20 H. X. WANG ET AL.
temperature sweep rate of 20˚C/min.
3. Results and Discussion
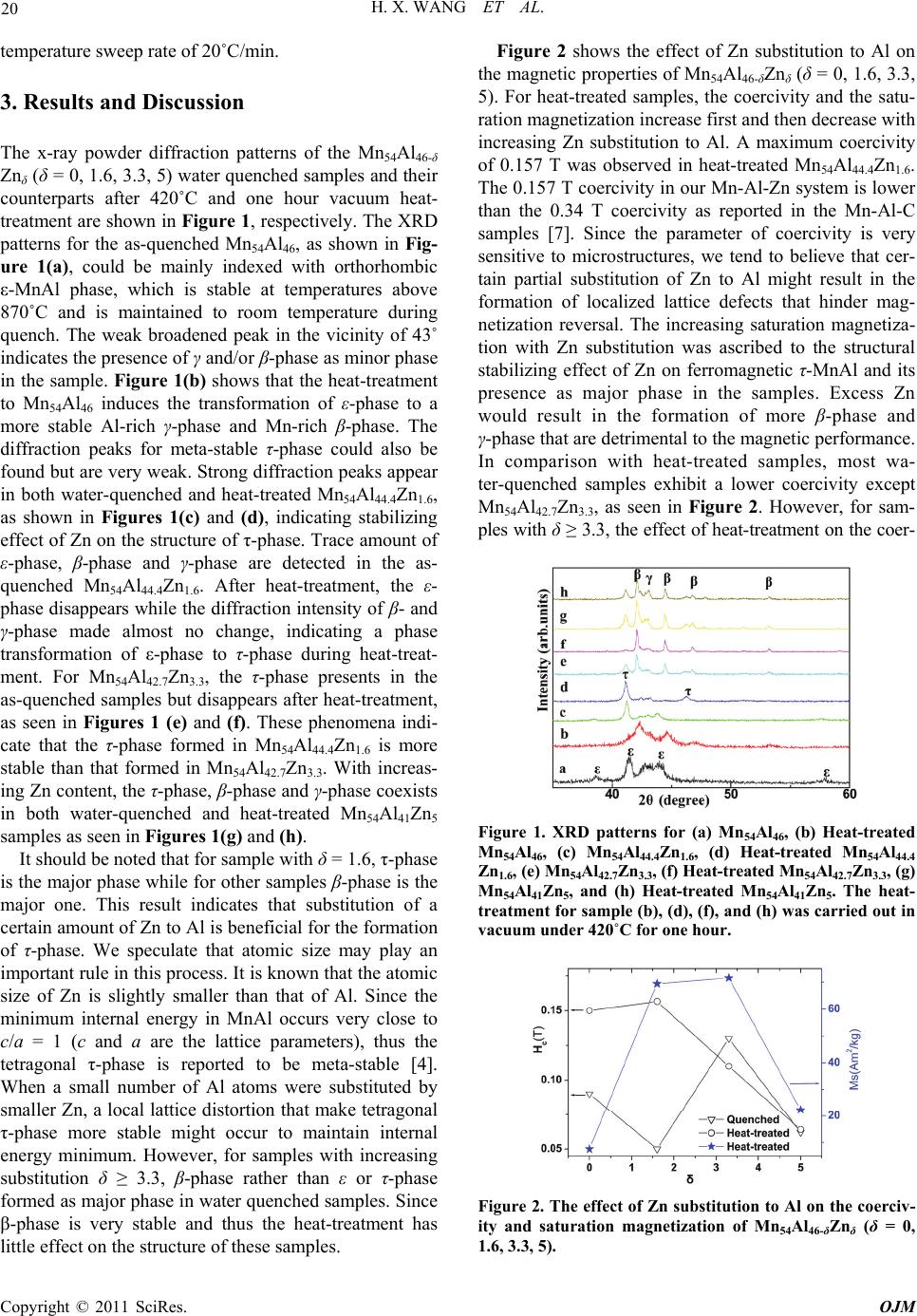
The x-ray powder diffraction patterns of the Mn54Al46-δ
Znδ (δ = 0, 1.6, 3.3, 5) water quenched samples and their
counterparts after 420˚C and one hour vacuum heat-
treatment are shown in Figure 1, respectively. The XRD
patterns for the as-quenched Mn54Al46, as shown in Fig-
ure 1(a), could be mainly indexed with orthorhombic
ε-MnAl phase, which is stable at temperatures above
870˚C and is maintained to room temperature during
quench. The weak broadened peak in the vicinity of 43˚
indicates the presence of γ and/or β-phase as minor phase
in the sample. Figure 1(b) shows that the heat-treatment
to Mn54Al46 induces the transformation of ε-phase to a
more stable Al-rich γ-phase and Mn-rich β-phase. The
diffraction peaks for meta-stable τ-phase could also be
found but are very weak. Strong diffraction peaks appear
in both water-quenched and heat-treated Mn54Al44.4Zn1.6,
as shown in Figures 1(c) and (d), indicating stabilizing
effect of Zn on the structure of τ-phase. Trace amount of
ε-phase, β-phase and γ-phase are detected in the as-
quenched Mn54Al44.4Zn1.6. After heat-treatment, the ε-
phase disappears while the diffraction intensity of β- and
γ-phase made almost no change, indicating a phase
transformation of ε-phase to τ-phase during heat-treat-
ment. For Mn54Al42.7Zn3.3, the τ-phase presents in the
as-quenched samples but disappears after heat-treatment,
as seen in Figures 1 (e) and (f). These phenomena indi-
cate that the τ-phase formed in Mn54Al44.4Zn1.6 is more
stable than that formed in Mn54Al42.7Zn3.3. With increas-
ing Zn content, the τ-phase, β-phase and γ-phase coexists
in both water-quenched and heat-treated Mn54Al41Zn5
samples as seen in Figures 1(g) and (h).
It should be noted that for sample with δ = 1.6, τ-phase
is the major phase while for other samples β-phase is the
major one. This result indicates that substitution of a
certain amount of Zn to Al is beneficial for the formation
of τ-phase. We speculate that atomic size may play an
important rule in this process. It is known that the atomic
size of Zn is slightly smaller than that of Al. Since the
minimum internal energy in MnAl occurs very close to
