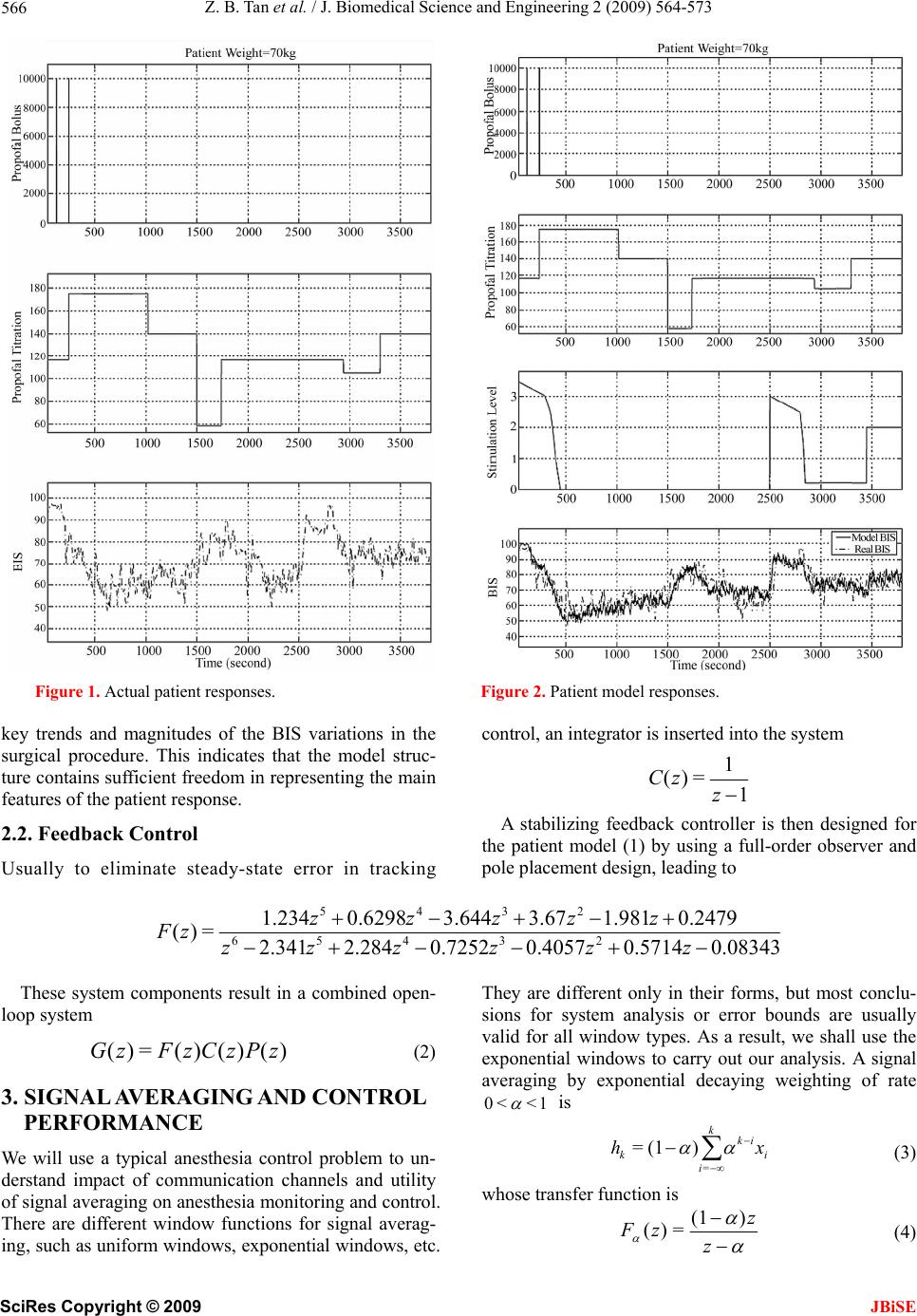 Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 566 Figure 1. Actual patient responses. Figure 2. Patient model responses. key trends and magnitudes of the BIS variations in the surgical procedure. This indicates that the model struc- ture contains sufficient freedom in representing the main features of the patient response. 2.2. Feedback Control Usually to eliminate steady-state error in tracking control, an integrator is inserted into the system 1 1 =)( z zC A stabilizing feedback controller is then designed for the patient model (1) by using a full-order observer and pole placement design, leading to 0.083430.57140.40570.72522.2842.341 0.24791.9813.673.6440.62981.234 =)(23456 2345 zzzzzz zzzzz zF These system components result in a combined open- loop system )()()(=)( zPzCzFzG (2) 3. SIGNAL AVERAGING AND CONTROL PERFORMANCE We will use a typical anesthesia control problem to un- derstand impact of communication channels and utility of signal averaging on anesthesia monitoring and control. There are different window functions for signal averag- ing, such as uniform windows, exponential windows, etc. They are different only in their forms, but most conclu- sions for system analysis or error bounds are usually valid for all window types. As a result, we shall use the exponential windows to carry out our analysis. A signal averaging by exponential decaying weighting of rate 1<<0 is i ik k i kxh = )(1= (3) whose transfer function is z z zF )(1 =)( (4) SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 567 Figure 3. Signal filtering in open-loop system. 3.1. Open-Loop Systems In wireless-based monitoring and diagnosis applications, the system is running in open-loop. In this case quanti- zation errors and communication noises can be grouped as an additive noise to the patient output y. When signal averaging is applied to reduce noise effects, the resulting system can be represented by the block diagram in Fig- ure 3. Figure 4 illustrates impact of filtering on open loop systems. In open loop applications, filtering will not de-stabilize the system. Consequently, one may choose a window of long horizon to reduce the effects of noise. It is apparent that the longer the averaging window, the less the noise effect on the signal. However, it is also observed that signal averaging slows down system’s Figure 4. Effects of signal averaging on open-loop systems. response to the input. In other words, filtering introduces a dynamic delay. This delay has very important implica- tion in the closed-loop applications. 3.2. Closed-Loop Systems On the other hand, if feedback control for anesthesia management decisions is intended, signal filtering be- comes part of a closed-loop system. When signal aver- aging is applied, the averaging filter Fa is inserted into the system, resulting in a modified closed-loop system shown in Figure 5. The close-loop system equations are: )(=,= kkkkkk dyFreGey (5) Then kkkk dGFyGFGry = and kkrk dHrHy = (6) where, GF GF H GF G Hr 1 =; 1 = (7) Figure 6 illustrates impact of filtering on closed loop systems. Although signal filtering can reduce the noise effect of the signal, it introduces a dynamic delay which has detrimental effects on the closed-loop system. The plots confirm that when filtering window is long the filter can destabilize the closed-loop system. Even when the filtering window size is small, its effectiveness is not very obvious. This example suggests that in closed-loop applications signal filtering has limited effectiveness. This understanding will be used to introduce new meth- ods to reduce noise effects in such applications. 