Journal of Behavioral and Brain Science
Vol. 2 No. 3 (2012) , Article ID: 22088 , 4 pages DOI:10.4236/jbbs.2012.23035
Development of the Gusto Facial Reflex in Perinatally Undernourished Rats*
Departamento de Neurobiología del Desarrollo y Neurofisiología, Instituto de Neurobiología, Universidad Nacional Autónoma de México, Campus UNAM Juriquilla, Querétaro, México
Email: masal@unam.mx
Received February 14, 2012; revised March 29, 2012; accepted April 18, 2012
Keywords: Gusto Facial Reflex; Newborn Rats; Undernutrition
ABSTRACT
In the rat, the taste system plays a critical role in motivating the animal to consume nutrients and avoid toxic substances. In neonatal rat the orofacial movements can be modified by the application of sucrose and quinine in the mouth, producing ingestion or rejection responses, respectively, but there is no information available on the gusto facial reflexes (GFR) under perinatal fasting. The aim of the present study was to determine how undernutrition can affect the development of orofacial responses to sucrose, quinine, and NaCl during early development. Pregnant dams were undernourished by being given 50% of a balanced diet from G6 to G12, 60% from G13 - G18, and 100% from G 19- G21. On postpartum days 0 - 9, prenatally underfed (UG) pups continued the undernourishment by remaining for 12 h with a foster dam and 12 h with a nipple-ligated mother. Stimuli were presented as a single droplet of sucrose (S), sodium chloride (NaCl), quinine hydro chloride (Q), and water (W) onto the lips at 1, 3, 5, 7, and 9 days of age, and mouth-opening frequency (MO) and lip-licking frequency (LL) were analyzed. The results show consistent effects associated with age, but not with diet, except in the cases of 0.01 M Q, which provoked in the UG subjects increased MO and the reduction of LL, and 0.1 M Q, which consistently reduced both responses. Moreover, S provoked increases in both MO and LL at 0.1 M while NaCl and water elicited minimal effects on GFR. These data suggest that perinatal undernutrition affects the basic components of the gustatory system necessary to produce GFR in the first postnatal days by possibly interfereing with the integration of taste input for food learning and the hedonic aspects of gustatory cues.
1. Introduction
In the past, a number of studies have shown that perinatal undernutrition in the rat delays the maturation of sensory systems and causes the expression of altered motor patterns; these effects lead to a significant deficiency of somatosensory, auditory, visual, and chemosensory stimulation that may be essential for long-term behavioral performance [1-8]. Perinatal undernutrition mainly interferes with the anatomical organization of central nervous system (CNS) areas which undergo a phase of postnatal cell proliferation; these areas include the cerebral and cerebellar cortices, the hippocampus, central amygdaloid nucleus, olfactory bulb, and solitary tract nucleus, and they participate in the head movements, mouth opening and closing, chewing, food ingestion, and chemosensory CNS integration [8,9-13]. Early undernutrition affects neuronal development by reducing the cell number and their dendritic branching, and by the formation of spines with small perikarya alterations that interfere with their connectivity and neuronal interactions [7,8,14-16].
The GFR evoked by the activation of gustatory receptors in the oral cavity generates two physiological processes, one related to the quality characteristics of the taste (sensorial) for food learning, and the other to the hedonic aspect (reward-aversion) of taste [17,18]. Because acceptance or rejection of taste stimuli depends on the hedonic rather than the sensory aspect, the hedonic component of taste stimulus may be indirectly evaluated by using relative fluid consumption tests or by analyzing taste mimetic responses [19-21]. Furthermore, the initial activation of specific taste receptors in the oral cavity triggers electrical impulses which are transmitted along two separate sensory and hedonic afferent pathways that are constituted by different relays and that ultimately reach the insular and the orbitofrontal cortices, respectively [22-24].
Although no facial motor reaction is specific to a particular taste sensation, there are clear variations of GFR with age and with the concentration of the different tastes. It is also known that an observer can make a good evaluation of a newborn’s facial expression because it shows reliable components of taste palatability or hedonic impact [17,25].
To our knowledge there is a lack of information about how perinatally underfed pups discriminate among different taste cues, and if the GFR evoked is different from that of normally fed pups. The current study investigates if the gustatory system of early underfed rats is functionally modified shortly after birth, as defined by MO and LL in response to different gustatory cues.
