Advances in Bioscience and Biotechnology
Vol.06 No.03(2015), Article ID:54821,9 pages
10.4236/abb.2015.63017
Using the Electromagnetics of Cancer’s Centrosome Clusters to Attract Therapeutic Nanoparticles
Ronald L. Huston
Mechanical and Materials Engineering, University of Cincinnati, Cincinnati, USA
Email: ron.huston@uc.edu
Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 27 February 2015; accepted 15 March 2015; published 19 March 2015
ABSTRACT
This paper summarizes recent research findings concerning centrioles, centriole duplication, centriole overduplication, supernumerary centrioles, centrosomes, and centrosome amplification. The paper then discusses the status of ongoing research on the use of nanoparticles for cancer treatment. The research findings show that a centriole produces an electromagnetic field apparently due to the longitudinal oscillation of its microtubules (MTs). A cluster of centrioles is therefore presumed to produce an enhanced electromagnetic field. Individual centrioles are immersed in a cloud of electron-dense material (proteins) which together with the centrioles is known as the centrosome. A cluster of centrioles thus produces a cluster of centrosomes―a hallmark of cancer cells. With enhanced electromagnetic fields, centrosome clusters provide an attraction for magnetically charged nanoparticles. These nanoparticles however are not attracted to normal cells which with only two (or at most four) centrioles, have a weaker magnetic field. The idea is simple: Magnetized and therapeutic nanoparticles are directed toward tumors and then attracted to the centrosome clusters of the tumor cells. Once inside the tumor cells, the nanoparticles can release their toxins.
Keywords:
Centrioles, Supernumerary Centrioles, Cell Electromagnetism, Theraputic Nanoparticles, Tumorigenesis, Cancer

1. Introduction
The recent widespread use of electron microscopy, and currently the use of atomic force measurements, in studying internal cellular structures have provided an improved understanding of the geometry, composition, and functioning of the centrosome, the centrioles, and the microtubules. Many questions still remain. But based on the findings to date, the imagings show that within a cell, adjacent to the nucleus, is a pair of tiny organelles (approximately 400 nm in major dimension) known as “centrioles”. The centrioles may be viewed as a pair of annular cylinders perpendicular to each other at the cylinder bases. The cylinders themselves are composed of nine blades of microtubule triplets as represented in Figures 1-3 [1] [2] .
The centrioles are the principal drivers of cell division (mitosis). They lead the mitosis by first duplicating themselves so that there are then two pairs of centrioles―a “mother” and a “daughter” pair. The daughter centriole then separates from the mother and moves around the nucleus to the opposite side.
At the same time the nucleus membrane softens so that the nucleus can be pulled apart by the mother and daughter centrioles on the opposite sides. Also, at the same time the DNA within the nucleus is being copied (duplicated) and separated with one part going with the mother centriole side of the nucleus and the other with
Figure 1. Cross-section of a typical eukaryotic (animal/human) cell.
Figure 2. A centriole pair.
Figure 3. Microtubule blades of a centriole.
the daughter side.
As the mitosis continues, the remainder of the original cell’s cytoplasm with its organelles is divided between the separating parts and then two new cells emerge.
Mechanically this activity can be regarded as due to forces exerted at a distance, as opposed to forces exerted by contact. Such distance exerted forces are characteristic of electromagnetic forces.
When this process goes awry the cell either dies or it becomes malignant. The malignancies arise by a failure of the centrioles to duplicate once and only once, but instead several centrioles are produced during the duplication. That is, a “mother centriole” produces more than one daughter. When this happens, the DNA copying is also disturbed leading to chromosome instability.
The overduplication of the centrioles then leads to supernumerary centrioles which in turn stimulate rapidly developing cell separation and then a tumor of malignant cells.
To see how this can happen consider that when there are supernumerary centrioles within a cell the centrioles tend to coalesce with each centriole pair bringing with it a surrounding cloud of electron dense proteins. This coalescence results in a cluster of centrioles and centrosomes or “centrosome amplification”.
Recent studies show that most, if not all cancerous tumors have supernumerary centrioles [3] -[40] . Excessive centrioles frequently gather together leading to the formation of centrosomal clusters.
With mitosis beginning with centriole duplication, supernumerary centrioles accelerate the rate of cell division, and in the process cause aneuploidy in the nucleus and chromosome instability (CIN).
Recent studies also show that the centrioles via their vibrating microtubules (MTs) develop an electromagnetic field within the cell [41] -[66] . Therefore, if there are supernumerary clustered centrioles, the overall electropolarity of the cell is believed to be enhanced.
Also in recent years, with the ongoing advances of nanotechnology, nanoparticles have been developed which are sufficiently small that they can penetrate cell membranes [65] -[80] . Consequently if the nanoparticles are magnetically charged, they could be attracted by the presumed magnetic field surrounding the clustered centrioles. If the nanoparticles adhere to the centrosomal cluster, toxins and/or heating can be externally released and/ or induced to destroy the cell.