3.3. Re-Sampling The plant in this case is identified as a 5t order differ- ence equation in (1). The system can be well approxi- h SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 568 Figure 5. System modules and their equivalent representation. Figure 6. Effects of signal averaging on closed-loop systems. Figur e 7. Step responses of the original system and the simplified system. mated by a continuous-time system that consists of a pure time delay and a first-order dynamics, sampled with sampling interval T=1 second. Let a continuous-time system be 173 0.93 =)( 5 s esP s (8) The step responses of the original system (1) and the simplified system P(s) are shown in Figure 7. This approximation allows us to use smaller sampling intervals to re-sample the output of the system. The benefits of re-sampling will become clear after some theoretical analysis in the next section. 4. ANALYSIS OF STABILITY AND PERFORMANCE Definition 1 The stability margin against exponential averaging, abbreviated as α-margin and denoted by )(G max , is the largest 10 such that for all )(<0 G max , the close-loop system (6) is stable and the system is unstable if . If the close-loop system is stable for all α, we denote )(> G max 1=)(G max . Suppose that the input to the filter is a noise corrupted constant =k d k x An exponential window of rate 1<<0 is applied to this signal and its output is ki ik x =i ik d = and identically distributed) k i )(1 = i ik k i d = ) k i k h )(1= = If i d is i.i.d. where k (1= (independent SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 569 wit 0= i Ed and 22 = i Ed , then 0= k E h and 2 2 1 1 = k E g k h as a C, usinn θ can re- du onsequently estimate of ce errors by 11 We will call . as the de- caying rate. Conv (in the mean sqre sense) is achieved when 1 ergence ua : 0= lim 2 1k E . Consider an entxpr oneial filte 1 1 =)( s sF onse is (9) wh resp ose impulse 0 ,=)(tetf t 1/ p of this (10) Now put-output relationshifilter is Note that , the in 1=)( 0 dttf d d )( )( with sampli ve xet t 1 =)/( Wgnals are sampled ng interval T, xtfty t)(=)( hen the si we denote )(= kTxxk and )(=kTyyk. If α is re- lated to λ and T /, we ha by =T e 1= )(1 lim 0 T T , is approxima ted by For small T)(ty i ik k i i ik x (1 analysis, we ma ponentia k i i ikT k i t k x T xe T ey == / = ( ))(1 )(1 =)( In other words, for systemy approxi- m Exl The between t dxkTy )/ )( 1 )( ate the discrete-time filter in (3) by its continuous-time counterpart in (9). These relationships between discrete- time averaging and continuous time averaging will be used to derive stability margins. 4.1. Stability Margin against Averaging relationship and will allow us to in ty margin in the co focus on stability analysis continuous time systems and then transform the results to the discrete-time filters. This is stated in the following theorem. Theorem 1 If the exponential stabili ntinuous-time domain is max , then max max T T = ln lim 0 Proof: This follows from the relationship We now concentrate on calculation of . nst nential av / =T e max Definition 2 The stability margin agai expo eraging for the continuous-time closed-loop system, abbreviated as continuous exponential A-margin and denoted by )(G max , is the smallest 0> under which the closed-loop system becomes able. If the closed-loop system remains stable for all 0> unst , we de- note =)(G max . Supp =N ose )()/(sDs ial functions of s )(sG where ar )(sn and )(sd )(sN e polynom and coprihat is, and )(sD do not have common zeros). Then max me (t is the t 0> larges before the closed-loop systee- comes unstablonsider the characteristic equation of the closed-loop system m b e. C 0= )( )( 1 1 1=)()(1 sD sN s sGsF or 0=)()()( sNsDssD (11) which leads to 0= )()( )( 1sNsD ssD (12) This expression leads to the following conclusion. Theorem 2 The exponential A-margin )(G max of )(sG is the gain margin of )()( )( =)( sNsD ssD sH (13) We make several interesting observations from (12). First, from (11), max may be calculated by using the Routh-Hurwitz test. Second, (12) is in a standard form for using root locus technique. So, we may plot the root locus of the system (13) (it