Health
Vol.6 No.5(2014), Article ID:43102,5 pages DOI:10.4236/health.2014.65044
Treatment success rate of tuberculosis patients in Dabat, northwest Ethiopia
![]()
Institute of Public Health, the University of Gondar, Gondar, Ethiopia; *Corresponding Author: sbsbtadesse90@gmail.com
Copyright © 2014 Sebsibe Tadesse, Takele Tadesse. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Sebsibe Tadesse, Takele Tadesse. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received 21 November 2013; revised 19 December 2013; accepted 26 December 2013
KEYWORDS
DOTS Strategy; Treatment Success Rate; Ethiopia
ABSTRACT
Evaluating the outcomes of DOTS program is essential for recognizing and amending system failures before the incidence and proportion of resistant isolates rise. In this study, we seek to evaluate the impact of DOTS strategy on tuberculosis treatment success rate in Dabat, northwest Ethiopia. Medical records of 1305 pulmonary and extrapulmonary tuberculosis patients registered from 2007 to 2012 at two health centers in Dabat district, northwest Ethiopia, were reviewed. Treatment outcomes and forms of tuberculosis were assessed according to WHO guidelines. Descriptive analyses were performed using frequencies and percentages. Treatment success rate was observed in 1146 (87.8%) patients. Out of these, 534 (89.1%) of the males and 612 (86.7%) of the females were successfully treated. It was also true in 338 (87.8%) of the smear-positive pulmonary tuberculosis patients, 473 (85.7%) of the smear-negative pulmonary tuberculosis patients, and 335 (91.0%) of the extrapulmonary tuberculosis patients. In conclusion, treatment success rate in the current study was slightly higher than the WHO international updated target for the period 2011 to 2015. However, this doesn’t mean that there will be no need for further enhancement of supervision and monitoring techniques, strengthening counseling and health education programs, and improving the quality of laboratory diagnostic services.
1. INTRODUCTION
Tuberculosis (TB) remains to be the major infectious disease causing high morbidity and mortality around the world. In 2010, there were an estimated 8.5 - 9.2 million new cases and 1.2 - 1.5 million deaths worldwide [1]. The World Health Organization (WHO) declared TB a global emergency and introduced the Directly Observed Treatment-Short course (DOTS) strategy for global TB control. In the face of this intensified effort to diagnose and treat TB, the rates in sub-Saharan Africa continue to climb [2].
Ethiopia is one of high TB endemic countries in the world, ranking 8th in the list of 22 high burden countries, and 3rd in Africa. In 2011, the estimated annual incidence and prevalence of all forms of TB were 258 and 237 per 100,000 populations, respectively [3]. A recent population-based survey showed that the prevalence of new sputum smear-positive TB was 174 per 100,000 populations [4]. The government initiated a pilot TB control project in 1992 based on the DOTS strategy. Since then the program has been subsequently scaled up in the country and implemented at the national level. The country’s DOTS coverage is estimated at 100.0% geographical and 95.0% health facility levels [5,6]. Currently, Ethiopia reports a treatment success rate of 83.0% for all forms of TB. Studies in some parts of the country—Tigray Region [7], Southern Region [8] and Gondar area [9] reported 89.2%, 74.8% and 29.5% treatment success rates in TB patients, respectively.
Monitoring treatment outcomes is essential in order to evaluate the effectiveness of the DOTS program [10]. This would make it possible to recognize and amend system failures before the incidence and proportion of resistant isolates rise [11]. However, only a few studies have been conducted to assess the treatment outcome of TB patients in northern Ethiopia [7,9]. Therefore, this study evaluates the treatment success rate of TB patients registered in Dabat district from 2007 to 2012.
2. METHODOLOGY
2.1. Study Area
This study was conducted in Dabat district, northwest Ethiopia. The district had an estimated population of 46,165. Like the rest of the districts in the northern part of the country, the livelihood of the community largely depended on subsistence agriculture. Only two health centers delivered DOTS service for TB patients in the community [12].
2.2. Study Design and Data Collection
A retrospective analysis of the profile of all TB patients registered at the two health centers delivering DOTS service between 2007 and 2012 was conducted. The registration documents reviewed contained basic information, such as age, sex, address, forms of TB, treatment card, TB register, and laboratory register.
2.3. Definitions
The following standard clinical case definitions of the National TB and Leprosy Control Program (NTLCP) guideline adopted from WHO [13] were used.
