Paper Menu >>
Journal Menu >>
 Vol.1, No.2, 33-40 (2011) Open Journal of Animal Sciences doi:10.4236/ojas.2011.12005 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ Characterization of proteins in cryopreserved and non-cryopreserved seminal plasma of dairy bulls of differing fertility ——Seminal plasma proteomics J. F. Odhiambo1,2, R. A. Dailey1* 1Division of Animal and Nutritional Sciences, West Virginia University, Morgantown, USA; *Corresponding Author: rdailey@wvu.edu 2Department of Agricultural, Food and Nutritional Science, University of Alberta, Edmonton, Canada; Received 6 May 2011, revised 25 May 2011, accepted 10 June 2011. ABSTRACT Seminal plasma is composed of secretions from accessory sex glands, which are mixed with sperm at ejaculation and contribute the majority of semen volume. Seminal plasma is considered a transport and support medium for sperm in the female reproductive tract. Because seminal plasma is not required for fertilization, the im- portance of its constituents to the establishment of normal pregnancy has been overlooked. Four seminal plasma proteins, Osteopontin, Sper- madhesin Z13, BSP 30 kDa and Phospholipase A2, have been identified as markers of fertility in dairy bulls [1-3]. The objective of the present study was to characterize the expression pat- terns of these proteins and other proteins found to be of interest in seminal plasma of cryopre- served and non-cryopreserved bull semen. Seminal plasma samples were obtained from 16 mature Holstein- Friesian bulls at Select Sires Inc. Samples were divided into two groups based on assigned fertility score expressed as the percentage point deviation (PD) of the bull’s non-return rate (NRR) from the average NRR of all bulls in the Select Sires Inc. reproductive management program. Group 1 (high fertility bulls, n = 8) 1.9% ≤ PD ≤ 2.7%, and group 2 (low fertility bulls, n = 8) - 6.5% ≤ PD ≤ 1.8%. Addi- tionally, the samples were categorized as proc- essed (cryopreserved) or unprocessed (non- cryopreserved) for protein analysis. Protein ex- pression was analyzed by 2 - D fluorescence difference gel electrophoresis (2D-DIGETM). Pro- tein spots were picked from a reference gel, analyzed by mass spectrometry and, subse- quently identified by MS/MS ion searches per- formed on the SwissProt database. Protein ex- pression did not differ (P > 0.05) with fertility grouping but displayed two distinct pa- tterns among the processing groups: majority of the functional proteins were highly expressed in seminal plasma of non-cryopreserved semen while the cryopreserved semen contained mainly structural/ extender derived proteins. Functional proteins identified included Spermadhesin Z13, BSP A1/2, BSP 30 kDa, Nucleobindin-1 and metalloproteinase inhibitor 2. Some of these proteins have been identified as anti-fertility or fertility enhancing agents in males. Whether this alteration in protein expression after processing might affect semen fertility is worthy of further evaluation. Keywords: Seminal Plasma; Proteomics; Bull Fertility 1. INTRODUCTION Secretions from the accessory sex glands are mixed with sperm at ejaculation and contribute to the majority of semen volume and components. However, during cryopreservation, most seminal plasma is replaced with semen extenders, mainly egg yolk or milk proteins. Se- minal fluid contains signaling agents that influence fe- male reproductive physiology to