2. Materials and Methods
2.1. Animals
Experiments were approved by local Animal Committees and were in accordance with the NIH Guide for Care and Use of Laboratory Animals. Subjects were male and female Wistar rats (Rattus norvegicus), born and reared in the animal colony at the Institute of Neurobiology, University of Mexico. All animals were maintained in an automatically controlled room at 22˚C ± 2˚C, 50% humidity, and a 12 h light/dark cycle (lights on at 7:00). For mating, a male was placed in a plastic cage (60 × 32 × 20 cm) containing three females (200 - 250 g). Spermpositive females were placed in individual plastic maternity cages (35 × 27 × 17 cm) with grill tops and wood shavings as nesting material one week before parturition. The day of birth was referred to as PN 0, and 24 h later pups were randomly mixed, redistributed and adjusted to 8 pups per mother (four males and four females). The redistribution was intended to balance possible genetic and prenatal biological differences between litters and give them equal probability of development.
2.2. Nutritional Procedures
2.2.1. Undernourished Group (UG)
The UG males and females (n = 56) came from at least nine different litters. Pregnant dams were fed from G6 to G12 with 50% (7.8 g) of a balanced diet (Purina chow) received by a normal dam, from G13 to G18 with 70% (10.9 g), and with the 100% (15.6 g) of the same diet until parturition, to avoid cannibalism of pups (Figure 1). After birth, prenatally underfed newborns were nursed by a pair of gestationally underfed dams, one of whose main galactophorous ducts had been subcutaneously tied [26]. To continue the neonatal underfeeding method, these two lactating dams were interchanged between litters every 12 h from postnatal days 1 to 9. This cross-fostering procedure reduces the effects on the newborns of maternal sensory deprivation that may interfere with the expression of GFR [2,26,27]. This paradigm was chosen because the oromotor reponse is regulated at the brainstem level, and most brain stem neurogenesis occurs

Figure 1. Schematic representation of the underfeeding procedure.
prenatally following first a rostrocaudal and then a mediolateral cytogenetic gradient; facial nerve efferent projections to the facial muscles and taste buds also occur during this period and continue during the early postnatal period [28,29]. Approximately 80% of the total underfed subjects included here were undernourished during the light phase and the remaining 20% during the dark phase of the cycle.
2.2.2. Control Group (CG)
The CG subjects consisted of 56 male and female pups obtained from nine, well-nourished litters, nursed by well-fed dams that had free access to water and balanced food (Purina chow) during gestation. After birth, CG pups were fed by two, normally lactating mothers, who were interchanged every 12 h between litters. To evaluate the effects of the nutritional treatment on body weights of subjects from the different dietary conditions a different group of six rats in each treatment was sampled.
2.3. Behavioral Testing
All GFR tests were performed between 10:00 and 12:00 in a sound-proof chamber that was illuminated with red light (60 W), maintained at 26˚C ± 2˚C, and separated from the ambient noise of the main laboratory. The GFR test was carried out with a minor modification of the procedure previously described for neonatal rats [20]. A total of fourteen groups were used, including control (CG) and undernourished groups (UG), each with 4 males and 4 females. The animals were labeled with different colors; for each stimulus, subjects came from different litters, and the sapid stimuli tested were as follows: water (CGW) and (UGW) and three different sapid solutions diluted with distilled water to two different concentrations: 0.1 M and 0.3 M sucrose (CGS) and (UGS), 0.1 M and 0.3 M NaCl (CG NaCL) and (UG NaCl); and 0.01 M and 0.1 M quinine (CGQ) and (UGQ); each group was tested at different postnatal ages: 1, 3, 5, 7, and 9 days. As a preliminary study in addition to the water stimulus we used only gustatory cues with an opposite palatability, and a neutral stimulus to characterize the GFR. Pups under the two dietary treatments were maintained with the mother in their habitat for at least 30 min before the gustatory stimulation test began. The taste stimuli were presented centrally on the pup’s lips as a single droplet (about 8 µl) via a 2-cm length of polyethylene tubing (P. E. 160) attached to a blunted tip of a 2.0-mm plastic micropipette. A single micropipette was used for each tastant during the recording tests. The hand-held and relaxed, swaddled pups were observed and videotaped for 10 sec before recording the sapid solution base line, and then for a 1-min interval under the effect of a tastant until the droplet disappeared into the pup’s mouth under red-lamp illumination (Figure 1). Because the newborn rats possess reliable mimetic rhythmic facial, head, and body movements in response to oral gustatory stimulation, in each behavioral test both the MO and LL of the pups were analyzed. These early mimetic responses have been shown to be a reliable index of taste sensitivity associated with the GFR [19,20,30]. To ensure blind observations with respect to taste stimulus presentation, the different dietary treatments and gustatory exposure sequences were randomly modified from one test to the next. Because of the small body size of neonatal rats, and because a certain level of experience is required for accurate recording of GFR events, some experiments included data from naive, partially experienced, and very experienced observers to assess the reliability of the measurements.