2.4. Smear-Positive Pulmonary TB (SPPTB)
A patient with at least two sputum specimens positive for acid-fast bacilli (AFB) by microscopy, or a patient with only one sputum specimen positive for AFB by microscopy, and chest radiographic abnormalities consistent with active pulmonary TB.
2.5. Smear-Negative Pulmonary TB (SNPTB)
A patient with symptoms suggestive of TB, with at least two sputum specimens negative for AFB by microscopy, and with chest radiographic abnormalities consistent with active pulmonary TB (including interstitial or miliary abnormal images), or a patient with two sets of at least two sputum specimens negative for AFB by microscopy taken at least two weeks apart, and radiographic abnormalities consistent with pulmonary TB and lack of clinical response to one week of broad spectrum antibiotic therapy.
2.6. Extrapulmonary TB (EPTB)
This included TB of organs other than the lungs, such as lymph nodes, the abdomen, the genitourinary tract, the skin, joints and bones, meninges, etc. The diagnosis of EPTB was based on fine needle aspiration cytology or biochemical analyses of the cerebrospinal/pleural/ascitic fluid or histopathological examination or strong clinical evidence consistent with active EPTB, followed by a decision of a clinician to treat with a full course of antiTB chemotherapy. In all the cases of EPTB, sputum examinations and chest radiographs were used to investigate the involvement of lung parenchyma.
2.7. Treatment Outcome
The treatment outcome was divided into seven categories according to NTLCP guideline. These categories were the following: cured (a patient whose sputum smear or culture was positive at the beginning of the treatment but who was smear or culture-negative in the last month of treatment and on at least one previous occasion); completed treatment (finished treatment, but without a bacteriology result at the end of treatment); failure (remaining smear positive at five months despite correct intake of medication); defaulted treatment (patients who interrupted their treatment for two consecutive months or more after registration); died (patients who died from any cause during the course of treatment); transferred out (patients whose treatment results were unknown due to transfer to another health facility) and successfully treated (a patient who was cured or completed treatment).
Death was determined by reviewing of the medical records followed by confirmation from DOTS contact persons.
2.8. Statistical Analyses and Ethical Considerations
Data were entered, cleaned and analyzed using the statistical package SPSS for windows, version 16. Inconsistencies in data entry were randomly checked by reentering 10% of the raw data. Frequencies and percentages were used to describe patient characteristics and treatment outcomes. The study was approved by the Institutional Review Board of the University of Gondar.
3. RESULTS
3.1. Patient Characteristics
A review of the treatment records of 1305 TB patients was retrieved from two health centers which were delivering DOTS services in the study area. Of these, 706 (54.1%) were females and 599 (45.9%) males. The majority, 1174 (90.0%) of the patients belonged to the economically productive age group, 15 to 64 years. More than three-fourths, 1025 (78.5%) were from the rural parts of the study area. The SNPTB patients accounted for 552 (42.3%), followed by 385 (29.5%) and 368 (28.2%) patients who had SPPTB and EPTB, respectively (Table 1).
3.2. Treatment Success Rate
Out of the total of 1305 TB patients evaluated for treatment outcomes from 2007-2012, treatment success rate was 1146 (87.8%), (see Figure 1). The treatment success rate was observed in 534 (89.1%) and 612 (86.7%) of males and females, respectively. It varied from 57 (80.3%) to 675 (89.5%) as the age group of patients varied from 0 - 14 years to ≥65 years. Two hundred fifty-one (89.7%) of the urban residents and 895 (87.3%) of the rural residents were successfully treated. Regarding the form of TB, treatment success rate was observed in 338 (87.8%) of the SPPTB patients, 473 (85.7%) of the SNPTB patients, and 335(91.0%) of the EPTB patients (Table 1).
4. DISCUSSION
DOTS is an internationally recognized patient-centered healthcare management system which provides support by observing patients on the process of taking their treatment, and thus ensures that they complete their treatment. It also helps identify patients who are in the infectious stage of the disease by monitoring sputum samples under the microscope, by providing effective drug treatment, and monitoring patients’ progress towards a cure [3]. The implementation of DOTS as a treatment strategy in Ethiopia has shown a marked success in improving patient adherence to treatment and consequently increasing the chance of patient cure [7].