Normal cells with only one pair of centrioles, and thus lower electropolarity of cancer cells, will not be an attraction for the magnetically charged nanoparticles.
The following paragraphs provide additional detail, and a basis for the foregoing assertions. Reference [1] provides additional background information.
2. Centrioles and Electromagnetism
To see how centrioles with their vibrating microtubules can establish an electromagnetic field of a cell, recall first some of the unique features of these organelles.
1) Centrioles occur as a pair of small orthogonal cylinders lying adjacent to the nucleus. See Figure 1.
2) Centrioles have precise and uniform geometry, being approximately 400 to 500 nanometers (nm) long and approximately 200 nm in diameter. See Figure 2.
3) Unlike all other organelles and organs, centrioles have no membrane cover.
4) Instead, centrioles are hollow cylinders with a perimeter of nine radially inclined “blades” of microtubule (MT) triplets―as represented in Figure 2.
5) As with DNA, centrioles are self duplicating.
6) Finally, and of greatest importance for our consideration, centrioles are the principal drivers of mitosis- from initiation through division.
Next, although centrioles occur in pairs with precise geometry, they are not identical. One (the “daughter”) is a bit shorter, and interestingly, it is attached to the side at the end (or base) of the other (the “mother”) as in Figure 3. This intersection is considered to be at the “proximal” end of the centrioles. The intersection itself is immersed in a cloud of electron dense pericentriolar matter known as the “microtubule organizing center” (MTOC).
The centrioles together with the MTOC are known as the “centrosome”.
With the high electron density of the MTOC, the electropolarity of the proximal centriole ends is taken as “negative” with the distal ends then being “positive”.
In more focused detail, the microtubules (MT) in the centriole blades are themselves hollow cylinders, but with varying lengths (approximately 400 to 500 nm) depending upon the overall centriole length. The MT inside and outside diameters are approximately 15 and 25 nm. The MT cylinder walls are composed of 13 tubulin filaments evenly spaced around the circumference and running lengthwise along the cylinder, as represented in Figure 4.
The MT filaments are composed of α and β-tubulin dimers laid end-to-end as in Figure 4. These filaments have smooth surfaces, allowing them to slide longitudinally. The tubulin dimers have ionic polarity with again the proximal end being negative and the distal end being positive.
The longitudinal movement of the MT filaments is oscillatory with a frequency of approximately 465 MHz [81] . It is this vibration that is believed to give rise to the electropolarity of the centriole and thus also to the cell itself.
3. Electro-Polarity of Dividing Cells and Cancer Cells
When a cell is about to divide the centrioles begin to duplicate by growing new “daughter”) centrioles on their sides at their adjoining bases which are emersed in the electron dense cloud of the MTOC. The nascent centriole appears to arise when a small deposit of the protein Plk4 (aka “SAK”) is placed on the outer MT of the “mother centriole” [82] (see also [22] and [83] -[91] ). The nine-fold cartwheel geometry of the MTs is then developed by the protein SAS-6 joined to the small SAK deposit [22] [84] [86] [87] [89] -[92] .
Once the MTs of the daughter centrioles become mature and begin to oscillate they enhance the overall electromagnetic field of the (now four) centrioles. That is, once the pair of centrioles becomes two pair the intensity of the electromagnetic field is potentially doubled.
Suppose now that there is an error in the centriole duplication where the mother centriole has two or more
Figure 4. A microtubule (MT).
daughter centrioles. Suppose further that this defect of overduplication persists so that there are numerous centrioles, or “supernumerary” centrioles within the cell. Suppose still further that due to electromagnetic field interactions these multiple centrioles cluster together as is characteristic of cancer cells [14] [21] [26] [28] [40] [88] [93] -[100] . Then the resulting electromagnetic field of the cluster is presumed to be many times greater than that of a single centriole pair.
For some cancerous tumors, specifically breast cancers, this increase in electromagnetism can even be measured at the breast surface [59] [62] .
4. Application of Nanotechnology
The modern era of nanotechnology began about 1995 with an emphasis upon micro-manufacturing. More recently applications have been expanding including a focus upon biosystems.
“Nano” normally refers to dimensions of the order of 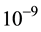 m. That is, 1 nanometer (nm) equates to
m. That is, 1 nanometer (nm) equates to 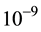 meters (m). A “nanoparticle” is thus one with nano-size dimension―that is, with major dimension
meters (m). A “nanoparticle” is thus one with nano-size dimension―that is, with major dimension 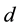 being:
being: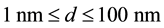 .
.
With the characteristic dimension of a cell being approximately 10 to 30 μm it is easy to visualize nanoparticles being able to penetrate a cellular surface.
In recent years many investigators have envisioned the use of therapeutic nanoparticles to treat malignant tumors. The idea is simple: If a nanoparticle can penetrate a cancer cell and release a toxin, while at the same time avoiding normal cells, an effective new cancer treatment will have been attained.
Here are a few quotes:
“Nanomedicines have enormous potential to improve the provision of cancer therapy, yet our ability to efficiently home these materials to regions of disease in vivo remains very limited”.