2.4. Statistical Analyses
The statistical package Statistica, version 6 was used to perform all data comparisons body weight differences between the CG and UG subjects were compared by using a two-way ANOVA, (diet) × (age). To analyze the MO and LL score differences in the case of the W stimulus, a two-way ANOVA (diet) × (age) was used. MO and LL differences in each gustatory cue were analyzed with a two-way ANOVA, 2 dietary conditions (control and undernutrition) × 5 (ages), followed by a post hoc, Fisher LSD test where appropriate. Statistical significance was defined as p < 0.05.
3. Results
3.1. Body Weight
Preliminary comparisons between males and females showed no significant differences, and they were then combined in the same group. Body weight comparisons among experimental groups showed that UG subjects weighed significantly less than CG. Body weight was also modified by age in both dietary conditions. Additionally, significant interactions between the diet and age factors were detected. Post hoc comparisons at each developmental age indicated consistently low body weight in the UG throughout the study as compared to their controls. In summary, the data showed that the perinatal undernutrition paradigm interfered consistently with the physical growth of undernourished subjects during the study (Table 1).
3.2. Effects of W on GFR
Water was utilized to examine the GFR to a neutral stimulus and also because the other sapid stimuli were diluted in water. The ANOVA comparisons of MO and LL following W application were not modified by diet or by age (data not shown). The mean values of the scores obtained over the study are indicated in Figure 2.
3.3. Effects of NaCl on GFR
The ANOVA comparisons of the MO after exposure to 0.1 M NaCl showed no modification by the diet, but a significant difference with age, F(4,56) = 51.20, (p < 0.05) and a significant interaction between diet and age, F(4,56) = 2.52, (p < 0.05). The LL following 0.1 M NaCl was affected by the diet, F(1,14) = 24.36, (p < 0.05) but was not modified by age; furthermore, a significant interaction between the diet and age, F(4,56) = 8.21, (p < 0.05) was observed. Post hoc comparison of LL between UG NaCl 0.1 and CG NaCl 0.1 shows a reduction in UG NaCl 0.1 on days 1, 5, and 9 of age, but a significant increase on day 3 of age (Figure 2(b), low left pannels).
The MO after exposure to 0.3 M NaCl was only modified by age, F(4,56) = 15.03, (p < 0.05) (Figure 2(b), upper right pannels). The ANOVA comparison of the LL shows significant differences associated only with age, F(4,56) = 2.95, (p < 0.05) (Figure 2(b), low right pannels).
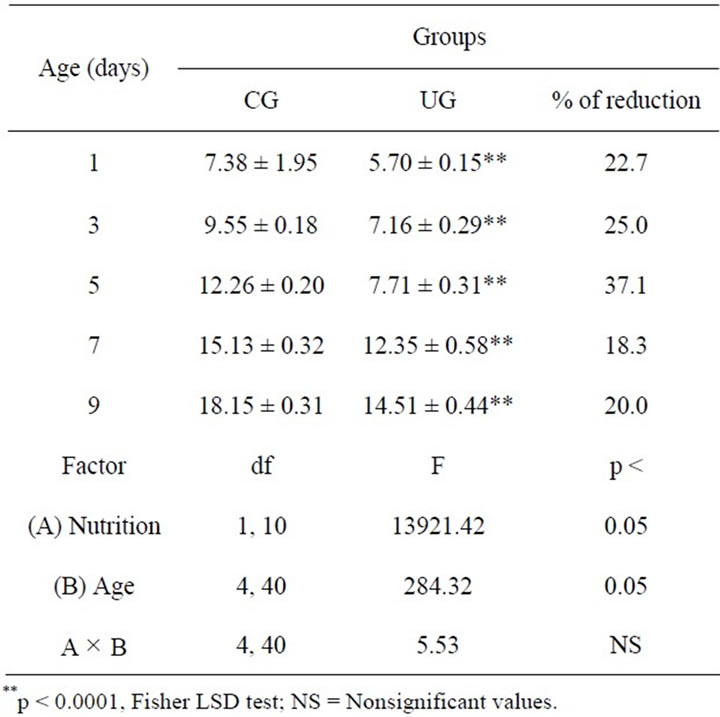
Table 1. Body weight (± SEM g) of pups reared by control and perinatally underfed dams (n = 6) during development.