In this study, treatment success rate was satisfactorily high (87.8%) in all TB patients treated in Dabat district. The result is slightly higher than the WHO international updated target of 87.0% for the period 2011 to 2015. The main reason for the improved successful treatment out come was the use of DOTS contact persons to closely monitor and trace patients who default from treatment. Studies conducted in other parts of Ethiopia [7,14] and Tanzania [15] reported higher successful treatment outcomes by using community health extension workers to follow-up patients. Another possible explanation for this may be better coordination of HIV and TB activities [5]. Studies show that HIV co-infection, which increases the risk of latent TB reactivation 20-fold, is the most powerful known risk factor for the progression of Mycobacterium tuberculosis infection to an active disease [16,17]. This higher successful treatment outcome in Dabat district implies that the performance of DOTS is encouraging, and the district is on the right track in achieving the WHO successful treatment outcome target in TB control.
This study was conducted in a typical densely populated agrarian community of northwest Ethiopia. The area is typical of the rural population of Ethiopia, representing 85.0% of the total population, where the prevalence of TB is high because of limited health service coverage, shortage of health workers, inadequate diagnostic and treatment facilities, and high social and perceived stigma [18]. Thus, we believe that our findings could be applicable in similar settings.
However, the major limitation of this study is that we have not had access to the full range of the socio-demographic and clinical variables in the medical records during the review process. Another limitation is that we analyzed the data from retrospective reviews of patient records which might affect the validity of the result. We recommend further studies which use longitudinal designs to develop the full pledged knowledge.
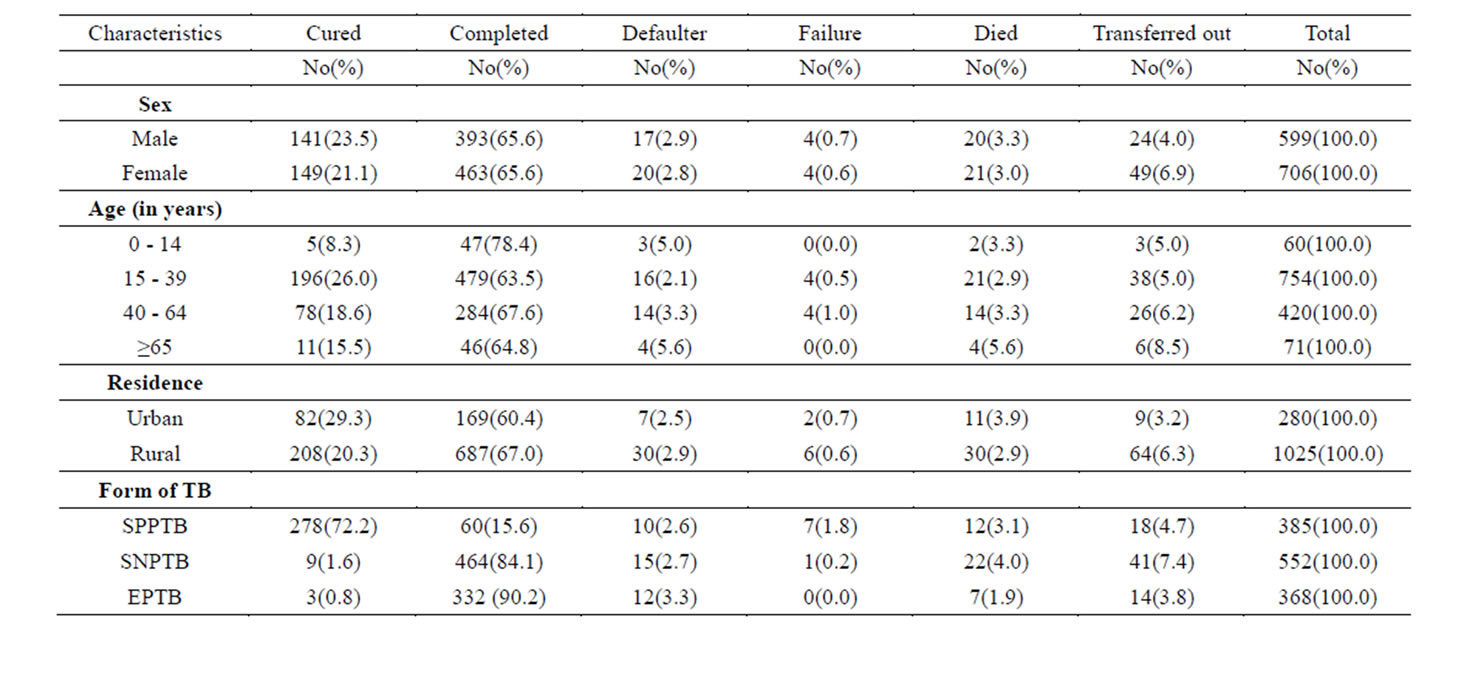
Table 1. Demographic characteristics of TB patients and treatment outcome, Dabat district, 2007-2012.