---G. von Maltzahn et al. [78]
“Within the family of nanomaterials, carbon nanotubes (CNT) have emerged as a new alternative and efficient tool for transporting and translocating therapeutic molecules”.
---A. Bianco et al. [100]
“To date, nanoparticles represent the most widely used carrier system for multifunctional drug delivery applications”.
---F. Wang et al. [73]
In spite of this optimism, the major problem is still how to effectively get the therapeutic nanoparticles into the cancer cells. D. Shi et al. [74] have suggested using magnetically charged nanoparticles and external magnetic fields to guide the particles to the tumors.
To carry this further, the way to have the therapeutic nanoparticles focus upon the cancer cells and yet avoid harming normal cells is to take advantage of the differences between cancer and normal cells, and specifically in this research, the differences in the electromagnetic properties of cancer and normal cells.
Advances in nanomaterials technology, and particularly in carbon nanotube technology [100] , are enabling the sensitizing and charging of nanoparticles to make them attracted to electromagnetic sources, and specifically with centrosome clusters.
5. Discussion
With some effective chemotherapies slowing the development of centrosome amplification, it follows that destruction of centrosome clusters will be an even more effective therapy [40] . The key to centrosome cluster destruction is getting therapeutic (toxic) nanoparticles to the clusters. The thesis advanced herein is that enhanced electropolarity of centrosome clusters will be an attraction for magnetically charged nanoparticles.
With nanoparticles being sufficiently small so that they can penetrate cell membranes, the nanoparticle size can arbitrarily be enlarged so that normal cell penetration is avoided, but cancer cells can be penetrated due to the electromagnetic attraction.
Although much remains to be done before this technology is efficacious, the steps forward are clear: More accurate empirical data is needed on the electromagnetic properties of microtubules, centrioles and centrosomes, and centrosome clusters. This data in turn will dictate the necessary additional testing and the development of the more effective nanoparticles.
Acknowledgements
Partial support for this research was provided by the Schafer Foundation and is sincerely appreciated. Also appreciated is the encouragement of Bardyl Tirana and Roger Adelman.
References
- Huston, R.L. (2014) On Centrioles, Microtubules, and Cellular Electromagnetism. Journal of Nanotechnology in Engineering in Medicine, 5, 031003-1-031003-5.
- Alberts, B., Bray, D., Lewis, J., Raff, M., Roberts, K. and Watson, J.D. (1994) Molecular Biology of the Cell. 3rd Edition, Garland Publishing, New York, Vol. 18, 803-818.
- Duensing, A., Liu, Y., Perdreau, S.A., Kleylein-Sohn, J., Nigg, E.G. and Duensing, S. (2007) Centriole Overduplication through the Concurrent Formation of Multiple Daughter Centrioles at Single Material Templates. Nature Oncogene, 26, 5280-6288.
- Schöeckel, L., Möeckel, M., Mayer, B., Boos, D. and Stemmann, O. (2011) Cleavage of Cohesion Rings Coordinates the Separation of Centrioles and Chromatids. Nature Cell Biology, 13, 966-972. http://dx.doi.org/10.1038/ncb2280
- Nigg, E.A. and Raft, J.W. (2009) Centrioles, Centrosomes, and Cilia in Health and Disease. Cell, 139, 663-678. http://dx.doi.org/10.1016/j.cell.2009.10.036
- Kobayashi, T. and Dynlacht, B.D. (2011) Regulating the Transition from Centriole to Basal Body. Journal of Cell Biology, 193, 435-444. http://dx.doi.org/10.1083/jcb.201101005
- Gansen, N.J., Godinho, S.A. and Pellman, D. (2009) A Mechanism Linking Extra Centrosomes to Chromosomal Instability. Nature, 460, 278-282. http://dx.doi.org/10.1038/nature08136
- Nigg, E.A. (2002) Centrosome Aberrations: Cause or Consequence of Cancer Progression? Nature Reviews Cancer, 2, 815-825. http://dx.doi.org/10.1038/nrc924
- Duensing, A., Liu, Y., Perdreau, S.A., Kleylein-Sohn, J., Nigg, E.A. and Duensing, S. (2007) Centriole Overduplication through the Concurrent Formation of Multiple Daughter Centrioles at Single Material Templates. Oncogene, 26, 6280-6288. http://dx.doi.org/10.1038/sj.onc.1210456
- Korzeniwski, N., Hohenfellner, M. and Duensing, S. (2012) CANDI Promotes PLK4-Mediated Centriole Overduplication and is Frequently Disrupted in Prostate Cancer. Neoplasia, 14, 799-806.
