 Pharmacology & Pharmacy, 2011, 2, 282-288 doi:10.4236/pp.2011.24036 Published Online October 2011 (http://www.SciRP.org/journal/pp) Copyright © 2011 SciRes. PP Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, Pyrazolic Chalcones, and Allylic Chalcones Tan Nhut Doan, Dao Thanh Tran Department of Medicinal Chemistry, School of Pharmacy, University of Medicine and Pharmacy, Ho Chi Minh City, Vietnam. Email: tan.doan@uqconnect.edu.au Received August 17th, 2011; revised September 11th, 2011; accepted September 30th, 2011. ABSTRACT A new series of chalcones (4a-c) and allylicchalcones (11a-b) have been prepared by the Claisen-Schmidt condensation. A novel series of pyrazolicchalcones (5a-c) have been synthesized by the reaction of respective chalcones (4a-c) and hydrazine hydrate. The structures of the compounds were confirmed by spectral data (infrared spectroscopy and 1H nuclear magnetic resonance). All of the compounds (4/5a-c and 11a-b) have been tested for their antimicrobial activi- ties (agar disc-diffusion method) and antioxidant activities (1,1-biphenyl-2-picrylhydrazyl free radical scavenging method). The test compounds failed to show antibacterial properties (4a-c, 5b, and 11a-b) or exhibited such properties poorly (5a and 5c). None of the test compounds displayed antifungal properties. Of the compounds tested, compounds 5a-c and 11a-b exhibited promising antioxidant activities. Keywords: AllylicChalcones, Antioxidant Activity, Antimicrobial Activity, PyrazolicChalcones 1. Introduction Flavonoids comprise a large family of plant-derived poly- phenolic compounds classified as anthocyanidins, fla- vonols, chalcones, aurones, flavanones, isoflavones, fla- vans, flavanonols, flavanols, and flavones differencing from each other in their structural group arrangements [1]. Chalcone, an important intermediate of flavonoid synthetic pathway, has been shown to exhibit diverse biological and pharmacological activities such as anti- cancer, antioxidant, anti-inflammatory, antimicrobial, anti- allergic,and antimalarial properties [2-4]. Oxidative damage which is implicated in various patho- logical events such as cancer and aging is induced by free radicals and reactive oxygen species [5]. Antioxidants are the compounds that prevent such oxidative damage due to their free radical scavenging ability [5]. In chal- cones, such ability is attributable to phenolic-OH group attached to the ring structure [6]. Chalcones with anti- oxidant activity (and compounds with such activity in general) have been demonstrated to have anticancer, anti- cardiovascular, anti-inflammatory, and many other active- ties [7,8]. As such, they have gained immense interest from bioorganic and medicinal chemistry research. Synthesized chalcones holding allylic substitutions and pyrazolicchalcones were recently reported as potent an- timicrobial and antioxidant agents [9-12]. In addition, the presence of enone function in chalcones having pyrazole moiety has been found to enhance the biological activity [13]. Prompted by all these observations, we report herein the synthesis, antioxidant and antimicrobial activities of novel chalcones, pyrazolicchalcones and allylicchalcones. 2. Chemistry In order to obtain pyrazolicchalcones 5a-c, the corre- sponding chalcones 4a-c were prepared by the Claisen- Schmidt condensation of respective acetophenones (1) and benzaldehydes (2) by the known literature method [14]. Subsequently, the corresponding pyrazolicchal- cones 5a-c were prepared by the addition of the obtained chalcones 4a-c to hydrazine hydrate in absolute ethanol (Scheme 1). All of the compounds were characterized by spectral data (infrared spectroscopy [IR] and 1H nuclear magnetic resonance [NMR]). The synthetic procedures and reaction conditions for allylicchalcones 11a-b are illustrated in Scheme 2. 