improve chances of conception and pregnancy success. These factors include cytokines, sex hormones, and prostaglandins [4]. Ex- periments in rodents and pigs showed that seminal plasma is the second most vital component of the ejacu- late, absence of which at mating reduced fertilization and increased fetal loss after implantation [5]. Artificial inseminations with adjunctive seminal plasma consis- tently tended to have little or no effect on pregnancy outcome [6]. The precise nature of active constituents  J. F. Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 34 that might influence pregnancy and their relative amounts remain relatively unknown. High concentrations of TGF-β cytokines were de- tected in boar seminal fluid [7], and their characteristic immunosuppressive activity was associated with protein fractions of appropriate size in boar seminal fluid [4]. Four proteins (osteopontin, spermadhesin Z13, bovine seminal plasma protein (BSP) 30 kDa, and phospholi- pase A2) were identified as markers of fertility in dairy bulls [1-3]. In those studies, proteins were visualized with Coomassie brilliant blue staining after 2-D gel separation, a process that might not reveal some of the low abundance proteins in seminal plasma that might be of biologic importance. Therefore, a more sensitive protein detection tech- nique that would give a broader approach was used in the present study to identify seminal plasma proteins. The multiplexing capability of 2 - D fluorescence dif- ference gel electrophoresis (2 - D DIGE) technology was expected to broaden the number of markers included in the assay and yield more robust predictions of bio- markers for bull fertility. The objectives of this study included: 1) large scale identification and differential expression of seminal plasma proteins between high and low fertility bulls, 2) correlation of expression of spe- cific proteins to fertility phenotype, and 3) comparison of the expression patterns pre- and post-cryopreservation. 2. MATERIALS AND METHODS Semen samples from 16 dairy bulls were obtained from Select Sires Inc. (Plain City, Ohio). All samples used (cryopreserved and non-cryopreserved) were ob- tained from the same ejaculate of each bull and proc- essed at Select Sires Inc. At each collection time, ejacu- lates from the same bull were pooled and an aliquot was obtained for the non-cryopreserved sample. The rest of the semen was processed and extended for cryopreserva- tion in 0.5 mL semen straws. Semen was extended in egg-yolk-citrate (EYC) com- posed of a 2.9% sodium citrate buffer supplemented with 20% egg yolk (vol/vol) prepared with a glycer- olated and non-glycerolated fractions containing 14 or 0% glycerol (vol/vol), respectively. Samples were di- luted 1:3 (vol/vol) with non-glycerolated semen extender, placed in a 200-mL water bath and allowed to equilibrate to 5˚C over a 2-h period. Samples were extended to half volume (80 × 106/ml) in non-glycerolated extender and then an equal volume of glycerolated extender was slowly titrated over a 10 min period. The final dilution yielded a sperm concentration of 20 × 106 per 0.5-mL French straw. Straws were then frozen in liquid nitrogen. To obtain seminal plasma, semen was centrifuged at 3,000 g for 10 min and then aspirated into Eppendorf tubes for volumes < 1 mL, or 15 mL centrifuge tubes for larger volumes. Samples were then further clarified in 0.45 micron