Figure 2. Mean frequency of MO and LL at different ages in response(s) to different tastants: (a) sucrose; (b) NaCl; and (c) quinine in both CG and UG groups. Squares (left) represent the low and triangles (right) the high concentration. Solid symbols represent the (CG) and empty symbols the underfed (UG). Note that the MO in UGS 0.1 M and UGQ 0.01 M are increased compared to the corresponding CGS on days 1 and 3 of age. Comparing the same groups at day 3 LL is increased in UGS 0.1 M, and in UGQ 0.01 M. Water (W);*Significant differences associated with diet (p < 0.05).
3.4. Effects of Sucrose (S) on GFR
Statistical comparisons of the MO provoked by S 0.1 was modified by diet, F(1,14) = 23.12, (p < 0.05) and by age, F(4,56) = 22.93, (p < 0.05). Moreover, a significant interaction between diet and age, F(4,56) = 22.93, (p < 0.05) was observed. Post hoc comparisons between UGS 0.1 and CGS 0.1 indicate a significant increase on days 1 and 3 of age without differences on day 5 and a significant reduction on days 7 and 9 (Figure 2(a), upper left pannels). ANOVA comparison of the LL elicited by S 0.1 shows that it was modified only by diet, F(1,14) = 14.23, (p < 0.05) with a significant interaction between diet and age, F(4,56) = 18.21, (p < 0.05). Post hoc comparison shows a significant reduction of LL in UGS 0.1 on day 1 compared with CGS 0.1; in contrast, on days 3, 7, and 9 it shows a significant increase in UGS 0.1 compared with CGS 0.1 (Figure 2(a), low left pannels).
MO in S 0.3 was modified by diet, F(1,14) = 9.09, (p < 0.05) and by age, F(4,56) = 26.55, (p < 0.05), and a significant interaction between these factors F(4,56) = 9.57, (p < 0.05). Post hoc comparison shows a significant increase in UGS 0.3 compared with CGS 0.3 on day 1 of age, but the other ages analyzed did not reveal significant effects (Figure 2(a), upper right pannels). ANOVA comparison of the LL elicited by S 0.3 shows significant differences only due to age, F(4,56) = 5.07, (p < 0.05) and a significant interaction diet by age, F(4,56) = 3.27, (p < 0.05). Post hoc comparisons show a significant decrease in UGS 0.3 relative to CGS 0.3 only on day 1 of age, with significant increase on day 3 (p < 0,05) (Figure 2(a), low right pannels).
3.5. Effects of Quinine (Q) on GFR
ANOVA comparison of the MO in response to Q 0.01 shows significant differences due to diet, F(1,14) = 34.79, (p < 0.05) and age, F(4,56) = 19.25, (p < 0.05) and a significant interaction between the diet and age, F(4,56) = 9.90, (p < 0.05). Post hoc comparisons between UGQ 0.01 and CGQ 0.01 indicated a significant increase (p < 0.05) on days 1 and 3 of age (Figure 2(c), upper left pannels). Statistical comparisons of the LL in responses to Q 0.01 did not show significant differences due to diet, but the LL changed with age, F(4,56) = 6.03, (p < 0.05), and there was a significant interaction between diet and age, F(4,56) = 12.73, (p < 0.05). Post hoc comparisons show that the LL in UGQ 0.01 is reduced on days 1 and 7 compared CGQ and there is a significant increase at 3 days of age (Figure 2(c), low left pannels).
Comparisons of the MO in response to Q 0.1 do not show significant differences due to diet, but MO was modified by age, F(4,56) = 15.60, (p < 0.05) without a significant interaction effect. Post hoc comparisons show a significant reduction (p < 0.05) in UGQ 0.1 compared to CGQ 0.1 on day 9 of age (Figure 2(c), upper right panels). ANOVA comparison of LL shows significant differences due to diet, F(1,14) = 24.03, (p < 0.05) and to age, F(4,56) = 3.90, (p < 0.05) without interaction. Post hoc comparisons showed a significant reduction of LL in UGQ 0.1 relative to CGQ on days 1, 3, 7, and 9 of age (Figure 2(c), low right pannels). In summary the ANOVA comparisons indicated consistent effects associated with age, with minimal effects of the diet, except in the UG subjects that exhibited an increase in MO and a decrease of LL with 0.01 M Q, and consistently reduced responses with 0.1 M Q.