c/a = 1 (c and a are the lattice parameters), thus the
tetragonal τ-phase is reported to be meta-stable [4].
When a small number of Al atoms were substituted by
smaller Zn, a local lattice distortion that make tetragonal
τ-phase more stable might occur to maintain internal
energy minimum. However, for samples with increasing
substitution δ ≥ 3.3, β-phase rather than ε or τ-phase
formed as major phase in water quenched samples. Since
β-phase is very stable and thus the heat-treatment has
little effect on the structure of these samples.
Figure 2 shows the effect of Zn substitution to Al on
the magnetic properties of Mn54Al46-δZnδ (δ = 0, 1.6, 3.3,
5). For heat-treated samples, the coercivity and the satu-
ration magnetization increase first and then decrease with
increasing Zn substitution to Al. A maximum coercivity
of 0.157 T was observed in heat-treated Mn54Al44.4Zn1.6.
The 0.157 T coercivity in our Mn-Al-Zn system is lower
than the 0.34 T coercivity as reported in the Mn-Al-C
samples [7]. Since the parameter of coercivity is very
sensitive to microstructures, we tend to believe that cer-
tain partial substitution of Zn to Al might result in the
formation of localized lattice defects that hinder mag-
netization reversal. The increasing saturation magnetiza-
tion with Zn substitution was ascribed to the structural
stabilizing effect of Zn on ferromagnetic τ-MnAl and its
presence as major phase in the samples. Excess Zn
would result in the formation of more β-phase and
γ-phase that are detrimental to the magnetic performance.
In comparison with heat-treated samples, most wa-
ter-quenched samples exhibit a lower coercivity except
Mn54Al42.7Zn3.3, as seen in Figure 2. However, for sam-
ples with δ ≥ 3.3, the effect of heat-treatment on the coer-
Figure 1. XRD patterns for (a) Mn54Al46, (b) Heat-treated
Mn54Al46, (c) Mn54Al44.4Zn1.6, (d) Heat-treated Mn54Al44.4
Zn1.6, (e) Mn54Al42.7Zn3.3, (f) Heat-treated Mn54Al42.7Zn3.3, (g)
Mn54Al41Zn5, and (h) Heat-treated Mn54Al41Zn5. The heat-
treatment for sample (b), (d), (f), and (h) was carried out in
vacuum under 420˚C for one hour.
Figure 2. The effect of Zn substitution to Al on the coerciv-
ity and saturation magnetization of Mn54Al46-δZnδ (δ = 0,
1.6, 3.3, 5).
Copyright © 2011 SciRes. OJM