syringe filter and finally frozen in 0.5 mL straws. Samples were assigned to high or low fertility groups based on fertility score expressed as the percentage point deviation (PD) of the bull’s non-return rate (NRR) from the average NRR of all bulls in the Select Sires Inc. re- productive management program. These scores were based on inseminations with frozen semen from the bulls under this program. Range of scores for high fertility bulls (n = 8) was 1.9% ≤ PD ≤ 2.7% and for low fertility bulls (n = 8) was –6.5% ≤ PD ≤ 1.8%. Following delivery, semen samples were thawed at room temperature and centrifuged at 10,000 × g for 15 min at 4˚C to remove sperm and associated debris. Ali- quots of 0.5 mL seminal samples were obtained from the supernatant and stored frozen at –80˚C until further use. 2.1. Electrophoresis Frozen samples of seminal plasma were thawed at room temperature and centrifuged at 10,000 × g for 60 min at 4˚C. The supernatant was processed by 2-D Clean-UP protocol (GE Healthcare, Piscataway, NJ) to remove impurities such as nucleic acids, lipids and salts. Samples were then assayed for protein content [8] using BSA as standard, and aliquots were frozen at –800˚C. Samples for electrophoresis were thawed at room tem- perature, concentrated to 1 - 10 mg protein/mL and la- beled with cyanine dye (CyDye) DIGE Fluor Cy3/5 (GE Healthcare, Piscataway, NJ) at a ratio of 50 µg protein to 400 pmol fluor. A pooled internal standard was created from equal aliquots of each sample and labeled with Cy2 dye. Samples were separated by isoelectric focusing on an Ettan™ IPGphor™ apparatus (GE Healthcare, Pis- cataway, NJ) using 24 cm Immobiline DryStrip gels (GE Healthcare, Piscataway, NJ) containing a mixture of ampholytes with pH ranging from 3 to 10. Following isoelectric focusing, strip gels were trans- ferred to 24 cm Tris-Tricine gradient gels (Bio-Rad La- boratories, Hercules, CA) mounted on low-fluorescence glass plates. Thereafter, proteins were separated by mo- lecular mass in the second dimension using Ettan™ Dalt II Electrophoresis System (GE Healthcare, Piscataway, NJ). Dalt gels were scanned using a Typhoon 9400 Va- riable Mode imaging densitometer (GE Healthcare, Pis- cataway, NJ) at 100 µm resolution. A fully automated image analysis software, Progenesis SameSpots™ (Non- linear Dynamics, Durham, NC), was used to analyze the protein expression data. 2.2. Statistical Analysis In 2D-DIGE experiments, the pooled internal stan-  J. F.Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 3535 dards were essential for assessing biological and ex- perimental (between gels) variations and increasing the robustness of statistical analysis. Individual protein data from sample groups (Cy3 or Cy5) were normalized against the Cy2 labeled internal standard. Scanned im- ages of the labeled proteins were sequentially analyzed by differential in-gel analysis (DIA) that performed Cy3/Cy5:Cy2 normalization, and then by biological var- iation analysis (BVA) that performed inter-gel statistical analysis to provide relative abundance in various groups. Log abundance ratios were then compared between sample groups using ANOVA and t-test in Progenesis SameSpots. The analyzed spots were ranked by their probability values and then grouped into experimental groups for further evaluation. Principal component analysis (PCA) was used to determine the presence of outliers in the data and also to compare how well the samples fitted to the experimental groups. The expres- sion profiles of the selected proteins were then examined by correlation analysis. 