4. Discussion
The present results indicated that perinatal undernutrition may induce functional alterations that may, in turn, modify the oromotor expressions in the UG subjects in response to aversive and pleasant gustatory cues. These behavioral alterations may be relevant because they could be reflecting the disruption of UG pup’s hedonic component during the early associative gustatory learning that occurred in the maternal environment and is fundamental for the newborn survival [31-33]. The present data also indicate that the versatile oromotor responses to different types and concentrations of tastants change depending on several factors, including age, mother-litter bonds, and nutritional conditions of the pups [33-35]. Although newborns exhibit a complex of mimetic facial responses, head, body, and flexor-extensor movements in responses to different tastants [20]; according to our previous observations the MO and LL responses were the more reliable measurements of taste responsivity. The statistical comparisons of the different low and high tastants concentrations indicated consistent significant interactions between diet and age in the case of S, and only to a low NaCl and Q concentrations. These findings suggest that the gustatory circuits organization of the C and U subjects at each developmental age, and the two tastants concentrations could be reciprocally influenced in the case of the S, and partially by the NaCl and Q stimuli concentrations. By contrast, the interactions between diet and age are lost when the NaCl and Q at high concentrations are presented, because they probably saturate the circuits functional capabilities; although, this possibility requires further investigation.
The GFR alterations in response to tastants found in the present study are in line with neuronal hypoplasia and the disorganization of the dendritic tree and spines observed along the relay nuclei of the gustatory and motor facial nucleus [7,13,15,36,37]. In this regard, it is known that descending input from the insular cortex, lateral hypothalamus, and central amygdale modulate the brainstem sensory and motor mechanisms of the newborn to produce fluid and food intake, nipple attachment, and the GFR integration that seems to be disrupted in the UG animals studied here [19,38-41]. Thus, it is possible that early in life brainstem structures that modulate GFR and are widely connected to forebrain areas may be vulnerable to food restriction and altered mother-litter bonds that disrupt various adaptive behavioral processes related to the early physiological needs of the newborn [35,42]. The fact that W and NaCl stimulation provoked negligible MO and LL responses may be related to the reduction of sensitivity to these two stimuli that maintain a relatively constant concentration as normal components of the amniotic fluid in the uterine environment during gestation [43-45]. Additionally, in the present study, the somatosensory system could participate in orosensory perception of different tastes, because the taste stimuli were diluted in water and oral movements would facilitate their spread to all parts of the oral cavity. Thus, we can not discard the possibility that tactile signals could contribute to these orofacial responses because palatability depends on their sensory characteristics of the food, which are detected by the taste trigeminal, and olfactory systems; more specifically, behavioral studies have shown that palatability can be modulated by trigeminal input [41,46].
Another point of interest concerns the fact that in the UG subjects there were reduced MO and LL, in response to Q administration, suggesting then that they preserve the aversive responsivity to unpleasant and possibly toxic tastants. This is a relevant finding because the UG animals maintain their capacity to gain experience through associative learning, and to revert the cognitive deficiencies commonly associated with perinatal undernutrition, by the combination of food and early sensory rehabilitation [5,6,33,47-49]. However, further studies are required to test that in the wild life these mechanisms are over expressed as in the case of UG subjects, surrounded by an environmental condition that frequently elicits stressful responses.
5. Conclusion
The basic brain stem mechanisms underlying the GFR arise during gestation and they are affected by prenatal and neonatal food restriction. This conclusion is supported by the increase in MO and LL in response to low sucrose concentration, and the increased MO and reduced LL in response to low Q concentration during the first days of life. By contrast W and NaCl stimulation provoked negligible affects on MO and LL. The current findings could be helpful in the design of protocols to study of early associated learning guided by gustatory stimulation.
6. Acknowledgement
This work was partly supported by DGAPA/UNAM. IN 207310. We thank Dr. D. Pless for editorial assistance.
REFERENCES
- D. Callison and J. Spencer, “Effect of Chronic Undernutrition and/or Visual Deprivation upon the Visual Evoked Potentials from the Developing Rat Brain,” Developmental Psychobiology, Vol. 1, No. 4, 1968, pp. 196-204. doi:10.1002/dev.420010308
- L. S. Crnic, J. M. Bell, M. Mangold, J. Gruenthal and S. Finger, “Separation-Induced Early Malnutrition: Maternal, Physiological and Behavioral Effects,” Physiology & Behavior, Vol. 26, No. 4, 1980, pp. 695-707. doi:10.1016/0031-9384(81)90147-5
- J. R. Galler and K. J. Propert, “Maternal Behavior Following Rehabilitation of Rats with Intergenerational Malnutrition. 1. Persistent Changes in Lactation-Related Behaviors,” Journal of Nutrition, Vol. 111, No. 8, 1981, pp. 1330-1336.