2-Al- lyloxybenzaldehyde (8) was prepared by the reaction of 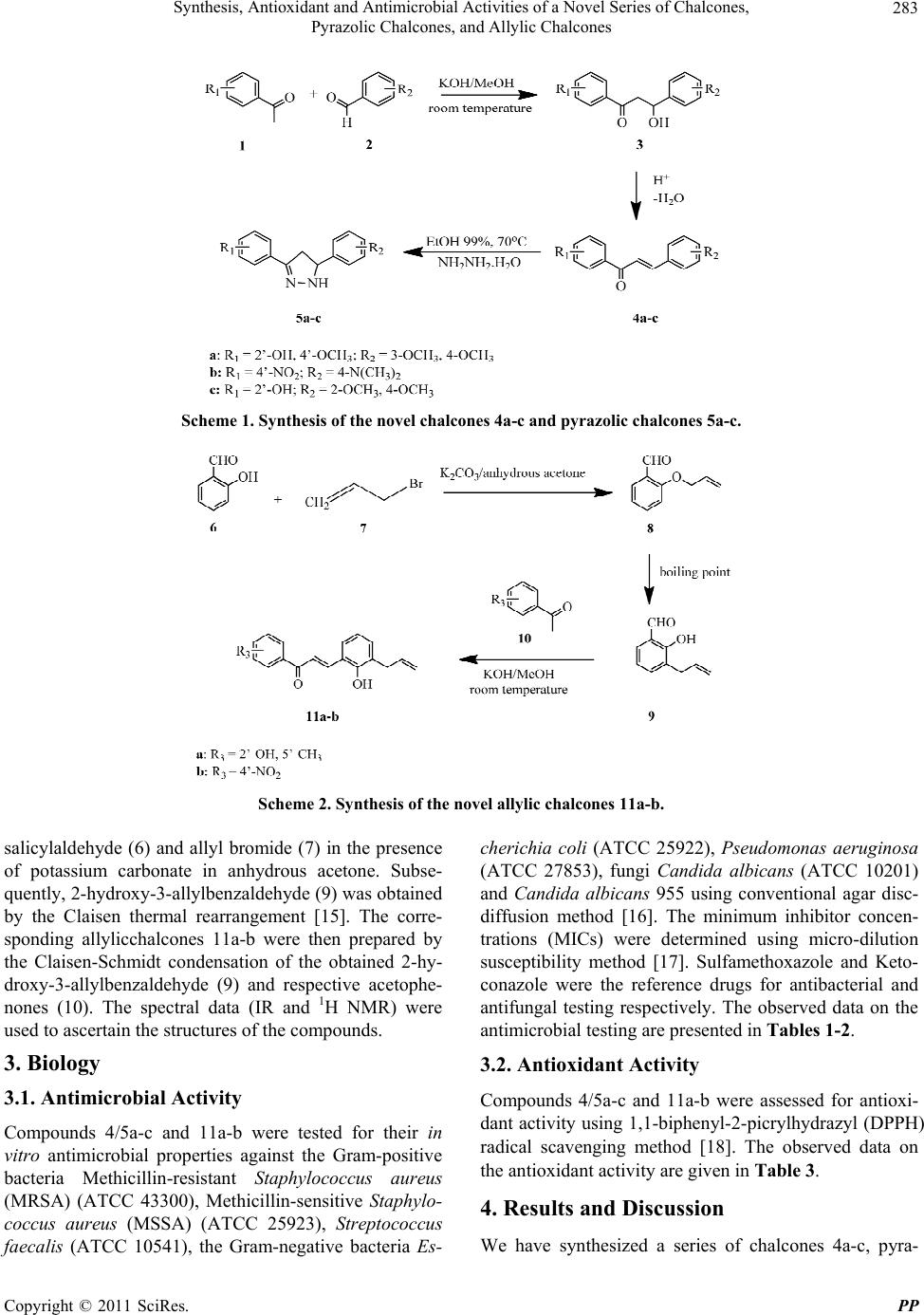 Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, 283 Pyrazolic Chalcones, and Allylic Chalcones Scheme 1. Synthesis of the novel chalcones 4a-c and pyrazolic chalcones 5a-c. Scheme 2. Synthesis of the novel allylic chalcones 11a-b. salicylaldehyde (6) and allyl bromide (7) in the presence of potassium carbonate in anhydrous acetone. Subse- quently, 2-hydroxy-3-allylbenzaldehyde (9) was obtained by the Claisen thermal rearrangement [15]. The corre- sponding allylicchalcones 11a-b were then prepared by the Claisen-Schmidt condensation of the obtained 2-hy- droxy-3-allylbenzaldehyde (9) and respective acetophe- nones (10). The spectral data (IR and 1H NMR) were used to ascertain the structures of the compounds. 3. Biology 3.1. Antimicrobial Activity Compounds 4/5a-c and 11a-b were tested for their in vitro antimicrobial properties against the Gram-positive bacteria Methicillin-resistant Staphylococcus aureus (MRSA) (ATCC 43300), Methicillin-sensitive Staphylo- coccus aureus (MSSA) (ATCC 25923), Streptococcus faecalis (ATCC 10541), the Gram-negative bacteria Es- cherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27853), fungi Candida albicans (ATCC 10201) and Candida albicans 955 using conventional agar disc- diffusion method [16]. The minimum inhibitor concen- trations (MICs) were determined using micro-dilution susceptibility method [17]. Sulfamethoxazole and Keto- conazole were the reference drugs for antibacterial and antifungal testing respectively. The observed data on the antimicrobial testing are presented in Tables 1-2. 3.2. Antioxidant Activity Compounds 4/5a-c and 11a-b were assessed for antioxi- dant activity using 1,1-biphenyl-2-picrylhydrazyl (DPPH) radical scavenging method [18]. The observed data on the antioxidant activity are given in Table 3. 4. Results and Discussion We have synthesized a series of chalcones 4a-c, pyra- Copyright © 2011 SciRes. PP  Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, 284 Pyrazolic Chalcones, and Allylic Chalcones zolicchalcones 5a-c and allylicchalcones 11a-b with ap- propriate nucleophiles using the procedures presented in Schemes 1 and 2. 4.1. Antimicrobial Activity The results of antimicrobial testing of compounds 4/5a-c and 11a-b are presented in Tables 1-2. The results indi- cated that compounds 4a-c, 5b and 11a-b did not exhibit antibacterial properties whereas compounds 5a and 5c displayed poor antibacterial activity towards Gram-posi- tive bacteria showing inhibitor zones between 10 and 16 mm and MIC values higher than 256 μg/ml for Strepto- coccus faecalis and 512 μg/ml for MSSA and MRSA compared to Sulfamethoxazole. These compounds (5a and 5c), however, did not exhibit antibacterial activity against Gram-negative bacteria. None of the synthesized compounds (4/5a-c and 11a-b) showed antifungal prop- erties. 4.2. Antioxidant Activity The antioxidant activity of compounds 4/5a-c and 11a-b were tested using DPPH radical scavenging method [18]. Except for compounds 4a-c which failed to show such activity, all of the other test compounds (5a-c and 11a-b) exhibited good antioxidant properties, with the strongest being observed in compound 5a (Table 3). However, all the synthesized compounds were less potent than vitamin C as the reference. The potencies for the antioxidant ac- tivity of the test compounds to the reference drug are in the following order: Vitamin C > 5a-b > 5c > 11a-b. 5. Conclusions In summary, we have synthesized a series of novel chal- cones, pyrazolicchalcones and allylicchalcones. We have also reported antimicrobial and antioxidant evaluations of these compounds. All the synthesized compounds showed poor antimicrobial properties or did not show such properties. A good antioxidant activity was ob- served in pyrazolicchalcones and allylicchalcones. 