2.3. Protein Identification A list of protein spots of interest (pick-list) was gener- ated using the image analysis software and exported di- rectly into a Spot Picking Ettan™ Spot Handling Work- station (GE Healthcare, Piscataway, NJ) equipped to automatically pick spots from the Dalt gels. Selected protein spots were washed by 50 mM ammonium bicar- bonate/50% (vol/vol) methanol in water, dried by vac- uum centrifugation, and incubated overnight at 37 0C in 140 ng of sequencing grade trypsin [9]. Tryptic digests were analyzed by capillary liquid chromatography- nanoelectrospray ionization-tandem mass spectrometry (CapLC-MS/MS, Thermo Finnigan, San Jose, CA). Pro- teins were identified by MS/MS ion searches performed on the processed spectra against the SwissProt and NCBI protein databases using a Bioworks Browser 3.1 (Thermo Finnigan, San Jose, CA) search engine. The identification of a protein was confirmed when the Bio- works confidence interval was greater than 95%. The protein mass and pI accuracy on the 2D gel was used as a guide to confirm protein identification. 3. RESULTS 3.1. Distribution of Protein Spots in Seminal Plasma of Dairy Bulls Three patterns of protein expression were observed consistently in seminal plasma samples of cryopreserved semen (Figure 1). Spot volume analysis and peptide identification indicated a higher expression of proteins from semen extender at the molecular weight range be- tween 30 to 60 kDa (Group 1) accounting for 55% of protein spots by Coomassie staining. A “train” of spots was visible within the 20 to 25 kDa range (Group 2) accounting for about 35% of the spot volume, while the remaining spots (10%) were expressed below the 20 kDa range (Group 3). There was a two-fold difference (P < 0.01) in total protein content between extended and non-extended se- minal plasma (41.6 ± 2.3 vs. 19.5 ± 2.1 ng/mL, respec- tively). However, the expression patterns of proteins in seminal plasma of high and low fertility bulls did not differ (P > 0.05). Therefore, subsequent analyses were done between cryopreserved (processed) and non-cryopre- served (unprocessed) seminal plasma. A total of 54 spots differed (P < 0.001) in expression pattern between proc- essed and unprocessed seminal plasma (Figure 2(a)). The spots were then examined by principal component analysis and clustered into two groups: those 31 spots (57.4%) that were highly expressed in processed seminal plasma but not in unprocessed seminal plasma and those 23 spots (42.6%) that were highly expressed in unproc- essed seminal plasma but not in processed seminal plasma (Figure 2(b)). 3.2. Protein Identification A reference image (Figure 3) was generated from the expression data and used to pick spots for protein identi- fication (Table 1). Extender derived proteins, mainly chicken vitellogenin-2 (MW 20.5 kDa), fibrinogen β chain (MW 52.70) and chicken albumin, were predomi- nant proteins identified from spots above 20 kDa in se- minal plasma of processed semen. In unprocessed semi- nal plasma, which lacked extender proteins, several spots above 20 kDa were identified. Notable among these proteins were nucleobindin-1, clusterin, phosphol- ipase A2 isoforms, seminal plasma protein BSP-30 kDa, metalloproteinase inhibitor-2 and cathepsins (B and D). Below 20 kDa range, greater amounts of spermadhesin (SPAD1 and Z13) isoforms were expressed than the ma- jor bovine seminal plasma proteins (PDC - 109 and BSP-A3) in seminal plasma of processed semen. How- ever, in unprocessed seminal plasma, PDC - 109 and BSP-A3 predominated over the spermadhesins below the 20 kDa range. 