- M. Salas, C. Torrero, M. Regalado, M. Martinez-Gomez and P. Pacheco, “Dendritic Arbor Alterations in the Medial Superior Olivary Neurons of Neonatally Underfed Rats,” Acta Anatomica, Vol. 151, No. 3, 1994, pp. 180- 187. doi:10.1159/000147661
- M. Salas, M. Regalado and C. Torrero, “Recovery of Long-Term Maternal Behavioral Deficiencies of Neonatally Underfed Rats by Early Sensory Stimulation: Effects of Successive Parturitions,” Nutritional Neuroscience, Vol. 4, No. 4, 2001, pp. 311-322.
- O. Soriano, M. Regalado, C. Torrero and M. Salas, “Contributions of Undernutrition and Handling to Huddling Development of Rats,” Physiology & Behavior, Vol. 89, No. 4, 2006, pp. 543-551. doi:10.1016/j.physbeh.2006.07.021
- L. Rubio, C. Torrero, M. Regalado and M. Salas, “Alterations in the Solitary Tract Nucleus of the Rat Following Perinatal Food Restriction and Subsequent Nutritional Rehabilitation,” Nutritional Neuroscience, Vol. 7, No. 5-6, 2004, pp. 291-300. doi:10.1080/10284150400019922
- C. Frias, C. Torrero, M. Regalado and M. Salas, “Development of Mitral Cells and Olfactory Bulb Layers in Neonatally Undernourished Rats,” Nutritional Neuroscience, Vol. 12, No. 3, 2009, pp. 96-104. doi:10.1179/147683009X423238
- N. H. Bass, M. G. Netsky and E. Young, “Effects of Neonatal Malnutrition on Developing Cerebrum.1. Microchemical and Histologic Study of Cellular Differentiation in the Rat,” Archives of Neurology, Vol. 23, No. 4, 1970, pp. 289-302. doi:10.1001/archneur.1970.00480280003001
- C. D. West and T. Kemper, “The Effect of a Low Protein Diet on the Anatomical Development of the Rat Brain,” Brain Research, Vol. 107, No. 2, 1976, pp. 221-237. doi:10.1016/0006-8993(76)90223-7
- P. McConnell and M. Berry, “The Effect of Refeeding after Neonatal Starvation of Purkinje Cell Dendritic Growth in the Rat,” The Journal of Comparative Neurology, Vol. 178, No. 2, 1978, pp. 759-772. doi:10.1002/cne.901780410
- V. C. Abrahams, A. A. Kori, G. E. Loeb, F. J. Richmond and S. A. Keirstead, “Facial Input to Neck Motoneurons: Trigemino-Cervical Reflexes in the Conscious and Anaesthetized Cat,” Experimental Brain Research, Vol. 97, No. 1, 1993, pp. 23-30. doi:10.1007/BF00228814
- E. Perez-Torrero, C. Torrero and M. Salas, “Effects of Perinatal Udernourishment on Neuronal Development of the Facial Motor Nucleus in the Rat,” Brain Research, Vol. 905, No. 1-2, 2001, pp. 54-62. doi:10.1016/S0006-8993(01)02500-8
- M. Salas, S. Diaz and A. Nieto, “Effects of Neonatal Food Deprivation on Cortical Spines and Dendritic Development of the Rat,” Brain Research, Vol. 73, No. 1, 1974, pp. 139-144. doi:10.1016/0006-8993(74)91012-9
- C. Escobar and M. Salas, “Neonatal Undernutrition and Amygdaloid Nuclear Complex Development: An Experimental Study in the Rat,” Experimental Neurology, Vol. 22, No. 2, 1993, pp. 311-318. doi:10.1006/exnr.1993.1130
- C. Frias, C. Torrero, M. Regalado and M. Salas, “Organization of Olfactory Glomeruli in Neonatally Undernourished Rats,” Nutritional Neuroscience, Vol. 9, No. 2, 2006, pp. 49-55. doi:10.1080/10284150500506042
- K. C. Berridge, “Measuring Hedonic Impact in Animals and Infants: Microstructure of Affective Taste Reactivity Patterns,” Neuroscience & Biobehavioral Reviews, Vol. 24, No. 2, 2000, pp. 173-198. doi:10.1016/S0149-7634(99)00072-X
- T. V. Sewards, “Dual Separate Pathways for Sensory and Hedonic Aspects of Taste,” Brain Research Bulletin, Vol. 62, No. 4, 2004, pp. 271-283. doi:10.1016/j.brainresbull.2003.10.004
- H. J. Grill and R. Norgren, “The Taste Reactivity Test. I. Mimetic Responses to Gustatory Stimuli in Neurologically Normal Rats,” Brain Research, Vol. 143, No. 2, 1978, pp. 263-279. doi:10.1016/0006-8993(78)90568-1
- J. D. Ganchrow, J. E. Steiner and S. Canetto, “Behavioral Displays to Gustatory Stimuli in Newborn Rat Pups,” Developmental Psychobiology, Vol. 19, No. 3, 1986, pp. 163-174. doi:10.1002/dev.420190303
- S. A. Shumake, S. E. Gaddis and K. A. Crane, “Liquid Baits for Rodent Control: A Comparison of Wild Norway versus Wild Ricefield Rat Response to Glucose Plus Saccharin Solutions,” Crop Protection, Vol. 16, No. 1, 1997, pp. 33-39. doi:10.1016/S0261-2194(96)00053-1
- T. Yamamoto, R. Matsuo, Y. Kiyomitsu and R. Kitamura, “Taste Responses of Cortical Neurons in Freely Ingesting Rats,” Journal of Neurophysiology, Vol. 61, No. 6, 1989, pp. 1244-1258.