6. Experimental 6.1. Chemistry All chemicals were purchased from commercial suppliers, and used without further purification. All solvents used for reaction were freshly distilled from proper dehydrate- ing agents. Melting points were determined in open cap- illaries on a Gallenkamp Melting Point Apparatus and are uncorrected. The purity of the compounds was checked by thin layer chromatography (TLC) (silica gel H, n-hexane- acetone 3:1). The IR spectra were performed on a Shi- madzu FTIR 8101 spectrometer in potassium bromide Table 1. Antimicrobial activity of 4/5a-c and 11a-b. Zone of inhibition at concentration of 1024 μg/ml (mm) Gram-positive bacteria Gram-negative bacteriaFungi Compound MSSA MRSA SF EC PA CA 10201 CA 955 4a - - - - - - - 4b - - - - - - - 4c - - - - - - - 5a 12 14 11 - - - - 5b - - - - - - - 5c 10 16 14 - - - - 11a - - - - - - - 11b - - - - - - - Sulfamethoxazole34 36 38 35 33 - - Ketoconazole - - - - - 41 39 Control (DMSO)- - - - - - - Data represent the mean of three replicates. MSSA—Methicillin-sensitive Staphylococcus aureus(ATCC 25923); MRSA— Methicillin-resistant Staphylococcus aureus(ATCC 43300); SF—Streptococcus faecalis (ATCC 10541); EC—Escherichia coli (ATCC 25922); PA—Pseudomonas aeruginosa(ATCC 27853); CA 10201—Candida albicans (ATCC 10201); CA 955—Candida albicans 955; DMSO—dimethyl sulfoxide. (-) indicates “not detected”. Copyright © 2011 SciRes. PP  Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, 285 Pyrazolic Chalcones, and Allylic Chalcones Table 2. Minimum inhibitor concentration (MIC), μg/ml of 5a and 5c. Minimum inhibitor concentration (MIC) (μg/ml) Gram-positive bacteria Compound MSSA MRSA SF 5a 512 512 256 5c 512 512 256 Sulfamethoxazole 4 4 4 MSSA—Methicillin-sensitive Staphylococcus aureus (ATCC 25923); MRSA— Methicillin-resistant Staphylococcus aureus (ATCC 43300); SF—Streptoco- ccus faecalis (ATCC 10541). Table 3. Antioxidant property of 4/5a-c and 11a-b. Compound % DPPH 4a 0.00 4b 0.00 4c 0.00 5a 89.64 5b 89.27 5c 77.14 11a 41.00 11b 40.90 Vitamin C* 97.92 DPPH—1,1-biphenyl-2-picrylhydrazyl. *Standard substance. (KBr) pellets and the wave numbers were given in cm–1. The 1H NMR spectra were run on a Bruker spectrometer operating at 500 MHz, using deuterated chloroform (CDCl3) as solvent and tetramethylsilane (TMS) as inter- nal standard. All chemical shifts are reported in parts per million (ppm) downfield relative to TMS on the δ scale. Data are reported as follows: chemical shift, multiplicity (s = single, d = double, t = triplet, q = quarter, m = multi- plier, br = broad), coupling constant (Hz) and integration. 6.1.1. 2-Hydroxy-4-methoxyacetophenone 2,4-Dihydroxyacetophenone (0.1 mol), potassium car- bonate (0.3 mol) and dimethyl sulphate (0.09 mol) in 20 ml anhydrous acetone were refluxed for 4 hours, moni- toring by TLC with solvent system of n-hexane—acetone (3:1). After removing potassium carbonate, the reaction solution was evaporated in vacuo, giving solid. The solid was filtered, washed with water, dried, and crystalized from methanol to yield 2-hydroxy-4-methoxyacetophe0 none as gray solid with overall yield of 81%. This prod- uct was used as the material for the synthesis of 4a. 6.1.2. 