4. DISCUSSION The main finding of this study was that expression pattern of seminal plasma proteins differed between, cryopreserved and non-cryopreserved dairy bull semen. Indeed, during cryopreservation, major seminal plasma proteins are replaced with extender proteins. Most artifi- cial inseminations in cattle involve use of processed (cryopreserved) semen, and as a consequence, most fer- tility data have been derived from inseminations with 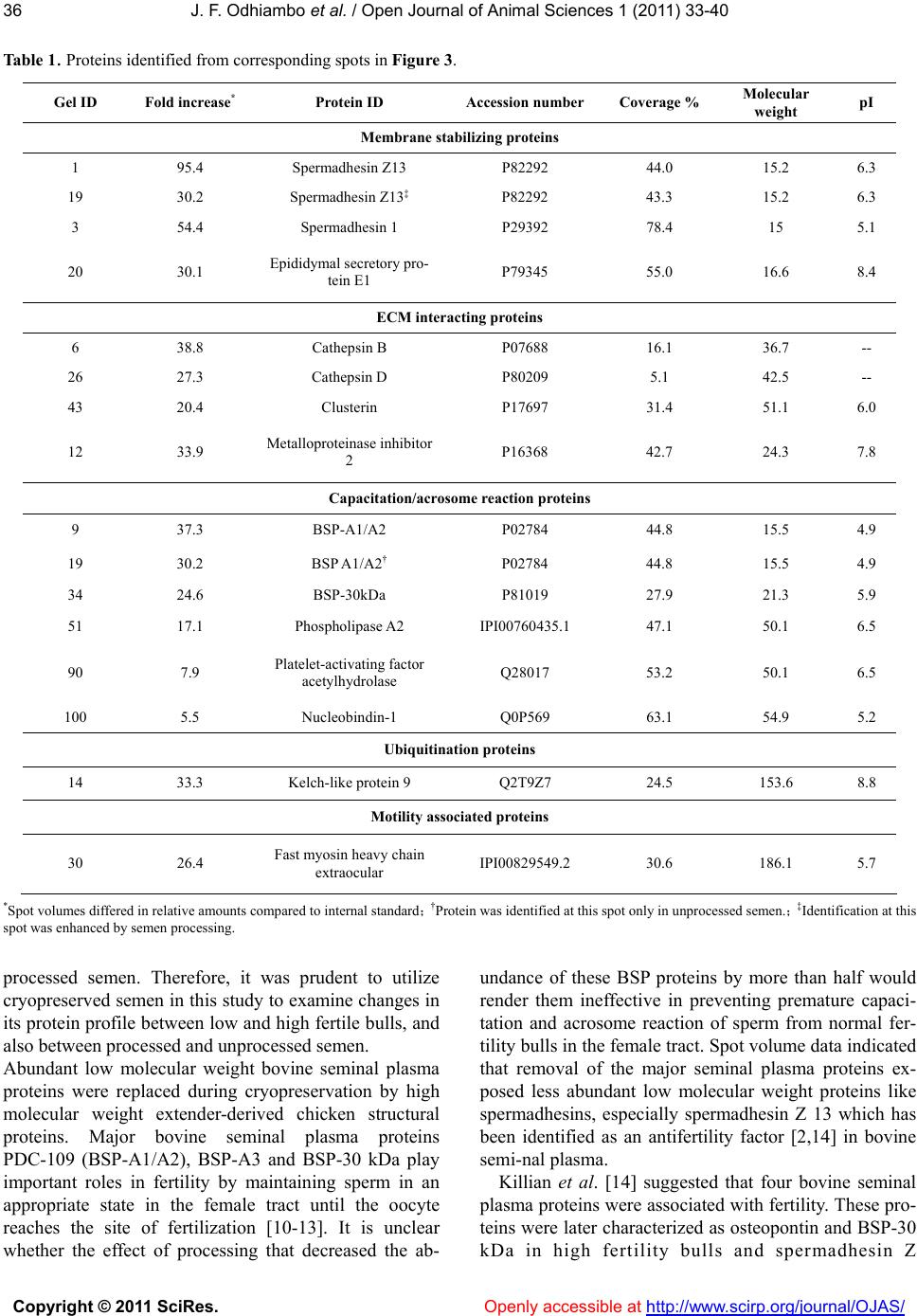 J. F. Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 36 Table 1. Proteins identified from corresponding spots in Figure 3. Gel ID Fold increase* Protein ID Accession number Coverage % Molecular weight pI Membrane stabilizing proteins 1 95.4 Spermadhesin Z13 P82292 44.0 15.2 6.3 19 30.2 Spermadhesin Z13‡ P82292 43.3 15.2 6.3 3 54.4 Spermadhesin 1 P29392 78.4 15 5.1 20 30.1 Epididymal secretory pro- tein E1 P79345 55.0 16.6 8.4 ECM interacting proteins 6 38.8 Cathepsin B P07688 16.1 36.7 -- 26 27.3 Cathepsin D P80209 5.1 42.5 -- 43 20.4 Clusterin P17697 31.4 51.1 6.0 12 33.9 Metalloproteinase inhibitor 2 P16368 42.7 24.3 7.8 Capacitation/acrosome reaction proteins 9 37.3 BSP-A1/A2 P02784 44.8 15.5 4.9 19 30.2 BSP A1/A2† P02784 44.8 15.5 4.9 34 24.6 BSP-30kDa P81019 27.9 21.3 5.9 51 17.1 Phospholipase A2 IPI00760435.1 47.1 50.1 6.5 90 7.9 Platelet-activating factor acetylhydrolase Q28017 53.2 50.1 6.5 100 5.5 Nucleobindin-1 Q0P569 63.1 54.9 5.2 Ubiquitination proteins 14 33.3 Kelch-like protein 9 Q2T9Z7 24.5 153.6 8.8 Motility associated proteins 30 26.4 Fast myosin heavy chain extraocular IPI00829549.2 30.6 186.1 5.7 *Spot volumes differed in relative amounts compared to internal standard; †Protein was identified at this spot only in unprocessed semen.; ‡Identification at this spot was enhanced by semen processing. processed semen. Therefore, it was prudent to utilize cryopreserved semen in this study to examine changes in its protein profile between low and high fertile bulls, and also between processed and unprocessed semen. Abundant low molecular weight bovine seminal plasma proteins were replaced during cryopreservation by high molecular weight extender-derived chicken structural proteins. Major bovine seminal plasma proteins PDC-109 (BSP-A1/A2), BSP-A3 and BSP-30 kDa play important roles in fertility by maintaining sperm in an appropriate state in the female tract until the oocyte reaches the site of fertilization [10-13]. It is unclear whether the effect of processing that decreased the ab- undance of these BSP proteins by more than half would render them ineffective in preventing premature capaci- tation and acrosome reaction of sperm from normal fer- tility bulls in the female tract. Spot volume data indicated that removal of the major seminal plasma proteins ex- posed less abundant low molecular weight proteins like spermadhesins, especially spermadhesin Z 13 which has been identified as an antifertility factor [2,14] in bovine semi-nal plasma. Killian et al. [14] suggested that four bovine seminal plasma proteins were associated with fertility. These pro- teins were later characterized as osteopontin and BSP-30 kDa in high fertility bulls and spermadhesin Z  J. F.Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 3737 Figure 1. Reference 2D gel depicting distribution of protein spots in seminal plasma of dairy bulls. Protein spots were characterized as: 1) probable extender-derived proteins, 2) probable medium and high molecular weight seminal plasma proteins, and 3) major bovine seminal plasma proteins. (a) 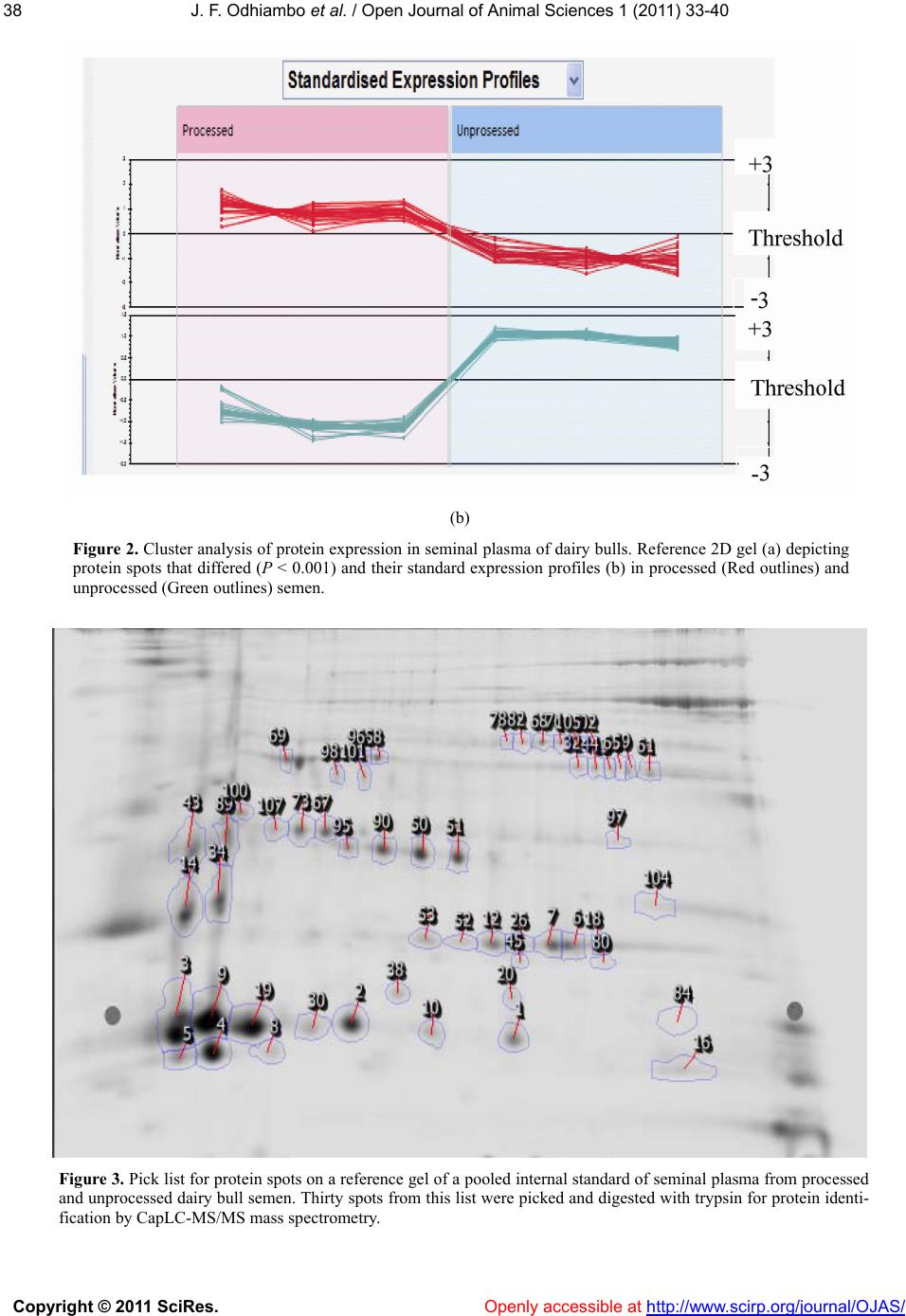 J. F. Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 38 (b) Figure 2. Cluster analysis of protein expression in seminal plasma of dairy bulls. Reference 2D gel (a) depicting protein spots that differed (P < 0.001) and their standard expression profiles (b) in processed (Red outlines) and unprocessed (Green outlines) semen. Figure 3. Pick list for protein spots on a reference gel of a pooled internal standard of seminal plasma from processed and unprocessed dairy bull semen. Thirty spots from this list were picked and digested with trypsin for protein identi- fication by CapLC-MS/MS mass spectrometry.  J. F.Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 3939 13 in low fertility bulls [2]. In the present study, seminal plasma protein expression did not differ between high and low fertility dairy bulls. A difference in protein ex- pression between fertility groups had been demonstrated in a previous study [2]. It was anticipated that by utiliz- ing the multiplexing capability of the 2-DIGE technol- ogy, experimental errors would be minimized, and a more robust analysis would be achieved as opposed to the densitometric analysis utilized in the former study. The discrepancy in outcome between the two studies can be attributed to sample type utilized or fertility grouping, or both. Samples in the present study were from proc- essed insemination straws and unprocessed seminal plasma from the same ejaculates as opposed to fresh seminal plasma samples utilized in the previous study that avoided secretions from epididymis and vas diferens. Therefore, differential expression of proteins in samples examined in this study might have been affected by sample type as well as semen processing as evidenced by the comparison of processed and unprocessed semi- nal plasma protein profile. The narrow range of fertility differences in the bulls used in this trial also impeded the classification of semen samples into distinct high and low fertile groups as was done in the previous study [2]. Bulls used in the present study had percentage point de- viations (PDs) from the average of +2.7% to –6.5%. In contrast, the previous study had PDs from +7.7% to –18.1%. Consequently, low and high fertility groups in the present study corresponded to the intermediate fertil- ity groups in that study. Because the previous authors reported no differences in expression levels of osteopon- tin, spermadhesin Z13, phospholipase A2 and BSP 30kDa among these two groups, it was not surprising that the groups did not differ in their seminal plasma protein profile in the present study. This is a major lim- iting factor in fertility studies in farm animals because it is rare to find sires at the lower extremes of fertility due selection pressure. Seminal plasma proteins have been characterized by other investigators and their association with male fertil- ity continues to