- Nigg, E.A. (2006) Origins and Consequences of Centrosome Aberrations in Human Cancers. International Journal of Cancer, 119, 2717-2723. http://dx.doi.org/10.1002/ijc.22245
- Godinho, S.A., Kwan, M. and Pellman, D. (2009) Centrosomes and Cancer: How Cancer Cells Divide with Too Many Centrosomes. Cancer and Metastasis Reviews, 28, 85-98. http://dx.doi.org/10.1007/s10555-008-9163-6
- Sluder, G. and Nordberg, J.J. (2004) The Good, the Bad and the Ugly: The Practical Consequences of Centrosome Amplification. Current Opinion in Cell Biology, 16, 49-54. http://dx.doi.org/10.1016/j.ceb.2003.11.006
- Brinkley, B.R. (2001) Managing the Centrosome Numbers Game: From Chaos to Stability in Cancer Cell Division. Trends in Cell Biology, 11, 18-21. http://dx.doi.org/10.1016/S0962-8924(00)01872-9
- D’Assoro, A.B., Lingle, W.L. and Salisbury, J.L. (2002) Centrosome Amplification and the Development of Cancer. Oncogene, 21, 6146-6153. http://dx.doi.org/10.1038/sj.onc.1205772
- Tsou, M.F. and Sterns, T. (2006) Mechanism Limiting Centrosome Duplication to Once Per Cell Cycle. Nature, 442, 947-951. http://dx.doi.org/10.1038/nature04985
- Bettencourt-Dias, M. and Glover, D.M. (2009) SnapShot: Centriole Biogenesis. Cell, 136, 188.e1-188.e2. http://dx.doi.org/10.1016/j.cell.2008.12.035
- Loffler, H., Fechter, A., Liu, F.Y., Poppelreuther, S. and Kramer, A. (2013) DNA Damage-Induced Centrosome Amplification Occurs via Excessive Formation of Centriolar Satellites. Oncogene, 32, 2963-2972. http://dx.doi.org/10.1038/onc.2012.310
- Vitre, B.D. and Cleveland, D.W. (2012) Centrosomes, Chromosomes Instability (CIN) and Aneuploidy. Current Opinion in Cell Biology, 24, 809-815. http://dx.doi.org/10.1016/j.ceb.2012.10.006
- Nigg, E.A. and Sterns, T. (2011) The Centrosome Cycle: Centriole Biogenesis, Duplication and Inherent Asymmetries. Nature Cell Biology, 13, 1154-1160. http://dx.doi.org/10.1038/ncb2345
- Lingle, W.L., Barrett, S.L., Negron, V.C., D’Assoro, A.B., Boeneman, K., Liu, W., et al. (2002) Centrosome Amplification Drives Chromosomal Instability in Breast Tumor Development. Proceedings of the National Academy of Sciences of the United States of America, 99, 1978-1983. http://dx.doi.org/10.1073/pnas.032479999
- Rodrigues-Martins, A., Riparbelli, M., Callaini, G., Glover, D.M. and Bettencourt-Dias, M. (2008) From Centriole Biogenesis to Cellular Function: Centrioles Are Essential for Cell Division at Critical Developmental Stages. Cell Cycle, 7, 11-16. http://dx.doi.org/10.4161/cc.7.1.5226
- Kramer, A., Neben, K. and Ho, A.D. (2002) Centrosome Replication, Genomic Instability, and Cancer. Leukemia, 16, 767-775. http://dx.doi.org/10.1038/sj.leu.2402454
- Ganem, N.J., Godinho, S.A. and Pellman, D. (2009) A Mechanism Linking Extra Centrosomes to Chromosomal Instability. Nature, 460, 278-282. http://dx.doi.org/10.1038/nature08136
- Vulprecht, J., David, A., Tibelius, A., Castiel, A., Konotop, G., Liu, F.Y., et al. (2012) STIL Is Required for Centriole Duplication in Human Cells. Journal of Cell Science, 125, 1353-1362. http://dx.doi.org/10.1242/jcs.104109
- Kwon, M., Godinho, S.A., Chandhok, N.S., Ganem, N.J., Azioune, A., Thery, M. and Pellman, D. (2008) Mechanisms to Suppress Multipolar Divisions in Cancer Cells with Extra Chromosomes. Genes & Development, 22, 2189-2203. http://dx.doi.org/10.1101/gad.1700908
- Phian, G.A., Wallace, J., Zhou, Y. and Doxsey, S.J. (2003) Centrosome Abnormalities and Chromosome Instability Occur Together in Pre-Invasive Carcinomas. Cancer Research, 63, 1398-1404.
- Holland, A.J., Lan, W., Niessen, S., Hoover, H. and Cleveland, D.W. (2010) Polo-Like Kinase 4 Kinase Activity Limits Centrosome Overduplication by Autoregulating Its Own Stability. The Journal of Cell Biology, 188, 191-198. http://dx.doi.org/10.1083/jcb.200911102
- Lingle, W.L. and Salisbury, J.L. (1999) The Role of the Centrosome in the Development of Malignant Tumors. Current Topics in Developmental Biology, 49, 313-329. http://dx.doi.org/10.1016/S0070-2153(99)49015-5
- Goepfert, T.M., Adigun, Y.E., Zhong, L., Gay, J., Medina, D. and Brinkley, W.R. (2002) Centrosome Amplification and Overexpression of Aurora A Are Early Events in Rat Mammary Carcinogenesis. Cancer Research, 62, 4115-4122.