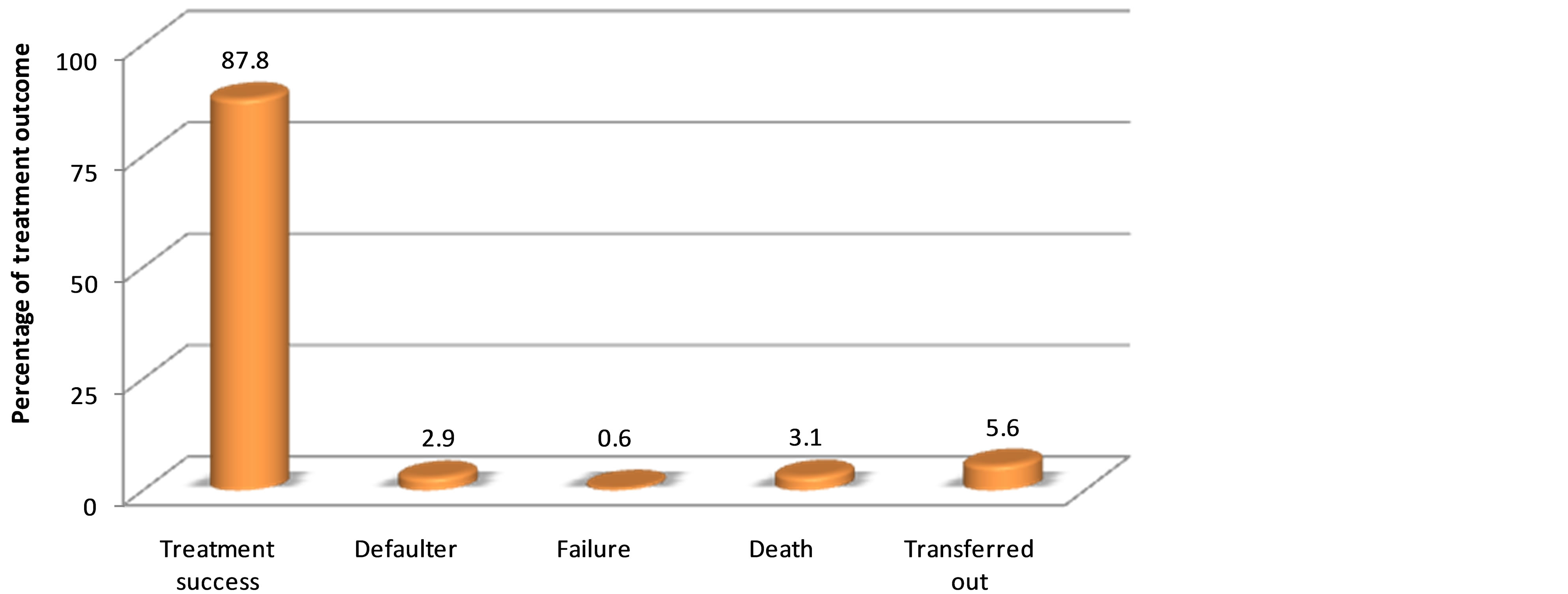
Figure 1. Proportion of TB treatment outcomes.
5. CONCLUSION
The treatment success rate in the current study was slightly higher than the WHO international updated target for the period 2011 to 2015. However, this doesn’t mean that there will be no need for further enhancement of supervision and monitoring techniques, strengthening counseling and health education programs, and improving the quality of laboratory diagnostic services.
ACKNOWLEDGEMENTS
Authors would like to thank the Armeaur Hansen Research Institute, and USAID/TB CARE-I for funding this study. They acknowledge all the staff members of Dabat and Woken health centers for the proper documentation of patient information and treatment outcome and for their genuine assistance during data collection.