be explored [2,3,15-17]. Functions of sperm that may be affected by seminal plasma proteins include capacitation, acrosome reaction, motility, DNA integrity and interaction with the oocyte [3,18]. Major BSP proteins (BSP-A1/A2, A3 and BSP-30 kDa) are known to influence capacitation by their ability to modulate membrane cholesterol [18]. Phospholipase A2 (PLA2) and osteopontin are involved in acrosome reaction and sperm-oocyte interaction and possibly early embryonic development [3]. Proteins that might be asso- ciated with interaction and modulation of extracellular matrix (ECM) components are TIMP-2, clusterin and cathepsins. These functions may be important during fertilization when the sperm is required to interact with and cross barriers established by the cumulus cells, zona pellucida and oocyte membrane. Albumin, aSFP and clusterin are involved either directly or indirectly in mechanisms aimed at preventing damage to sperm membrane, oxidative stress, and immune attack. Proteins associated with sperm motility in the female reproduc- tive tract include BSP A1/A2, aSFP, PLA2 and ecto 5'-nucleotidase (5'-NT). Spermadhesin Z13 might also be included in the motility associated group because it shares 50% homology with aSFP. However, expression of spermadhesin Z13 in seminal plasma of dairy bulls was inversely related to fertility [2]. 5. CONCLUSIONS Although expression pattern of seminal proteins was not associated with fertility ranking in this study, altera- tion of protein expression might affect fertility in bulls that show more divergence in fertility ranking. In addi- tion, expression patterns of seminal plasma proteins might be altered during cryopreservation as has been demonstrated in this study. Whether this alteration might affect fertility remains to be explored. 6. ACKNOWLEDGEMENTS The authors acknowledge the support of Select Sires Inc for provi- sion of fertility data and semen samples. We greatly acknowledge the support of Ms. Linda Corrum, Mr. Steve Wolf and Dr. Peter Perotta of the Core Proteomics facility at West Virginia University. This work is published with the approval of the director of the WV Agriculture and Forestry Experiment Station as a scientific paper from the Division of Animal and Nutritional Sciences and was supported by Hatch Project 421, NE1007 and Select Sires grant. REFERENCES [1] Cancel, A.M., Chapman, D.A. and Killian, G.J. (1997) Osteopontin is the 55-kilodalton fertility-associated pro- tein in Holstein bull seminal plasma. Biology of Repro- duction, 57, 1293-1301. [2] Moura, A.A., Koc H., Chapman D.A. and Killian G.J. (2006) Identification of proteins in the accessory sex gland fluid associated with fertility indexes of dairy bulls: a proteomic approach. Journal of Andrology, 27, 201-211. doi:10.2164/jandrol.05089 [3] Moura, A.A., Chapman, D.A. and Killian, G.J. (2007) Proteins of the accessory sex glands associated with the oocyte-penetrating capacity of cauda epididymal sperm from holstein bulls of documented fertility. Molecular Reproduction & Development, 74, 214-222. doi:10.1002/mrd.20590 [4] Robertson, S.A. (2007) Seminal fluid signaling in the female reproductive tract: Lessons from rodents and pigs. Journal of Animal Science, 85, E36-44. doi:10.2527/jas.2006-578  J. F. Odhiambo et al. / Open Journal of Animal Sciences 1 (2011) 33-40 Copyright © 2011 SciRes. Openly accessible at http://www.scirp.org/journal/OJAS/ 