Open Journal of Clinical Diagnostics
Vol.4 No.1(2014), Article ID:43746,4 pages DOI:10.4236/ojcd.2014.41006
Cardiac Amyloidosis: The Role of Magnetic Resonance Imaging
Linda Sim, Christopher Semsarian, David Celermajer, Rajesh Puranik
Department Cardiology, Royal Prince Alfred Hospital, Sydney, Australia
Email: raj.puranik@cmrs.org.au
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/


Received 1 January 2014; revised 1 February 2014; accepted 10 February 2014
ABSTRACT
Amyloidosis, an infiltrative disease characterized by deposition of high molecular weight insoluble protein has a low detection rate and poor prognosis with congestive heart failure being the most common cause of death. Although definitive diagnosis is based on endomyocardial biopsy, this is rarely performed due to procedural risk and sampling error. Traditional non-invasive imaging, such as transthoracic echocardiography (TTE) and electrocardiogram (ECG) lacks sensitivity detecting the disease in its early stages. This case report demonstrates the utility of cardiac Magnetic Resonance Imaging in addition to traditional imaging modalities in this often clinically challenging problem.
Keywords:Cardiac Amyloidosis; Cardiac Magnetic Resonance Imaging (CMR); Transthoracic Echocardiography (TTE); Electrocardiography (ECG); Congestive Cardiac Failure (CCF)

1. Introduction
Amyloidosis is an infiltrative disease leading to either systemic or localized deposition of an extracellular high molecular weight insoluble protein, known as amyloid. Congestive heart failure and arrhythmia are the most common clinical presentations and amyloid can lead to pathologic thickening of cardiac structures and restrictive cardiomyopathy [1] . Cardiac amyloidosis (CA) is usually diagnosed late and in the disease course and therapies are relatively ineffective, leading to poor prognosis, with a median survival of less than 6 months after the onset of heart failure [2] [3] . Although definitive diagnosis is based on endomyocardial biopsy, this is rarely performed due to procedural risk and the possibility of sampling error [1] [4] . Traditionally, non-invasive methods of diagnosis such as transthoracic echocardiography (TTE) and electrocardiogram (ECG) [5] , may lack sensitivity in the early stages of the disease. The advent of Late Gadolinium-Enhancement studies using Cardiovascular Magnetic Resonance Imaging (LGE-CMRI) and T2 weighted imaging aids diagnosis of CA due to its superior ability to perform myocardial tissue characterization [5] . We demonstrate the utility of such imaging in addition to traditional imaging modalities in this often clinically challenging problem.
2. Case Report
2.1. Case 1—Primary Cardiac Amyloidosis
An 85-year-old female patient presented with blood stained sputum and complained of episodes of dyspnoea but no chest pain. Her past cardiac history included known primary cardiac amyloid confirmed by identification of the transthyretin gene mutation diagnosed 12 months prior along with an incidental finding of a right atrial myxoma. On examination she had HR of 100 bpm, BP of 133/79, oxygen saturation of 96% - 98% and signs of cardiac failure. Her ECG showed sinus rhythm with mild intra-ventricular conduction defect (Figure 1(A)). Recent blood results showed a raised C reactive protein of 16.6mg/L and platelet count of 570 × 10^9/L. Her most recent TTE demonstrated the development of moderate concentric LV hypertrophy with septal and posterior wall thickness of 14mm compared to a corresponding wall thickness of 9 mm in 2008 and preserved systolic function [6] . The E/A ratio was reversed, consistent with diastolic dysfunction. Additionally, the right atrial mxyoma was poorly visualised. CMRI demonstrated severe hypertrophy affecting the basal septal wall (20 mm), inferior wall (24 mm) and posterolateral wall (16 mm) (Figure 1(B)). LV systolic function was preserved with EF of 60% [6] . There was inhomogeneous increase of myocardial signal on T2 weighted imaging along with patchy diffuse hyper-intense LGE in the LV septum and postero-lateral walls (Figure 1(C)). The RV was normal size with normal systolic function and unusual patchy hypertrophy of the basal RV wall. These findngs were consistent with CA, specifically demonstrating features of a severe phenotype. The incidental right atrial myxoma measuring 8 mm × 12 mm was well visualised and attached to the interatrial septum within the RA. The patient had improvement clinically with antibiosis for suspected intercurrent pneumonia and gentle IV diuresis. The right atrial myxoma was thought to be slow growing, immobile and non-obstructive and hence left for surveillance and not surgical excision. The prognosis for this patient was guarded on the basis of the progression of LV hypertrophy and infiltration shown on the CMR.