2’-Hydroxy-3,4,4’-trimethoxychalcone (4a) 2-Hydroxy-4-methoxyacetophenone (1.05 equivalence) and 3,4-dimethoxybenzaldehyde were dissolved in methanol. To the above mixture, potassium hydroxide (3 equiva- lence) was added in portions to give a blood-red solution. The reaction mixture was stirred at room temperature for 32 hours, during which 2’-hydroxy-3,4,4’-trimethoxy- chalcone (4a) precipitated as the potassium salt. The re- action mixture was poured into cold 1N hydrochloride acid (HCl) solution and was further added concentrated HCl (c-HCl) until the solution became acidic. The result- ing precipitate was filtered, washed with water, and crystalized from methanol to give product 4a as crystals. Yield 61%. Light yellow solid, melting point (mp): 170˚C, IR (KBr), ν (cm–1): 1633 (C=O), 1564 (C=C aryl), 1126 (C-O). 1H-NMR (500 MHz, CDCl3): 13.50 (s, 1 H, Ar-OH), 7.85 - 7.82 (d, J = 15.5 Hz, 1 H, Hβ), 7.84 - 7.82 (d, J = 8 Hz, 1 H, H6’), 7.44 - 7.41 (d, J = 15.5 Hz, 1H, Hα), 7.25 - 7.23 (d, J = 2 Hz, 8.5 Hz, 1H, H6), 7.16 (d, J = 2 Hz, 1H, H3’), 6.91 - 6.89 (d, J = 8 Hz, 1H, H5), 6.49 - 6.47 (d, J = 2.5 Hz, 8.5 Hz, 1H, H5’), 6.47 (s, J = 2 Hz, 1H, H2), 3.96 (s, 3H, Ar-OCH3), 3.93 (s, 3H, Ar-OCH3), 3.85 (s, 3H, Ar-OCH3). 6.1.3. 4’-Nitro-4-dimethylaminochalcone (4b) 4-Nitroacetophenone (1.05 equivalence) and 4-dimethy- laminobenzaldehyde were dissolved in methanol. To the above mixture, potassium hydroxide (3 equivalence) was added in portions to give a blood-red solution. The reac- tion mixture was stirred at room temperature for 32 hours, during which 4’-nitro-4-dimethylaminochalcone (4b) pre- cipitated as the potassium salt. The reaction mixture was poured into cold 1N-HCl solution and was further added c-HCl until the solution became acidic. The resulting precipitate was filtered, washed with water, and crystal- ized from methanol to give product 4b as crystals. Yield 79%. Light yellow solid, mp: 206˚C, IR (KBr), ν (cm–1): 1647 (C=O), 1518 (C=C aryl), 1258 (C-O). 1H-NMR (500 MHz, CDCl3): 7.93 - 7.90 (d, J = 15 Hz, 1H, Hβ), 7.93 - 7.91 (d, J = 1.5 Hz, 7.5 Hz, 1H, H6), 7.58 - 7.55 (d, J = 8 Hz, 2H, H2’, H6’), 7.47 - 7.44 (d, J = 15 Hz, 1H, Hα), 7.47 - 7.44 (t, 1H, H4), 7.02 - 7.00 (d, J = 8 Hz, 1H, H3), 6.93 - 6.90 (t, 1H, H4’), 6.71 - 6.69 (d, J = 2 Hz, 8 Hz, 2H, H3, H5), 3.05 (s, 6H, 2 × CH3). 6.1.4. 2’-Hydroxy-2,4-dimethoxychalcone (4c) 2-Hydroxyacetophenone (1.05 equivalence) and 2,4-di- methoxybenzaldehyde were dissolved in methanol. To the above mixture, potassium hydroxide (3 equivalence) was added in portions to give a blood-red solution. The reaction mixture was stirred at room temperature for 20 Copyright © 2011 SciRes. PP  Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, 286 Pyrazolic Chalcones, and Allylic Chalcones hours, during which 2’-hydroxy-2,4-dimethoxychalcone (4c) precipitated as the potassium salt. The reaction mix- ture was poured into cold 1N-HCl solution and was fur- ther added c-HCl until the solution became acidic. The resulting precipitate was filtered, washed with water, and crystalized from methanol to give product 4c as crystals. Yield 59%. Light yellow solid, mp: 111˚C, IR (KBr), ν (cm–1): 1637 (C=O), 1558 (C=C aryl), 1170 (C-O). 1H- NMR (500 MHz, CDCl3): 13.10 (s, 1H, Ar-OH), 8.14 - 8.22 (d, J = 15.4 Hz, 1H, Hβ), 7.90 - 7.94 (d, J = 8.2 Hz, 1H, H6’), 7.64 - 7.75 (d, J = 15.4, 1H, Hα), 7.58 - 7.62 (d, J = 8.6 Hz, 1H, H6), 7.44 - 7.52 (m, J = 8 Hz, 1H, H4’), 6.99 - 7.03 (d, J = 8.6 Hz, 1H, H5’), 6.93 - 6.97 (d, J = 8.4 Hz, 1H, H3’), 6.55 - 6.59 (d, J = 7.6 Hz, 1H, H5), 6.5 (ds, J = 1.4 Hz, 1H, H3), 3.93 (s, 3H, Ar-OCH3), 3.87 (s, 3H, Ar-OCH3). 6.1.5. 2-(5-(3,4-Dimethoxyphenyl)-4,5-dihydro-1H- pyrazol-3-yl)-5-methoxyphenol (5a) 2’-Hydroxy-3,4,4’-trimethoxychalcone (4a, 2 mmol) was dissolved in 100 ml absolute ethanol. To this mixture, hydrazine hydrate (4 mmol) was added dropwise at room temperature. The reaction mixture was refluxed for 7 hours, monitoring by TLC with solvent system of n- hexane-acetone (3:1). The reaction mixture was cooled in ice bath. The resulting white solid was filtered and wash- ed with cold water. The filtrate was extracted with ether and evaporated in vacuo yielding more solid. The solid was collected and crystalized from ethanol to yield 2-(5- (3,4-dimethoxyphenyl)-4,5-dihydro-1H-pyrazol-3-yl)-5- methoxyphenol (5a). White solid, mp: 115˚C, IR (KBr), ν (cm–1): 3336 (N-H), 1591 (C=N), 1155 (C-O). 