is an improper system) and detect the value that reaches marginal stability, which will b max e . The root locus plot starts at the poles of system (13) which are precisely the poles of the closed-loop system without the averaging filter. Since SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 570 Figure 8. Using bode plots to obtain the gain margin. the closed-loop system is stable, for small the closed- ppose . Then, loop system with the filter will remain stable. The root locus plot moves towards the zeros of system (13) which are the poles of the open-loop system. Hence, if the open-loop system is unstable, the exponential A-margin is always finite. Example 1 Su1)2)/((=)( sssG 12= )()( )( =)( 2 sss sNsD ssD sH The gain margin can be obtained by using the Matlab function “margin” (which gives 2= max ) or by plotting the bode plot as shown in Fi which gives 2=d 6.02=B max gure 1 . Alternatively, from 0=1)(2=)(1)(2 22 sssss we can calculate 2= max by the Routh-Hurwitz method. Analysis e benefit of signal aver- ilarly, the continuous time close-loop system eq 4.2. Performance Within the A-margin, what is th aging? On one hand, signal averaging can reduce noise effect. On the other hand, averaging introduces delays and reduces closed-loop system performance. Conse- quently, an optimal choice of averaging becomes an is- sue. Sim uations are: d GF GF r GF G y 11 = (14) Here, we denote (15) If d is a white noise, noise attenuation aim the L2 norm of Hλ. Naturally, for op we should select s to reduce timal noise reduction, GF GF H 1 = 2 <<0 inf = H max (16) Example 2 For the system in Exa takes values 0,0.1,…0.9, the correspo for the closed-loop system Hλ are The monotone increase of the L2 norms indicates that fsmegnneduce n iac tus ts th mple 1, when λ nding H 2 norms or this yste, avragin caot roisempt on he outpt. A a result,here hould be no averaging for is system. Example 3 For another example, consider a system 221 2 () =4 s Gs ss The closed-loop system’s characteristic equation is 32 =(2)(14)3=0ss s () () ()sD sD sNs It can be calculated by the Routh-Hurwitz method that . The 1.366= max 2 norm of as a fution of λ Figur he optiml averaging occurs at H a nc is plotted in e 3. T 0.59= with the norm 2.5263= 0.59 PP H. ip / =T eor small sampling interval T, T/ 2 From the relationsh f l rate for averaging in the discrete-time do- main. For example, if , we obtain , TT e1.7/0.59 ee === is the optima 0.01=T0.983= . λ 0 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 η6.227.00 7.87 9.0010.49 12.59 15.73 20.9631.4462.85 Figure 9. Optimal averaging rate. SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 571 4.3. Fast Sampling for Disturbance Attenuation Although the optimal 2 performance in (16) can- not be improved for the continuous-time system, noise attenuation in the sampled system can be further im- proved. We first establish a relationship between the 2 of - to be- norm of the continuous-time system and the norm its sampled system. Suppose that the disbance se quence passes through a ZOH of inT come . The continuous-time system is stable with imesponse . Then, 2 l tur terval H k d )(td pulse r)(th 0 ()=()() t ythtd d Suppose is a pulse sequence, , and . Th, a w k d en, d 1= 0 d nd (td 0= k d, , other- 0Tt <0 ise. Under this input k1=)(t, 0=) 0 ()= () T ytht d Hence, the sampled values of )(ty , which form the pulse response of the sampled system, become 0 =( )=() T k yyKThkT d For small T this can be approximated by =() k yThkT We note that for smal l T, ) 22 2 20=0 =() ( k htdt ThT PP k Consequently, if we use and to dene led systese respe have )(= k h o ote th sampm and its imlnse, wpu kTThhk and 2222 22 =0 == ()= k k 2 2 hThkTTH PP PPPP From k dhy ~ =iik i k 0= if is i.i.d., mean zero and variance n 2 H In fact, 2 k d 2 , the 22 kk =0 =0 2 2 22 =0 == = kk kiijkj ij k i EyhEd dh T P PP 22 2 = k ki hh P 222 2 =sup max k k TH PP If 2 2 PP is optimized, then 22 2 H= PP as in (16). Consequently, the noise reduction ratio can be expressed as 22 =T (17) This is a relationship between noise reduction in the sastem and the optimal L2 norm of the continu- ous-time system. This analysis concludes that using faster sampling (smaller T) can reduce the noise effects. V ING Findings from Section 4 provide some useful design guidelines. 