40 [5] O, W.S., Chen, H.Q. and Chow P.H. (1998) Effects of male accessory sex gland secretions on early embryonic development in the golden hamster. Journal of Repro- duction Fertility, 84, 341-344. [6] Odhiambo, J.F., Poole, D.H., Hughes, L., DeJarnette, M., Inskeep, E.K. and Dailey, R.A. (2009) Pregnancy out- come in dairy and beef cattle after artificial insemination and treatment with seminal plasma or transforming growth factor beta-1. Theriogenology, 72, 566-571. doi:10.1016/j.theriogenology.2009.04.013 [7] O’Leary, S., Robertson, S.A. and Armstrong, D.T. (2002) The influence of seminal plasma on ovarian function in pigs—a novel inflammatory mechanism? Journal of Re- productive Immunology, 57, 225-238. [8] Lowry, O.H., Rosebrough, N.J., Farr, A. L. and Randall, R.J. (1951) Protein measurement with the Folin-Phenol reagents. The Journal of Biological Chemistry, 193, 265-275. [9] Koc, E.C., Burkhart, W., Blackburn, K., Koc, H., Mose- ley, A. and Spremulli L.L. (2001) Identification of four proteins from the small subunit of the mammalian mito- chondrial ribosome using a proteomics approach. Protein Science, 10, 471-481. [10] Thérien, I., Soubeyrand, S. and Manjunath, P. (1997) Major proteins of bovine seminal plasma modulate sperm capacitation by high-density lipoprotein. Biology of Re- production, 57, 1080-1088. doi:10.1095/biolreprod57.5.1080 [11] Thérien, I., Moreau, R. and Manjunath, P. (1998) Major proteins of bovine seminal plasma and high-density lipo- protein induce cholesterol efflux from epididymal sperm. Biology of Reproduction, 59, 768-776. doi:10.1095/biolreprod59.4.768 [12] Yu, B., Zhao, Y., Zhao, W., Chen, F., Liu, Y., Zhang, J., Fu, W., Zong, Z., Yu, A. and Guan, Y. (2003) The inhibi- tory effect of BSP-A1/-A2 on protein kinase C and tyro- sine protein kinase. Cell Biochemistry and Function, 21, 183-188. doi:10.1002/cbf.1018 [13] Gwathmey, T.M., Ignotz, G.G., Mueller, J.L., Manjunath, P. and Suarez S.S. (2006) Bovine seminal plasma pro- teins PDC-109, BSP-A3, and BSP-30-kDa share func- tional roles in storing sperm in the oviduct. Biology of Reproduction, 75, 501-507. doi:10.1095/biolreprod.106.053306 [14] Killian, G.J., Chapman, D.A. and Rogowski, L.A. (1993) Fertility-associated proteins in Holstein bull seminal plasma. Biology of Reproduction, 49, 1202-1207. doi:10.1095/biolreprod49.6.1202 [15] Mortarino, M., Tedeschi, G., Negri, A., Ceciliani, F., Gottardi, L, Maffeo, G. and Ronchi, S. (1998) Two-di- mensional polyacrylamide gel electrophoresis map of bull seminal plasma proteins. Electrophoresis, 19, 797- 801. doi:10.1002/elps.1150190532 [16] Gwathmey, T.M., Ignotz, G.G. and Suarez, S.S. (2003) PDC-109 (BSP-A1/A2) promotes bull sperm binding to oviductal epithelium in vitro and may be involved in forming the oviductal sperm reservoir. Biology of Re- production, 69, 809-815. doi:10.1095/biolreprod.102.010827 [17] Jobim, M.I., Oberst, E.R., Salbego, C.G., Souza, D.O., Wald, V.B., Tramontina, F. and Mattos, R.C. (2004) Two- dimensional polyacrylamide gel electrophoresis of bo- vine seminal plasma proteins and their relation with se- men freezability. Theriogenology, 61, 255-266. doi:10.1016/S0093-691X(03)00230-9 [18] Manjunath, P., Bergeron, A., Lefebvre, J. and Fan, J. (2007) Seminal plasma proteins: Functions and interac- tion with protective agents during semen preservation. Society of Reproduction and Fertility Supplement, 65, 217-228. |