2.2. Case 2—Secondary Cardiac Amyloidosis
A 42-year-old female patient presented with dyspnoea for the past 1 - 2 months with clinical signs of bilateral effusions. Her past medical history included scleroderma/ dermatomyosistis syndrome in 1999 and Raynaud’s phenomenon, ECGs showed LV hypertrophy with repolarisation abnormality (Figure 2(A)) and blood test results showed elevated C reactive protein (99.2 mg/L), protein/ creatinine ratio (67.6 mg/ mmol) and Ur protein of 0.25 g/ L. These blood results indicated chronic inflammatory disease and the pattern of urinary protein loss was consistent with non-selective glomerular proteinuria. There was also elevated free lambda light chain (34.10 mg/ L) with normal Lambda/ Kappa ratio (0.48) along with positive immunofixation reaction, consistent with systemic secondary amyoidosis [2] [7] . TTE revealed normal LV cavity size with severely increased wall thick
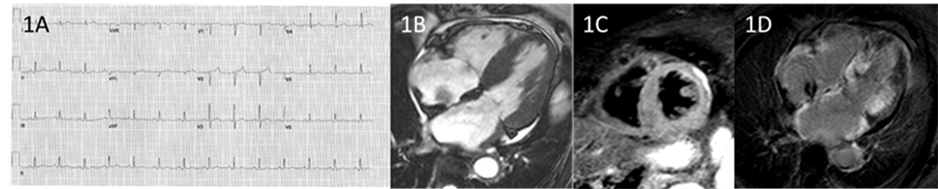
Figure 1. (A) 12 lead ECG with normal sinus rhythm with reduced amplitude in QRS complexes; (B) CMRI of the 4-chamber view (b-SSFP imaging) demonstrating increased left ventricular wall thickness, small circum ferential pericardial effusion and biatrial enlargement. The right atrial myxoma is seen as hypointense signal attached to the interatrial septum; (C) T2-STIR image in a mid-short axis slice at the papillary muscle level, demonstrating hyper-intense signal in the posterior, lateral and septal walls; (D) LGE-CMRI image of the 4-chamber view showing patchy increased signal intensity of the myocardium in the septum and lateral walls. Note is made of hyper-intense signal in the atrial walls also.
ness (19 mm septum and 17 mm posterior wall) and moderately impaired contraction with diffuse hypokinesis and an estimated LV EF 37% (Figure 2(B)) [6] . The RV was mildly dilated and mildly impaired with Tricuspid Annular Plane Systolic Excursion (TAPSE) of 14 mm (normal range >16 mm) [8] . The E and A waves were fused, making detailed assessment of diastolic function difficult. There was also a small to moderate pericardial effusion. Given the marked LV hypertrophy and systemic inflammatory disease, myocardial infiltration was suspected. CMRI confirmed moderate to severe LV hypertrophy in all segments, with moderate to severe systolic impairment (LVEF 30%). The RV was mildly to moderately dilated and severely impaired (EF 30%) [6] . There was evidence of patchy diffuse basal mid-wall LGE of the LV with increased myocardial signal on T-2 weighted imaging in the basal septal, posterior and lateral walls, consistent with secondary or reactive form of CA (Figure 2(C)) [1] [3] -[5] [7] [9] . It was recommended that there be an escalation of systemic anti-inflammatory therapies in addition to conventional heart failure therapies, and the patient made marked clinical improvement in the short term. Endomyocardial biopsy was offered to the patient who declined on the basis of the risks explained for such a procedure. Progress CMRI was planned to assess the success of anti-inflammatory therapies.