- Marx, J. (2001) Do Centrosome Abnormalities Lead to Cancer? Science, 292, 426-429. http://dx.doi.org/10.1126/science.292.5516.426
- Marthiens, V., Rujano, M.A., Pennetier, C., Tessier, S., Paul-Gilloteaux, P. and Basto, R. (2013) Centrosome Amplification Causes Microcephaly. Nature Cell Biology, 15, 731-740. http://dx.doi.org/10.1038/ncb2746
- Maiato, H. and Logarinho, E. (2014) Mitotic Spindle Multipolarity without Centrosome Amplifications. Nature Cell Biology, 16, 386-394. http://dx.doi.org/10.1038/ncb2958
- Lingle, W.L. and Salisbury, J.L. (1999) Altered Centrosome Structure Is Associated with Abnormal Mitoses in Human Breast Tumors. American Journal of Pathology, 155, 1941-1951. http://dx.doi.org/10.1016/S0002-9440(10)65513-7
- Yang, Z., Loncarek, J., Khodjakov, A. and Rieder, C.L. (2008) Extra Centrosomes and/or Chromosomes Prolong Mitosis in Human Cells. Nature Cell Biology, 10, 748-751. http://dx.doi.org/10.1038/ncb1738
- Leber, B., Maier, B., Fuchs, F., Chi, J., Riffel, P., Anderhub, S., et al. (2010) Proteins Required for Centrosome Clustering in Cancer Cells. Science Translational Medicine, 2, 33-38. http://dx.doi.org/10.1126/scitranslmed.3000915
- Pinhan, G.A., Purohit, A., Wallace, J., Knecht, H., Woda, B., Quesenberry, P. and Doxsey, S.J. (1998) Centrosome Defects and Genetic Instability in Malignant Tumors. Cancer Research, 58, 3974-3985.
- Lingle, W.L., Kutz, W.H., Ingle, J.N., Maible, N.J. and Salisbury, J.L. (1998) Centrosome Hypertrophy in Human Breast Tumors: Implications for Genomic Stability and Cell Polarity. Proceedings of the National Academy of Sciences of the United States of America, 95, 2950-2955. http://dx.doi.org/10.1073/pnas.95.6.2950
- Bornens, M. (2008) Organelle Positioning and Cell Polarity. Nature Reviews Molecular Cell Biology, 9, 874-886. http://dx.doi.org/10.1038/nrm2524
- Fukasawa, K. (2007) Oncogenes and Tumor Suppressors Take on Centrosomes. Nature Reviews Cancer, 7, 911-924. http://dx.doi.org/10.1038/nrc2249
- Azimzadeh, J. and Marshall, W.F. (2010) Building the Centriole. Current Biology, 20, R816-R825. http://dx.doi.org/10.1016/j.cub.2010.08.010
- Meggs, W.J. (1988) Electric Fields Deforming the Spatial Organization of Microtubules and Action Filaments. Medical Hypotheses, 26, 165-170. http://dx.doi.org/10.1016/0306-9877(88)90095-3
- Kucera, O. and Havelka, D. (2012) Mechano-Electrical Vibrations of Microtubules―Link to Subcellular Morphology. Biosystems, 109, 346-355. http://dx.doi.org/10.1016/j.biosystems.2012.04.009
- Tyner, K.M., Kopelman, R. and Philbert, M. (2007) “Nanosized Voltmeter” Enables Cellular-Wide Electric Field Mapping. Biophysical Journal, 93, 1163-1174. http://dx.doi.org/10.1529/biophysj.106.092452
- Zhao, Y. and Zhan, Q. (2012) Electric Oscillation and Coupling of Chromatics Regulate Chromosome Packaging and Transcription in Eukaryotic Cells. Theoretical Biology and Medical Modelling, 9, 27-38. http://dx.doi.org/10.1186/1742-4682-9-27
- Brown, J.A. and Tuszynski, J.A. (1999) A Review of the Ferroelectric Model of Microtubules. Ferroelectrics, 220, 141-155. http://dx.doi.org/10.1080/00150199908216213
- Pokorny, J. (2011) Electrodynamic Activity of Healthy and Cancer Cells. 19th International Fröhlick’s Symposium, Journal of Physics: Conference Series, 329, Paper No. 012007.
- Cifro, M., Havelka, D. and Kucera, O. (2010) Electric Oscillations Generated by Collective Vibration Modes of Microtubules. In: Kinnamen, M. and Myllyla, R., Eds., Laser Applications in Life Sciences, Proceedings of SPIE, Vol. 7376.
- Pokorny, J., Hasek, J., Vanis, J. and Jelinek, F. (2008) Biophysical Aspects of Cancer―Electromagnetic Mechanism. Indian Journal of Experimental Biology, 46, 310-321.