1H- NMR (500 MHz, CDCl3): 11.19 (s, 1H, Ar-OH), 7.07 - 7.06 (d, J = 8.5 Hz, 1H, H6’), 6.93 - 6.92 (s, J = 1 Hz, 8 Hz, 1H, H2”), 6.90 - 6.88 (d, J =1.5 Hz, 8 Hz, 1H, H5”), 6.84 - 6.82 (d, J = 8.5 Hz, 1H, H6”), 6.56 (s, J = 1.5 Hz, 1H, H3’), 6.46 - 6.44 (d, J = 2.5 Hz, 8 Hz, 1H, H5’), 5.87 (s, 1H, NH), 4.82 - 4.78 (t, 1H, H3), 3.87 (s, 6H, 2 × Ar- OCH3), 3.81 (s, 3H, Ar-OCH3), 3.52 - 3.47 (q, 1H, H4β), 3.09 - 3.05 (q, 1H, H4α). 6.1.6. N,N-Dimethyl-4-(3-(4-nitrophenyl)-4,5- dihydro-1H-pyrazol-5-yl)-benzenamin (5b) 4’-Nitro-4-dimethylaminochalcone (4b, 2 mmol) was dis- solved in 100 ml absolute ethanol. To this mixture, hydra- zine hydrate (4 mmol) was added dropwise at room tem- perature. The reaction mixture was refluxed for 7 hours, monitoring by TLC with solvent system of n-hexane- acetone (3:1). The reaction mixture was cooled in ice bath. The resulting white solid was filtered and washed with cold water. The filtrate was extracted with ether and evaporated in vacuo yielding more solid. The solid was collected and crystalized from ethanol to yield N,N-di- methyl-4-(3-(4-nitrophenyl)-4,5-dihydro-1H-pyrazol-5- yl)-benzenamin (5b). White solid, mp: 154˚C - 154.7˚C, IR (KBr), ν (cm–1): 3334 (N-H), 1614 (C=N). 1H-NMR (500 MHz, CDCl3): 8.22 - 8.20 (d, J = 2 Hz, 9 Hz, 2H, H3’, H4’), 7.78 - 7.76 (d, J = 1.5 Hz, 9 Hz, 2H, H2’, H6’), 7.21 - 7.19 (d, J = 8.5 Hz, 2H, H2”, H6”), 6.71 - 6.69 (d, J = 2 Hz, 9 Hz, 2H, H3”, H4”), 6.19 (s, 1H, NH), 4.95 - 4.92 (t, 1H, H3), 3.45-3.39 (dd, 1H, H4), 2.94 (s, 6H, 2 × CH3). 6.1.7. 2-(5-(2,4-Dimethoxyphenyl)-4,5-dihydro- 1H-pyrazol-3-yl)-phenol (5c) 2’-Hydroxy-2,4-dimethoxychalcone (4c, 2 mmol) was dis- solved in 100 ml absolute ethanol. To this mixture, hy- drazine hydrate (4 mmol) was added dropwise at room temperature. The reaction mixture was refluxed for 7 hours, monitoring by TLC with solvent system of n- hexane-acetone (3:1). The reaction mixture was cooled in ice bath. The resulting white solid was filtered and washed with cold water. The filtrate was extracted with ether and evaporated in vacuo yielding more solid. The solid was collected and crystalized from ethanol to yield 2-(5-(2,4-dimethoxyphenyl)-4,5-dihydro-1H-pyrazol-3-yl)- phenol (5c). White solid, mp: 154˚C - 154.7˚C, IR (KBr), ν (cm–1): 3327 (N-H), 1591 (C=N), 1153 (C-O). 1H- NMR (500 MHz, CDCl3): 11.09 (s, 1H, Ar-OH), 7.24 - 7.18 (m, 3H, H3’, H4’, H6’), 6.99 - 6.97 (d, J = 1Hz, 8 Hz, 1H, H6”), 6.87 - 6.84 (t, J = 8.5 Hz, 1H, H5’), 6.47 (sb, 1H, NH), 6.47 - 6.44 (m, 2H, H3”, H5”), 5.13 - 5.09 (t, 1H, H3), 3.82 (s, 3H, Ar-OCH3), 3.79 (s, 3H, Ar-OCH3), 3.50 - 3.45 (q, 1H, H4β), 3.08 - 3.03 (q, 1H, H4α). 6.1.8. 2-Allyloxybenzaldehyde (8) To a solution of salicylaldehyde (6, 0.1 mol) and allyl bromide (7, 0.11 mol) in anhydrous acetone (30 ml), po- tassium carbonate (0.12 mol) was added. The reaction mixture was refluxed for 3 hours. At the completion of the reaction (monitoring by TLC with solvent system of n-hexane-acetone 3:1), the resulting mixture was cooled to room temperature and poured into water (150 ml). The aqueous phase was then extracted with dichloromethane. The obtained extract was washed with 5% sodium hy- droxide (NaOH) solution and water. The organic phase was dried over anhydrous magnesium sulfate and con- centrated under reduced pressure. After distillation under reduced pressure, 2-allyloxybenzaldehyde (8) was ob- tained as a yellow liquid. Yield 87%, boiling point (bp): 273.5˚C/760 mmHg. 1H-NMR (500 MHz, CDCl3): 4.75 (dd, J = 5.0 Hz, 1.6 Hz, 2H), 5.13 - 5.43 (m, 2H), 5.4 - 5.9 (m, 1H), 6.65 - 8.12 (m, 4H), 10.62 (s, 1H). 