3. Discussion
Amyloidosis can be either localised or systemic where multiple organs are involved [2] . The most common types of amyloidosis are: systemic light chain amyloidosis (AL) (previously known as primary amyloidosis); familial amyloid polyneuropathy (FAP); senile amyloidosis; and systemic amyloid A amyloidosis (previously known as secondary amyloidosis) [2] [10] [11] . In AL, involvement is usually systemic and multi-organ failure is the usual cause of death with heart failure as the commonest presentation [2] [10] [11] . FAP is an autosomal dominant disease where the inherited abnormal form of protein transthyretin (TTR) constitutes the amyloid fibre and commonly presents with conducting system disease, such as tachyarrhythmias [2] [10] . Senile amyloidosis affects those usually over 80 years of age where small amount of TTR protein is deposited in the myocardium over time, often presenting clinically as heart failure and frequently associated with carpal tunnel syndrome [2] . Secondary amyloidosis is usually associated with chronic inflammatory diseases with increased production of amyloid A (SAA) protein or less frequently associated with renal cell carcinoma or Hodgkin’s disease with clinical presentation often including renal insufficiency or proteinuria [2] [10] . We present two cases where increasing dyspnoea was the chief complaint associated with LV hypertrophy. There was evidence of diastolic dysfunction with bi-atrial dilatation in both patients and systolic dysfunction in case 2. On LGE-CMRI studies both patients showed patchy diffuse enhancement of the ventricular walls and increased signal on T2 imaging, the hall mark features of LV infiltration such as that which occurs with CA [5] [9] . The diagnosis of CA can be difficult in the early stages of the disease as it is often clinically silent [3] . The clinical presentation of more advanced CA commonly involves signs and symptoms of heart failure including dyspnoea, restrictive filling pattern due to elevated LVEDP, peripheral oedema, ascites and weight loss [7] [11] . Although, endomyocardial biopsy remains the gold standard in diagnosing CA with a specificity of 100%, it is rarely performed due to procedural risks and possibility of sampling error [4] . In systemic amyloidosis biopsy of the rectum, gingival and abdominal fat pad are preferred over myocardium [11] . A positive test for amyloidosis is present when
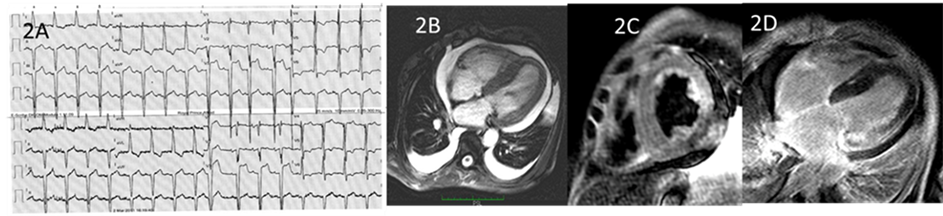
Figure 2. (A) 12 lead ECG demonstrating sinus rhythm with evidence of left atrial enlargement, non-specific intraventricular conduction block and left ventricular hypertrophy with repolarization abnormality; (B) CMRI showing a 4-chamber view (b-SSFP image) with increased left ventricular wall thickness and moderate circumferential pericardial effusion. Bilateral pleural effusions are also present; (C) T2-STIR image in a mid-short axis slice at the papillary muscle level, demonstrating hyper-intense signal in the posterior and lateral walls. (D) 4 chamber LGE-CMRI image demonstrating enhancement of the basal lateral wall.
Congo red staining produces an apple-green birefringence when viewed under a polarised microscope [2] . More often than not the diagnosis and management of patients with CA is dependent on non-invasive imaging [2] . LGE-CMRI and T2 weighted imaging has a role in diagnosing CA by enabling myocardial tissue characterization [12] . In particular, it is the ability of MRI to delineate LV infiltration with high spatial resolution and is a highly reproducible technique [1] [4] . Myocardial regions affected by the disease are typically hyperintense on T2 weighted images segments and enhance after contrast administration with LGE studies [4] [5] . CMR to date is the most sensitive non-invasive imaging technique in detecting CA (sensitivity 88%, specificity 95%) with high positive and negative predictive values of 93% and 90% respectively [5] . Additionally, where endomyocardial biopsy is planned in an affected patient, CMRI findings may assist in localising the most affected myocardial segments and therefore increasing diagnostic yield of sampling [4] [9] .
4. Conclusion
CA is an infiltrative disease, which is often clinically challenging to diagnose, and usually has late presentation and poor prognosis if left untreated [2] [3] . Non-invasive imaging techniques remain the diagnostic tools of choice. TTE provides useful information regarding diastolic dysfunction and increased wall thickness. We recommend the addition of CMRI to the screening protocol for CA, so that earlier non-invasive detection through the ability to better characterise the myocardium for infiltrative disease, may lead to better clinical outcomes.