- Pokorny, J., Hasek, J. and Jelinek, F. (2005) Electromagnetic Field of Microtubules: Effects on Transfer of Mass Particles and Electrons. Journal of Biological Physics, 31, 501-514. http://dx.doi.org/10.1007/s10867-005-1286-1
- Pokorny, J., Pokorny, J. and Kobikova, J. (2013) Postulates on Electromagnetic Activity in Biological Systems and Cancer. Integrative Biology, 5, 1439-1446. http://dx.doi.org/10.1039/c3ib40166a
- Cifra, M., Pokorny, J., Havelka, D. and Kucera, O. (2010) Electric Field Generated by Axial Longitudinal Vibration Modes of Microtubule. Biosystems, 100, 122-131. http://dx.doi.org/10.1016/j.biosystems.2010.02.007
- Pokorny, J., Hasek, J. and Jelinek, F. (2005) Endogenous Electric Field and Organization of Living Matter. Electromagnetic Biology and Medicine, 24, 185-197. http://dx.doi.org/10.1080/15368370500379566
- Tsai, M.C. and Ahringer, J. (2007) Microtubules Are Involved in Anterior-Posterior Axis Formation in C. elegans Embryos. Journal of Cell Biology, 179, 397-402. http://dx.doi.org/10.1083/jcb.200708101
- McKean, P.G., Vaughan, S. and Gull, K. (2001) The Extended Tubulin Superfamily. Journal of Cell Science, 114, 2723-2733.
- Tassin, A.M. and Bornens, M. (1999) Centrosome Structure and Microtubule Nucleation in Animal Cells. Biology of the Cell, 91, 343-354. http://dx.doi.org/10.1111/j.1768-322X.1999.tb01092.x
- Raynaud-Messina, B. and Merdes, A. (2007) γ-Tubulin Complexes and Microtubule Organization. Current Opinion in Cell Biology, 19, 24-30. http://dx.doi.org/10.1016/j.ceb.2006.12.008
- McCaig, C.D., Rajnicek, A.M., Song, B. and Zhao, M. (2005) Controlling Cell Behavior Electrically: Current Views and Future Potential. Physiological Reviews, 85, 943-978. http://dx.doi.org/10.1152/physrev.00020.2004
- Cuzick, J., Holland, R., Barth, V., Davies, R., Faupel, M., Fentiman, I., Frischbier, H.J., LaMarque, J.L., Merson, M., Sacchini, V., Vanel, D. and Veronesi, U. (1998) Electropotential Measurements as a New Diagnostic Modality for Breast Cancer. The Lancet, 352, 359-363. http://dx.doi.org/10.1016/S0140-6736(97)10002-2
- Djamqoz, M.B.A., Mycielska, M., Madeja, Z., Fraser, S.P. and Korohoda, W. (2001) Directional Movement of Rat Prostate Cancer Cells in Direct-Current Electric Field: Involvement of Voltagegated Na+ Channel Activity. Journal of Cell Science, 114, 2697-2705.
- Mycielska, M.E. and Djamqoz, M.B.A. (2004) Cellular Mechanisms of Direct-Current Electric Field Effects: Galvanotaxis and Metastatic Disease. Journal of Cell Science, 117, 1631-1639. http://dx.doi.org/10.1242/jcs.01125
- Zhoo, M., Bai, H., Wang, E., Forrester, J.V. and McCaig, C.D. (2003) Electrical Stimulation Directly Induces Pre-Angio- genic Responses in Vascular Endothelia Cells by Signaling through VEGF Receptors. Journal of Cell Science, 117, 397-405. http://dx.doi.org/10.1242/jcs.00868
- Olivotto, M., Arcanqeli, A., Carla, M. and Wanke, E. (1996) Electric Fields at the Plasma Membrane Level: A Neglected Element in the Mechanism of Cell Signaling. BioEssays, 18, 495-504. http://dx.doi.org/10.1002/bies.950180612
- Davies, R.J., Joseph, R., Asbun, H. and Sedwitz, M. (1989) Detection of the Cancer-Prone Colon, Using Transepithelial Impedence Analysis. JAMA Surgery, 124, 480-484. http://dx.doi.org/10.1001/archsurg.1989.01410040090021
- Binggeli, R. and Weinstein, R.C. (1986) Membrane Potentials and Sodium Channels: Hypotheses for Growth Regulation and Cancer Formation Based on Changes in Sodium Channels and Gap Junctions. Journal of Theoretical Biology, 123, 377-401. http://dx.doi.org/10.1016/S0022-5193(86)80209-0
- Garci-Bennett, A., Hess, M. and Fadeel, B. (2011) In Search of the Holy Grail: Folate-Targeted Nanoparticles for Cancer Therapy. Biochemical Pharmacology, 81, 976-984. http://dx.doi.org/10.1016/j.bcp.2011.01.023
- Ridley, A. and Heald, R. (2011) Cell Structure and Dynamics―Editorial Overview. Current Opinion in Cell Biology, 23, 1-3. http://dx.doi.org/10.1016/j.ceb.2010.12.003
- Kim, D.H., Rozhkova, E.A., Ulasov, I.V., Bader, S.D., Rajh, T., Lesniak, M.S. and Novosad, V. (2010) Biofunctionalized Magentic-Vortex Microdiscs for Targeted Cancer-Cell Destruction. Nature Materials, 9, 165-171. http://dx.doi.org/10.1038/nmat2591
- Dobson, J. (2008) Remote Control of Cellular Behavior with Magnetic Nanoparticles. Nature Nanotechnology, 3, 139- 143. http://dx.doi.org/10.1038/nnano.2008.39
- Nogales, E., Whittaker, M., Milligan, R.A. and Downing, K.H. (1999) High-Resolution Model of the Microtubule. Cell, 96, 79-88. http://dx.doi.org/10.1016/S0092-8674(00)80961-7
- Mannix, R.J., Kumar, S., Cassiola, F., Montoya-Zavala, M., Feinstein, E., Prentiss, M. and Inber, D.E. (2007) Nano- magnetic Actuation of Receptor-Mediated Signal Transduction. Nature Nanotechnology, 3, 36-40.