- E. T. Rolls, “The Orbitofrontal Cortex and Reward,” Cerebral Cortex, Vol. 10, No. 3, 2000, pp. 284-294. doi:10.1093/cercor/10.3.284
- T. V. Sewards and M. Sewards, “Separate, Parallel Sensory and Hedonic Pathways in the Mammalian Somatosensory System,” Brain Research Bulletin, Vol. 58, No. 3, 2002, pp. 243-260. doi:10.1016/S0361-9230(02)00783-9
- J. E. Steiner, D. Glaser, M. E. Hawilo and K. C. Berridge, “Comparative Expression of Hedonic Impact: Affective Reactions to Taste by Human Infants and Other Primates,” Neuroscience & Biobehavioral Reviews, Vol. 25, No. 1, 2001, pp. 53-74. doi:10.1016/S0149-7634(00)00051-8
- A. Lynch, “Postnatal Undernutrition: An Alternative Method,” Developmental Psychobiology, Vol. 9, No. 1, 1976, pp. 39-48. doi:10.1002/dev.420090107
- D. L. A. Van den Hove, H. W. M. Steinbusch, A. Scheepens, W. D. J. Van de Berg, L. A. M. Kooiman, B. J. G. Boosten, J. Prickaerts and C. E. Blanco, “Prenatal Stress and Neonatal Rat Brain Development,” Neuroscience, Vol. 137, No. 1, 2006, pp. 145-155. doi:10.1016/j.neuroscience.2005.08.060
- J. S. Altman and S. Bayer, “Development of the Brain Stem in the Rat. II. Thymidine-Radiographic Study of the Time of Origins of Neurons of the Upper Medulla, Excluding the Vestibular and Auditory Nuclei,” The Journal of Comparative Neurology, Vol. 194, No. 1, 1980, pp. 37- 56. doi:10.1002/cne.901940103
- S. Harada, K. Yamaguchi, N. Kanemaru and Y. Kasahara, “Maturation of Taste Buds on the Soft Palate of the Postnatal Rat,” Physiology & Behavior, Vol. 68, No. 3, 2000, pp. 333-339. doi:10.1016/S0031-9384(99)00184-5
- W. G. Hall and T. E. Bryan, “The Ontogeny of Feeding in Rats: IV. Taste Development as Measured by Intake and Behavioral Responses to Oral Infusions of Sucrose and Quinine,” Journal of Comparative & Physiological Psychology, Vol. 95, No. 2, 1981, pp. 240-251. doi:10.1037/h0077771
- R. M. Sullivan and D. A. Wilson, “Molecular Biology of Early Olfactory Memory,” Learning & Memory, Vol. 10, No. 1, 2003, pp. 1-4. doi:10.1101/lm.58203
- R. M. Sullivan, R. D. A. Wilson, J. Feldon, B. K. Yee, U. Meyer, G. Richter-Levin, A. Avi, T. Michael, M. Gruss, J. Bock, C. Helmeke and K. Braun, “The International Society for Developmental Psychobiology Annual Meeting Symposium: Impact of Early Life Experiences on Brain and Behavioral Development,” Developmental Psychobiology, Vol. 48, No. 7, 2006, pp. 583-602. doi:10.1002/dev.20170
- M. Ruiz-Diaz, C. Torrero, M. Regalado and M. Salas, “Perinatal Undernourishment and Handling: Effects on Olfactory Discrimination in the Newborn Rat,” Nutritional Neuroscience, Vol. 13, No. 4, 2010, pp. 1-9.