References
- Seeger, A., Klumpp, B., Kramer, U., Stauder, N.I., Fenchel, M., Claussen, C.D. and Miller, S. (2009) MRI Assessment of Cardiac Amyloidosis: Experience of Six Cases with Review of the Current Literature. The British Journal of Radiology, 82, 337-342. http://dx.doi.org/10.1259/bjr/76104551
- Halwani, O. and Delgado, D.H. (2010) Cardiac Amyloidosis: An Approach to Diagnosis and Management. Expert Review of Cardiovascular, 8, 1007-1013. http://dx.doi.org/10.1586/erc.10.41
- Mussi, T.C., Szarf, G., Furtado, P.G.R., Duarte, F., Nakano, E., Pinetti, R., Szejnfeld, D., Franco, M.F., Antonio Carlos de, C., Carvalho, C. and Ajzen, S.A. (2010) Cardiac Amyloidosis Secondary to Multiple Myeloma. European Journal of Radiology Extra, 75, e13-e16. http://dx.doi.org/10.1016/j.ejrex.2010.04.011
- Wang, J., Kong, X., Xu, H., Zhou, G., Chang, D., Liu, D., Zhang, L. and Xie, M. (2010) Noninvasive Diagnosis of Cardiac Amyloidosis by MRI and Echocardiography. Journal of Huazhong University of Science and Technology [Medical Science], 30, 536-540.
- Pennell, D.J. and Maceira, A.M. (2009) Magnetic Resonance Imaging in Cardiac Amyloidosis. JACC: Cardiovascular Imaging, 2, 1369-1380. http://dx.doi.org/10.1016/j.jcmg.2009.10.001
- Lang, M.R., Bierig, M., Devereux, R.B., Flachskampf, F.A., Foster, E., Pellikka, P.A., Picard, M.H., Roman, M.J., Seward, J., Shanewise, J.S., Solomon, S.D., Spencer, K.T., St John Sutton, M. and Stewart, W.J. (2005) Recommendations for Chamber Quantification: A Report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, Developed in Conjunction with the European Association of Echocardiography, a Branch of the European Society of Cardiology. Journal of the American Society of Echocardiography, 18, 1440-1463. http://dx.doi.org/10.1016/j.echo.2005.10.005
- Garcia-Pavia, P., Tome-Esteban, M.T. and Rapezzi, C. (2011) Amyloidosis. Also a Heart Disease. Revista Española de Cardiología, 64, 797-808.
- Rudski, L.G., Lai, W.W., Afilalo, J., Hua, L., Handschumacher, M.D., Chandrasekaran, K., Solomon, S.D., Louie, E.K. and Schiller, N.B. (2010) Guidelines for the Echocardiographic Assessment of the Right Heart in Adults: A Report from the American Society of Echocardiography. Endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. Journal of The American Society of Echocardiography, 23, 685-713. http://dx.doi.org/10.1016/j.echo.2010.05.010
- Wassmuth, R., Abdel-Aty, H., Bohl, S. and Schulz-Menger, J. (2011) Prognostic Impact of T2-Weighted CMR Imaging for Cardiac Amyloidosis. European Radiology, 21, 1643-1650. http://dx.doi.org/10.1007/s00330-011-2109-3
- Falk, R.H. and Dubrey, S.W. (2010) Amyloid Heart Disease. Progress in Cardiovascular Diseases, 52, 347-361. http://dx.doi.org/10.1016/j.pcad.2009.11.007
- Maredia, N. and Ray, S. (2005) Cardiac Amyloidosis. Clinical Medicine, 5, 504-509. http://dx.doi.org/10.7861/clinmedicine.5-5-504
- Mekinian, A., Lions, C., Leleu, X., Duhamel, A., Lamblin, N., Coiteux, V., De Groote, P., Hatron, P.-Y., Facon, T., Beregi, J.-P., Hachulla, E. and Launay, D. (2010) Prognosis Assessment of Cardiac Involvement in Systemic AL Amyloidosis by Magnetic Resonance Imaging. The American Journal of Medicine, 123, 864-868. http://dx.doi.org/10.1016/j.amjmed.2010.03.022