- Neuberger, T., Schöpf, B., Hofmann, H., Hofmann, M. and von Rechenberg, B. (2005) Superparamagnetic Nanoparticles for Biomedical Applications: Possibilities and Limitations of a New Drug Delivery System. Journal of Magnetism and Magnetic Materials, 293, 483-496. http://dx.doi.org/10.1016/j.jmmm.2005.01.064
- Weng, F., Pauletti, G.M., Wang, J., Zhang, J., Ewing, R.C., Wang, Y. and Shi, D. (2013) Dual Surface-Functionalized Janus Nanocomposites of Polystyrene/Fe3O4@SiO2 for Simultaneous Tumor Cell Targeting and Stimulus-Induced Drug Release. Advanced Materials, 25, 3485-3489. www.advmat.de
- Shi, D., Bedford, N.M. and Cho, H.S. (2011) Engineered Multifunctional Nanocarriers for Cancer Diagnosis and Therapeutics. Small, 7, 2549-2567.
- Chow, E.K.H. and Ho, D. (2013) Cancer Nanomedicine: From Drug Delivery to Imaging. Science Translational Medicine, 5, 216rv4. http://dx.doi.org/10.1126/scitranslmed.3005872
- Pouponneau, P., Leroux, J.C., Soulez, G., Gaboury, L. and Martel, S. (2011) Co-Encapsulation of Magnetic Nanoparticles and Doxorubicin into Biodegradable Microcarriers for Deep Tissue Targeting by Vascular MRI Navigation. Biomaterials, 32, 3481-3486. http://dx.doi.org/10.1016/j.biomaterials.2010.12.059
- Liu, Z., Chen, K., Davis, C., Sherlock, S., Cao, Q.Z., Chen, X. and Dai, H. (2008) Drug Delivery with Carbon Nano- tubes for in Vivo Cancer Treatment. Cancer Research, 68, 6652-6660. http://dx.doi.org/10.1158/0008-5472.CAN-08-1468
- von Maltzahn, G., Park, J.H., Lin, K.Y., Singh, N., Schwöppe, C., Mesters, R., Berdel, W.E., Ruoslahti, W., Sailor, M.J. and Bhatia, S.N. (2011) Nanoparticles that Communicate in Vivo to Amplify Tumour Targeting. Nature Materials, 10, 545-552. http://dx.doi.org/10.1038/nmat3049
- Fonseca, N.A., Gregó, A.C., Valério-Fernandes, A., Simões, S. and Moreira, J.N. (2014) Bridging Cancer Biology and the Patient’s Needs with Nanotechnology-Based Approaches. Cancer Treatment Reviews, 40, 626-635. http://dx.doi.org/10.1016/j.ctrv.2014.02.002
- Avvakumova, S., Colombo, M., Tortoya, P. and Prosperi, D. (2014) Biotechnological Approaches toward Nanoparticle Biofunctionalization. Trends in Biotechnology, 32, 11-20. http://dx.doi.org/10.1016/j.tibtech.2013.09.006
- Pokorny, J., Vedruccio, C., Cifra, M. and Kucera, O. (2011) Cancer Physics: Diagnostics Based on Damped Cellular Elastoelectrical Vibrations in Microtubules. European Biophysics Journal, 40, 747-759. http://dx.doi.org/10.1007/s00249-011-0688-1
- Sonnen, K.F., Schmellen, L., Leonhardt, H. and Nigg, E.A. (2012) 3D-Structured Illumination Microscopy Provides Novel Insight into Architecture of Human Centrosomes. Biology Open, 1, 965-976. http://dx.doi.org/10.1242/bio.20122337
- Azimzadeh, J. and Bornens, M. (2007) Structure and Duplication of the Centrosome. Journal of Cell Science, 120, 2139-2142. http://dx.doi.org/10.1242/jcs.005231
- Habedanek, R., Stierhof, Y.D., Wilkinson, C.J. and Nigg, E.A. (2005) The Polo Kinase Plk4 Functions in Centriole Duplication. Nature Cell Biology, 7, 1140-1146. http://dx.doi.org/10.1038/ncb1320
- Baffet, A.D., Martin, C.A., Scarfone, I., Daly, O.M., David, A., Tibelius, A., Lattao, R., Hussain, M.S. and Woodruff, J.B. (2013) Meeting Report―Building a Centrosome. Journal of Cell Science, 126, 3259-3262. http://dx.doi.org/10.1242/jcs.136721