- T. Hiroshige, K. Honma and K. Watanabe, “Possible Zeitgebers for External Entrainment of the Circadian Rhythm of Plasma Corticosterone in Blind Infantile Rats,” Journal of Physiology (London), Vol. 325, No. 1, 1982, pp. 507-519.
- V. K. Sharma, R. Chindambaran, R. Subbaraj and M. K. Chandrashekaran, “Effects of Restricted Feeding Cycle on the Locomotor Activity in the Mouse Musbooduga,” Physiology & Behavior, Vol. 70, No. 1-2, 2000, pp. 81-87. doi:10.1016/S0031-9384(00)00217-1
- M. Salas, “Effects of Early Undernutrition on Dendritic Spines of Cortical Pyramidal Cells in the Rat,” Developmental Neuroscience, Vol. 3, No. 3, 1980, pp. 109-117. doi:10.1159/000112384
- J. P. Andrade, M. D. Madeira and M. Paula-Barbosa, “Effects of Long-Term Malnutrition and Rehabilitation on the Hippocampal Formation of the Adult Rat. A Morphometric Study,” Journal of Anatomy, Vol. 187, No. 2, 1995, pp. 379-393.
- P. M. Di Lorenzo and S. Monroe, “Corticofugal Influence on Taste Responses in the Nucleus of the Solitary Tract in the Rat,” Journal of Neurophysiology, Vol. 74, No. 1, 1995, pp. 258-272.
- H. Nishijo, T. Uwano, R. Tamura and T. Ono, “Gustatory and Multimodal Neuronal Responses in the Amygdala during Licking and Discrimination of Sensory Stimuli in Awake Rats,” Journal of Neurophysiology, Vol. 79, No. 1, 1998, pp. 21-36.
- Y. K. Cho, C. S. Li and D. V. Smith, “Descending Influences from the Lateral Hypothalamus and Amygdala Converge onto Medullary Taste Neurons,” Chemical Senses, Vol. 28, No. 2, 2003, pp. 155-171. doi:10.1093/chemse/28.2.155
- R. F. Lundy Jr., “Gustatory Hedonic Value: Potential Function for Forebrain Control of Brainstem Taste Processing,” Neuroscience & Biobehavioral Reviews, Vol. 32, No. 8, 2008, pp. 1601-1606. doi:10.1016/j.neubiorev.2008.07.002
- N. Viswanathan and M. K. Chandrashekaran, “Cycles of Presence and Absence of Mother Mouse Entrain the Circadian Clock of the Pups,” Nature, Vol. 317, No. 10-16, 1985, pp. 530-531.
- P. P. L. Tam and S. T. H. Chan, “Changes in the Composition of Maternal Plasma, Fetal Plasma and Fetal Extraembryonic Fluid during Gestation in the Rat,” Journal of Reproduction and Fertility, Vol. 51, No. 1, 1977, pp. 41-51. doi:10.1530/jrf.0.0510041
- H. B. Greizerstei, “Placental and Fetal Composition during the Last Trimester of Gestational in the Rat,” Biology of Reproduction, Vol. 26, No. 5, 1982, pp. 847-853. doi:10.1095/biolreprod26.5.847
- E. Erkadius, T. O. Morgan and R. Di Nicolantonio, “Amniotic Fluid Composition and Fetal and Placental Growth Rates in Genetically Hypertensive and Normotensive Rats,” Reproduction, Fertility and Development, Vol. 7, No. 6, 1995, pp. 1563-1567. doi:10.1071/RD9951563
- K. C. Berridge and J. C. Fentress, “Trigeminal-Taste Interaction in Palatability Processing,” Science, Vol. 228, No. 10, 1985, pp. 747-750. doi:10.1126/science.3992242
- T. C. Jordan, S. E. Cane and K. F. Howells, “Deficits in Spatial Memory Performance Induced by Undernutrition,” Developmental Psychobiology, Vol. 14, No. 4, 1981, pp. 317-325. doi:10.1002/dev.420140404
- M. R. Rosenzweig and E. L. Bennett, “Psychobiology of Plasticity: Effects of Training and Experience on Brain and Behavior,” Behavioural Brain Research, Vol. 78, No. 5, 1996, pp. 57-65. doi:10.1016/0166-4328(95)00216-2
- C. A. Turner, M. H. Lewis and M. A. King, “Environmental Enrichment: Effects on Stereotyped Behavior and Dendritic Morphology,” Developmental Psychobiology, Vol. 43, No. 1, 2003, pp. 20-27. doi:10.1002/dev.10116
NOTES
*The authors declare that there are no conflicts of interests concerning this research.