REFERENCES
- World Health Organization (2011) Global tuberculosis control. World Health Organization, Geneva.
- World Health Organization (2010) Global tuberculosis control. World Health Organization, Geneva.
- World Health Organization (2012) Global tuberculosis control. World Health Organization, Geneva.
- Tadesse, T., Demissie, M., Berhane, Y., Kebede, Y. and Abebe, M. (2011) Two-thirds of smear-positive tuberculosis cases in the community were undiagnosed in Northwest Ethiopia: Population-based cross-sectional study. PLoS One, 6, 28258. http://dx.doi.org/10.1371/journal.pone.0028258
- Federal Ministry of Health (2011) Health and health related indicators, Ethiopia. Federal Ministry of Health, 37.
- Federal Ministry of Health (2011) Overview of National TB Control Implementation status. Sixth National TB Research Workshop, Gondar University, Gondar.
- Berhe, G., Enquselassie, F. and Aseffa, A. (2012) Treatment outcome of smear-positive pulmonary tuberculosis patients in Tigray Region, Northern Ethiopia. BMC Public Health, 12, 537. http://dx.doi.org/10.1186/1471-2458-12-537
- Munoz, S.M., Cuevas, L.E., Tumato, M., Merid, Y. and Yassin, M.A. (2010) Factors associated with poor tuberculosis treatment outcome in the Southern Region of Ethiopia. The International Journal of Tuberculosis and Lung Disease, 14, 973-979.
- Tessema, B., Muche, A., Bekele, A., Reissig, D., Emmrich, F. and Sack U. (2009) Treatment outcome of tuberculosis patients at Gondar University Teaching Hospital, Northwest Ethiopia: A five-year retrospective study. BMC Public Health, 9, 371. http://dx.doi.org/10.1186/1471-2458-9-371
- Veen, J., Raviglione, M., Rieder, H.L., Migliori, G.B., Graf, P., Grzemska, M. and Zalesky, R. (1998) Standardized tuberculosis treatment outcome monitoring in Europe. Recommendations of a Working Group of the WHO and the European Region of the International Union against Tuberculosis and Lung Disease (IUATLD) for uniform reporting by cohort analysis of treatment outcome in tuberculosis patients. European Respiratory Journal, 12, 505-510. http://dx.doi.org/10.1183/09031936.98.12020505
- Tuula, V., Pekka, H., Jukka, O., Kari, L., Maarit, K. and Petri, R. (2007) Risk factors for poor tuberculosis treatment outcome in Finland: a cohort study. BMC Public Health, 7, 291-299. http://dx.doi.org/10.1186/1471-2458-7-291
- Central Statistical Authority (2007) Population and Housing Census of Ethiopia: Results for Amhara Regional State. Addis Ababa.
- Ethiopian Federal Ministry of Health (2008) Tuberculosis, Leprosy and TB/HIV Prevention and Control Program Manual. Addis Ababa.
- Datiko, D.G. and Lindtjorn, B. (2009) Health Extension Workers improve tuberculosis case detection and treatment success in Southern Ethiopia: A community randomized trial. PLoS One, 4, e5443. http://dx.doi.org/10.1371/journal.pone.0005443
- Van, D.B.J., Lyimo, R., Irongo, C.F., Boeree, M.J., Schaalma, H., Aarnoutse, R.E. and Kibiki, G.S. (2009) Community vs. facility-based directly observed treatment for tuberculosis in Tanzania’s Kilimanjaro Region. The International Journal of Tuberculosis and Lung Disease, 13, 1524-1529.
- Getahun, H., Gunneberg, C., Granich, R. and Nunn, P. (2010) HIV infection associated tuberculosis: The epidemiology and the response. Clinical Infectious Diseases, 50, 201-207. http://dx.doi.org/10.1086/651492
- Kwan, C.K. and Ernst, J.D. (2011) HIV and tuberculosis: A deadly human syndemic. Clinical Microbiology Reviews, 24, 351-376. http://dx.doi.org/10.1128/CMR.00042-10
- Deribew, A., Hailemichael, Y., Tesfaye, M., Desalegn, D., Wogi, A. and Daba, S. (2010) The synergy between TB and HIV co-infection on perceived stigma in Ethiopia. BMC Research Notes, 3, 249. http://dx.doi.org/10.1186/1756-0500-3-249