- Bettencourt-Dias, M., Rodrigues-Martins, A., Carpenter, L., Riparbelli, M., Lehmann, L., Gatt, M.K., Carmo, N., Balloux, F., Callainj, G. and Glover, D.M. (2005) SAK/PLK4 Is Required for Centriole Duplication and Flagella Development. Current Biology, 15, 2199-2207. http://dx.doi.org/10.1016/j.cub.2005.11.042
- Brownlee, C.W. and Rogers, G.C. (2013) Show Me Your License, Please: Deregulation of Centriole Duplication Mechanisms that Promote Amplification. Cellular and Molecular Life Sciences, 70, 1021-1034. http://dx.doi.org/10.1007/s00018-012-1102-6
- Marthiens, V., Piel, M. and Basto, R. (2012) Never Tear Us Apart―The Importance of Centrosome Clustering. Journal of Cell Science, 125, 3281-3292. http://dx.doi.org/10.1242/jcs.094797
- Rodrigues-Martins, A., Riparbelli, M., Callaini, G., Glover, D.M. and Bettencourt-Dias, M. (2007) Revisiting the Role of the Mother Centriole in Centriole Biogenesis. Science, 316, 1046-1050. http://dx.doi.org/10.1126/science.1142950
- Bettencourt-Dias, M. and Glover, D.M. (2007) Centrosome Biogenesis and Function: Centrosomics Brings New Understanding. Nature Reviews Molecular Cell Biology, 8, 451-463. http://dx.doi.org/10.1038/nrm2180
- Strnad, P. and Gonczy, P. (2008) Mechanisms of Procentriole Formation. Trends in Cell Biology, 18, 389-396. http://dx.doi.org/10.1016/j.tcb.2008.06.004
- van Breugel, M., Hirono, M., Andreeva, A., Yanagisawa, H.A., Yamaguchi, S., Nakazawa, Y., Morgner, N., Petrovich, M., Ebong, I.O., Robinson, C.V., Johnson, C.M., Veprintsev, D. and Zuber, B. (2011) Structures of SAS-6 Suggest Its Organization in Centrioles. Science, 331, 1196-1199. http://dx.doi.org/10.1126/science.1199325
- Fukasawa, K. (2005) Centrosome Amplifications, Chromosome Instability and Cancer Development. Cancer Letters, 230, 6-19. http://dx.doi.org/10.1016/j.canlet.2004.12.028
- Chan, J.Y. (2011) A Clinical Overview of Centrosome Amplification in Human Cancers. International Journal of Biological Sciences, 7, 1122-1144. http://dx.doi.org/10.7150/ijbs.7.1122
- Kawamura, K., Izumi, H., Ma, Z., Ikeda, R., Moriyama, M., Tanaka, T., Nojima, T., Levin, L.S., Fujikawa-Yamamoto, K., Suzuki, K. and Fukasawa, K. (2004) Induction of Centrosome Amplification and Chromosome Instability in Human Bladder Cancer Cells by p53 Mutation and Cyclin E Overexpression. Cancer Research, 64, 4800-4809. http://dx.doi.org/10.1158/0008-5472.CAN-03-3908
- Sato, N., Mizumoto, K., Nakamura, M., Ueno, H., Minamishima, Y.A., Farber, J.L. and Tanaka, M. (2000) A Possible Role for Centrosome Overduplication in Radiation-Induced Cell Death. Oncogene, 19, 5281-5290. http://dx.doi.org/10.1038/sj.onc.1203902
- Kasbek, C., Yang, C.H., Uysof, A.M., Chapman, H.M., Winey, M. and Fink, H.A. (2007) Preventing the Degradation of Mps1 at Centrosomes Is Sufficient to Cause Centrosome Reduplication in Human Cells. Molecular Biology of the Cell, 18, 4457-4469. http://dx.doi.org/10.1091/mbc.E07-03-0283
- Loncarek, J., Herget, P. and Khodjakou, A. (2010) Centriole Reduplication during Prolonged Interphase Requires Procentriole Maturation Governed by Plk1. Current Biology, 20, 1277-1282. http://dx.doi.org/10.1016/j.cub.2010.05.050
- Bettencourt-Dias, M., Hildebrandt, F., Pellman, D., Woods, G. and Godinho, S.A. (2011) Centrosomes and Cilia in Human Disease. Trends in Genetics, 27, 307-315. http://dx.doi.org/10.1016/j.tig.2011.05.004
- Bianco, A., Kostarelos, K. and Prato, M. (2005) Applications of Carbon Nanotubes in Drug Delivery. Current Opinion in Chemical Biology, 9, 674-679. http://dx.doi.org/10.1016/j.cbpa.2005.10.005