1) Signal averaging is beneficial in reducing noise effects. 2) Effectiveness of signal averaging in closed-loop systems varies substantially with the filter windows or decaying rates. There is an optimal decaying rate at which signal filtering becomes most effective. 3) filter window is optimally selected, further noise attenuation can only be achieved byng the sampling rates. 4) Increasing sampling rates incurs hi ban for com performance limit for noise attenuation. This is a unique feature for closed-loop applications, convergence can be si mpled sy 5. CONTROL WITH SIGNAL AERAG When the increasi gherdwidth requirementsmunications. When channel bandwidths are limited, there is a funda- mental systems. In open-loop obtained by applying gnal averaging over a very long horizon. However, this cannot be applied to closed-loop systems since long windows of filtering destabilize the feedback system. 5.1. Anesthesia Applications We now apply these understandings to anesthesia control systems. The open-loop transfer function in (2) can be derived as () ()= () Nz Gz Dz with 76 543 2 () =0.023110.09699 0.012430.4466 0.689 0.51010.2005 0.02235 Nz zz zzz zz and 12 11109 876 5 432 () =4.59.24811.48 9.576 5.6842.528 0.7518 0.2721 0.6608 0.507 Dz zzzz zzz z zzz 0.2003 0.02234z Since the open loop system is unstable, the stability margin of the closed-loop system with inserted averag- ing window is always limited. The closed-loop system’s stability concerns have already been depicted in Figure SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 572 6. The closed-loop system’s H2 no, which defines the system’s ability in noise attenuation, is shown in Figure 10. , the closed-loop system’s step response is simu- lated when the filter is optimally selected and shown in Figure 11. The system inherent sampling rate is T=1 second. While re-sampling is performed with T=1, the H2 norm of the closed-loop system will be reduced further. uced sampling intervals, improvements of noise tion are illustrated in Figure 12. 5.2. Discussions It can be seen from Figure 10 that the optimal filter de- caying rate is with the corresponding H2 norm 9.0872 wh closed-loop system is stable and it’ rm Then For red attenua 0.1300= opt en T=1. The s step response has much fluctuation in steady state. From the relationship, optopt T opt ee 1//== , we obtain 0.49= opt . This leads to the optimal choice of Figure 10. Closed-loop system performance vs. filter decaying rates. Figure 12. The closed-loop system performance for reduced sampling intervals. decaying rate when the sampling interval T is reduced from 1 as When sampling rate is increased to 1T, the H2 norm of the closed-loop system will be reduced to 9.0872T as established in (17). Figure 12 illustrates the step re- sponses of the closed-loop system with sampling interval T=0.5, T=0.1 and T=0.01 second respectively. The steady state fluctuation of the step response is decreasing with the reduced sampling intervals. 6. CONCLUSIONS ed sys- tems was investigated in this paper. Such systems in- volve communication channels which are corrupted by noises and have limited bandwidth resources. Signal averaging is the fundamental method in dealing with stochastic noises and errors. It is used effectively in re- ducing noise effects when only remote monitoring and diagnosis are involved. However, the case is different when feedback is intended. Our results show that the decaying rate of the averag- ing window has significant impact on the performance of the close-loop system. When α is larger than some value, the close-loop system becomes unstable. A concept of stability margins against exponential averaging is intro- duced. Its calculation can be performed by either the Routh-Hurwitz method or the root-locus method on a modified system. Furthermore, the strategy for choosing the optimal decaying rate is derived. Our results con- and design method is applied to anesthesia patient con- .