6.1.9. 2-Hydroxy-3-allylbenzaldehyde (9) 14 g of 2-allyloxybenzaldehyde (8) was heated in a sealed tube. After 12 hours, the rearrangement was complete. Copyright © 2011 SciRes. PP  Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, 287 Pyrazolic Chalcones, and Allylic Chalcones The reaction was cooled to room temperature and diluted with 50 ml diethyl ether. The diluted solution was then extracted with 10% NaOH. The alkaline extract was added 10% HCl until the solution became slightly acidic (pH 6). The above solution was then extracted with di- ethyl ether. The ethereal extract was dried with anhy- drous magnesium sulfate and was concentrated in vacuo. The oily residue was distilled under reduced pressure to get 2-hydroxy-3-allylbenzaldehyde (9) as a light yellow liquid. Yield 72.8% (10.2 g), bp: 243.5˚C/760 mmHg. 1H-NMR (500 MHz, CDCl3): 11.30 (s, 1H, Ar-OH), 9.85 (s, 1H, CHO), 7.41 - 7.39 (m, 2H, H4, H6), 6.97 - 6.94 (t, 1H, H5), 6.03 - 5.95 (m, 1H, H2’), 5.12 - 5.08 (m, 2H, =CH2), 3.44 - 3.41 (d, 1H, H1’). Compound 9 was used as the material for the synthesis of compounds 11a-b. 6.1.10. 2’-Hydroxy-5’-methyl-2-hydroxy-3- allylchalcone (11a) 2-Hydroxy-5-methylacetophenone (1.05 equivalence) and 2-hydroxy-3-allylbenzaldehyde (9) were dissolved in me- thanol. To the above mixture, potassium hydroxide (3 equivalence) was added in portions to give a blood-red solution. The reaction mixture was stirred at room tem- perature for 52 hours, during which 2’-hydroxy-5’-me- thyl-2-hydroxy-3-allylchalcone (11a) precipitated as the potassium salt. The reaction mixture was poured into cold 1N-HCl solution and was further added c-HCl until the solution became acidic. The resulting precipitate was filtered, washed with water, and crystalized from metha- nol to give product 11a as crystals. Yield 40%. Light purple solid, mp: 156˚C, IR (KBr), ν (cm–1): 1706 (C=O), 1559 (C=C aryl), 1166 (C-O). 1H-NMR (500 MHz, CDCl3): 12.65 (s, 1H, Ar-OH), 8.20 - 8.17 (d, J = 15.5 Hz, 1H, Hβ), 7.71 - 7.68 (d, J = 15.5, 1H, Hα), 7.66 (s, 1H, H6), 7.51 - 7.49 (d, J = 1 Hz, 7.5 Hz, 1H, H6’), 7.28 (s, J = 2 Hz, 1H, H2’), 7.16 - 7.14 (d, J = 1 Hz, 7.5 Hz, 1H, H4), 7.04 - 7.02 (d, J = 1 Hz, 8 Hz, 1H, H3’), 6.91 - 6.88 (t, 1H, H5’), 6.89 - 6.87 (d, J = 8.5 Hz, 1H, H4’), 6.03 - 5.95 (m, 1H, H2”), 5.16 - 5.12 (m, 2H, =CH2), 3.42 - 3.41 (d, 1H, H1”), 2.30 (s, 3H, Ar-CH3). 6.1.11. 4’-Nitro-2-hydroxy-3-allylchalcone (11b) 4’-Nitroacetophenone (1.05 equivalence) and 2-hydroxy- 3-allylbenzaldehyde (9) were dissolved in methanol. To the above mixture, potassium hydroxide (3 equivalence) was added in portions to give a blood-red solution. The reaction mixture was stirred at room temperature for 50 hours, during which 4’-nitro-2-hydroxy-3-allylchalcone (11b) precipitated as the potassium salt. The reaction mixture was poured into cold 1N-HCl solution and was further added c-HCl until the solution became acidic. The resulting precipitate was filtered, washed with water, and crystalized from methanol to give product 11b as crystals. Yield 45%. Light purple solid, mp: 180˚C, IR (KBr), ν (cm–1): 1651 (C=O), 1573 (C=C aryl), 1170 (C-O). 1H- NMR (500 MHz, CDCl3): 8.28 - 8.26 (d, J = 8.5 Hz, 2H, H3, H4), 8.141 - 8.11 (d, J = 16 Hz, 1H, Hβ), 8.08 - 8.06 (d, J = 8.5 Hz, 2H, H2, H6), 7.53 - 7.49 (d, J = 16 Hz, 1H, Hα), 7.47 - 7.45 (s, J = 8 Hz, 1H, H6’), 7.15 - 7.13 (d, J = 7.5 Hz, 1H, H4’), 6.88 - 6.86 (t, 1H, H5’), 5.98 - 5.93 (m, 1H, H2”), 5.12 - 5.07 (m, 2H, =CH2), 3.39-3.38 (d, 1H, H1”). 