=== 2.04/0.49 /TT opt Teee The impact of communication channels on feedback control in anesthesia applications in wireless bas clude that fast sampling must be used for improving noise reduction after optimal filter design. The analysis Figure 11. Step response of the closed-loop system when the filter is optimally selected, and sampling interval T=1. SciRes Copyright © 2009 JBiSE  Z. B. Tan et al. / J. Biomedical Science and Engineering 2 (2009) 564-573 SciRes Copyright © 2009 573 sis is conducted on the basis of the linear systems. Actually, anesthesia patient models contain non- linearity. Our future work will consider analysis of non- l anean Conference on Athens-Greece. erosa, M., and Morari, M., trol problems. Our analy [6] Talbot, S. L. and Boroujeny, B. F., (2008) Spectral of the method of blind carrier tracking for OFDM, IEEE Trans- actions on Signal Processing, 56(7). [7] Bataillou, E., Thierry, E., Rix, H., and Meste, O., (1995) Weighted averaging using adaptive estimation inear systems. REFERENCES [1] Nunes, C. S., Mendonca, T., Lemos, J. M., and Amorim, P., (2007) Predictive adaptive control of the bispectral index of the EEG (BIS): Exploring electromyography as an accessible disturbance, Mediterr weights, Signal Processing, 44, 5l−66. [8] Eisenach, J. C., (1999) Reports of Scientific Meetings— Workshop on Safe Feedback Control of Anesthetic Drug Delivery, Anesthesilogy, 91, 600–601. [9] Linkens, D. A., (1992) Adaptive and intelligent control in anesthesia, IEEE Control Systems Magazine, 6–11. [10] Wang, L. Y. and Wang, H., (2002) Control-oriented mod- eling of BIS-based patient response to anesthesia infu- sion, Internat. Conf. Math. Eng. Techniques in Medicine Control and Automation, [2] Gentilini, A., Rossoni-Get al, and Bio. Sci., Las Vegas. [11] Wang, L. Y. and Wang, H., (2002) Feedback and predic- tive control of anesthesia infusion using control-oriented patient models, Internat. Conf. Math. Eng. Techniques in Medicine and Bio. Sci., Las Vegas. (2001) Modeling and closed-loop control of hypnosis by means of bispectral index (BIS) with isoflurane, IEEE Trans. on Biomedical Engineering, 48, 874−889,. [3] Dong, C., Kehoe, J., Henry, J., Ifeachor, E. C., Reeve, C. D., and Sneyd, J. R., (1999) Closed-loop computer con- trolled sedation with propofol, Proc. of the Anaesthetic Research Society, 631. [4] Zhang, X. S., Roy, R. J., and Huang, J. W., (1998) Closed- loop system for total intravenous anesthesia by simulta- neously administering two anesthetic drugs, Proc. of the 20th Annual International Conference of the IEEE Engi- [12] Wang, L. Y., Wang, H., and Yin, G., (2002) Anesthesia infusion models: Knowledge-based real-time identifica- tion via stochastic approximation, 41st IEEE Cont. and Dec. Conf., Las Vegas. [13] Gan, T. J., et al., (1997) Bispectral index monitoring allows faster emergence and improved recovery from propofol, Alfentanil, and Nitrous Oxide Anesthesia, An- esthesiology, 87, 808–815. [14] Rosow, C. and Manberg, P. J., (1998) Bispectral index neering in Medicine and Biology, 3052–3055. [5] Goldman, J. M., (2006) Medical device connectivity for improving safety and efficiency, American Society of Anesthesiologists Newsletters, 70(5), http://www.asahq.org/Newsletters/2006/05−06/goldman0 5_06.html. monitoring, Annual of Anesthetic Pharmacology, 2, 1084– 2098. [15] Ljung, L. and Söderström, T., (1983) Theory and Practice of Recursive Identification, MIT Press, Cambridge, MA. JBiSE
|