6.2. Biological Evaluation 6.2.1. Antimicrobial Testing Compounds 4/5a-c and 11a-b were evaluated for their in vitro antimicrobial activity by agar disc-diffusion method [16]. Stock solutions of test compounds were diluted in dimethyl sulfoxide (DMSO) (1%) to give a final concen- tration of 1024 μg/ml. The DMSO (1%) alone was used as a control. Sterile filter paper discs (6 mm diameter) moistened with the test compound solution were care- fully placed on the agar culture plates which had been previously inoculated separately with the microorgan- isms. The plates were incubated at 37˚C for 24 hours (in the case of bacteria) or 48 hours (in the case of fungi). After incubation, growth was surveyed by measuring the diameter of the growth inhibition zones. All determina- tions were made in triplicate for each compound. Aver- age of three independent readings for each compound was recorded. The results are given in Table 1. MIC was defined as the lowest concentration of com- pound required for a complete inhibition of inoculated bacteria or fungi after incubation time. Sulfamethoxazole and Ketoconazole were used as reference agents for an- tibacterial and antifungal activities respectively. The MICs of the test compounds were determined using micro- dilution method [17]. The test compounds, Sulfameth- oxazole and Ketoconazole were dissolved in DMSO (1%) to give a concentration of 1024 μg/ml; and two-fold dilu- tion of the solution was prepared (512, 256, 128, 64, 32, 16, 8, and 4 μg/ml). The microorganism suspensions were inoculated to the corresponding wells. The plates were incubated at 37˚C for 24 hours (in the case of bac- teria) or 48 hours (in the case of fungi). The MIC values are displayed in Table 2. 6.2.2. Antioxidant Testing Compounds 4/5a-c and 11a-b were tested for antioxidant activity by DPPH radical scavenging method [18]. The nitrogen centered stable free radical DPPH has been in widespread use in spectrophotometric studies to charac- terize antioxidants [19]. This is based on the fact that the odd electron in the DPPH free radical gives a strong ab- Copyright © 2011 SciRes. PP  Synthesis, Antioxidant and Antimicrobial Activities of a Novel Series of Chalcones, Pyrazolic Chalcones, and Allylic Chalcones Copyright © 2011 SciRes. PP 288 sorption maximum at λ 517 nm, which is purple in color [18]. A radical scavenging antioxidant reacts with DPPH stable free radical, resulting in the decolorization which is stoichiometric with respect to the number of electrons captured [18]. The change on the absorbance produced in this reaction is used to measure antioxidant properties [18]. Stock solutions of different compounds (1 mM) were mixed with 0.5 ml of 0.3 mMDPPH in methanol. Final volume was adjusted to 3 ml. Reaction mixtures were variously shaken and allowed to react for 30 minutes in the dark at room temperature. Absorbance values were measured at 517 nm using anultraviolet-visible (UV-VIS) spectrophotometer. 0.5 ml of 0.3 mM DPPH diluted in 2.5 ml of methanol was used as control. Absorbance was converted to % antioxidant activity using the following equation: S (%) = 100 (A0 – AS)/A0 where A0 is the ab- sorbance of the control (containing all reagents except the test compound) and AS represents absorbance of the test compound. Results were compared with activity of vitamin C which was used as the standard. The data are summarized in Table 3. 7. Authors’ Disclosures of Potential Conflicts of Interest The authors indicated no potential conflicts of interest. REFERENCES [1] E. Grotewold, “The Science of Flavonoids,” Springer, New York, 2006. doi:10.1007/978-0-387-28822-2 [2] Y. M. Lin, Y. Zhou, M. Flavin, L. M. Zhou, W. Nie and F. C. Chen, “Chalcones and Flavonoids as Anti-Tuberculo- sis Agents,” Bioorganic & Medicinal Chemistry, Vol. 10, No. 8, 2002, pp. 2795-2802. doi:10.1016/S0968-0896(02)00094-9 [3] M. L. Go, X. Wu and X. L. Liu, “Chalcones: An Update on Cytotoxic and Chemoprotective Properties,” Current Medicinal Chemistry, Vol. 12, No. 4, 2005, pp. 483-499. [4] Z. Nowakowska, “A Review of Anti-Infective and Anti- Inflammatory Chalcones,” European Journal of Medicinal Chemistry, Vol. 42, No. 2, 2007, pp. 125-137. doi:10.1016/j.ejmech.2006.09.019 [5] S. Velavan, K. Nagulendran, R. Mahesh and V. Begum, “In Vitro Antioxidant Activity of Asparagus racemosus- root,” Pharmacognosy Magazine, Vol. 3, No. 9, 2007, pp. 26-33. [6] M. Okawa, J. Kinjo, T. Nohara and M. Ono, “DPPH (1,1-Diphenyl-2-picrylhydrazyl) Radical Scavenging Ac- tivity of Flavonoids Obtained from Some Medicinal Plants,” Biological and Pharmaceutical Bulletin, Vol. 24, No. 10, 2001, pp. 1202-1205. doi:10.1248/bpb.24.1202 [7] E. Cadenas and L. Packer, “Handbook of Antioxidants,” Marcel Dekker, New York, 1996. [8] C. Rice-Evans and L. Packer, “Flavonoids in Health and Disease,” Marcel Dekker, New York, 2003. [9] O. Prakash, R. Kumar and V. Parkash, “Synthesis and Antifungal Activity of Some New 3-Hydroxy-2-(1-phenyl- 3-aryl-4-pyrazolyl)chromones”, European Journal of Me- dicinal Chemistry, Vol. 43, No. 2, 2008, pp. 435-440. doi:10.1016/j.ejmech.2007.04.004 [10] O. Prakash, R. Kumar and R. Sehrawat, “Synthesis and Antibacterial Activity of Some New 2,3-Dimethoxy-3-hy- droxy-2-(1-phenyl-3-aryl-4-pyrazolyl)chromanones,” Eu- ropean Journal of Medicinal Chemistry, Vol. 44, No. 4, 2009, pp. 1763-1767. doi:10.1016/j.ejmech.2008.03.028 [11] B. Bandgar, S. Gawande, R. Bodade, N. Gawande and C. Khobragade, “Synthesis and Biological Evaluation of a Novel Series of Pyrazolechalcones as Anti-Inflammatory, Antioxidant and Antimicrobial Agents,” Bioorganic & Medicinal Chemistry, Vol. 17, No. 24, 2009, pp. 8168- 8173. doi:10.1016/j.bmc.2009.10.035 [12] H. Adibi, J. S. Mojarrad, H. Asgharloo and G. Zarrini, “Synthesis, in Vitro Antimicrobial and Antioxidant Ac- tivities of Chalcone and Flavone Derivatives Holding Al- lylic Substitutions,” Medicinal Chemistry Research, Vol. 20, No. 8, 2010, pp. 1318-1324. [13] N. S. Joshi, A. A. Shaikh, A. P. Deshpande, B. K. Karale, S. B. Bhirud and C. H. Gill, “Synthesis, Characterization and Antimicrobial Activities of Some Fluorine Contain- ing 2-(1-Phenyl-3-aryl-1H-pyrazol-4-yl)-3-chlorochromo- nes, 2-(1-Phenyl-3-aryl-1H-pyrazol-4-yl)chromones and 5-(1-Phenyl-3-aryl-1H-py razol-4-yl)-3-(2- hydroxypheny l)- 4,5-dihydropyrazolines,” Indian Journal of Chemistry, Vol. 44B, No. 2, 2005, pp. 422-425. [14] T. T. Dao, Y. S. Chi, J. Kim, H. P. Kim, S. Kim and H. Park, “Synthesis and PGE2 Inhibitor Activity of 5,7-Di- hydroxyflavones and Their O-Methylated Flavone Ana- logs,” Archives of Pharmacal Research, Vol. 26, No. 5, 2003, pp. 345-350. doi:10.1007/BF02976690 [15] M. Hiersemann and U. Nubbemeyer, “The Claisen Rear- rangement: Methods and Applications,” Wiley-VCH Ver- lag GmbH, Weinheim, 2007. [16] National Committee for Clinical Laboratory Standards (NCCLS), “Approved Standard Document M-7A,” Vil- lanova, P.A., 1985. [17] P. R. Murray, E. Baron, J. Jorgensen, M. Landry and M. Pfaller, “Manual of Clinical Microbiology,” 9th Edition, American Society of Microbiology, Washington DC, 2007. [18] K. Kato, S. Terao, N. Shimamoto and M. Hirata, “Studies on Scavengers of Active Oxygen Species. 1. Synthesis and Biological Activity of 2-O-Alkylascorbic Acids,” Journal of Medicinal Chemistry, Vol. 31, No. 4, 1988, pp. 793-798. doi:10.1021/jm00399a019 [19] Padmaja, T. Payani, G. D. Reddy and V. Padmavathi, “Synthesis, Antimicrobial and Antioxidant Activities of Substituted Pyrazoles, Isoxazoles, Pyrimidine and Thi- oxopyrimidine Derivatives,” European Journal of Me- dicinal Chemistry, Vol. 44, No. 11, 2009, pp. 4557-4566. doi:10.1016/j.ejmech.2009.06.024
|