 Open Journal of Endocrine and Metabolic Diseases, 2013, 3, 252-258 http://dx.doi.org/10.4236/ojemd.2013.35034 Published Online September 2013 (http://www.scirp.org/journal/ojemd) Amelioration of Albuminuria in Japanese Type 2 Diabetic Patients by Maximal Dose of Candesartan* Yoichi Oikawa1,2, Akira Shimada1, Mizumi Kyo2 1Department of Internal Medicine, Tokyo Saiseikai Central Hospital, Tokyo, Japan 2Department of Internal Medicine, Eiju General Hospital, Tokyo, Japan Email: oikawa@saichu.jp Received August 12, 2013; revised August 31, 2013; accepted September 10, 2013 Copyright © 2013 Yoichi Oikawa et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. ABSTRACT Introduction: It was recently reported that candesartan, an angiotensin II receptor blocker, had a protective effect against cardiovascular events, comparable to that of calcium channel antagonists. Moreover, a renoprotective effect and anti-diabetic action of candesartan had also been demonstrated. However, whether the renoprotective effect of cande- sartan, especially in diabetes, was dose-dependent or not remain to be fully elucidated. The present study attempted to clarify the dose effect of renoprotection by candesartan in Japanese type 2 diabetic patients. Subjects and Method: In this case series study, we recruited 26 type 2 diabetic patients with albuminuria whose blood pressure did not reach the target BP level (<130/80 mmHg) despite administration of 4 or 8 mg/day of candesartan. Subsequently, these lower doses of candesartan were increased to the maximal dose in Japan, 12 mg/day. Clinical parameters were examined be- fore, at 6 and 12 months after the increase in dose. Results: An ameliorating effect of the increased dose of candesartan on albuminuria and hypertension was distinctly observed. No severe adverse effect was observed. Conclusion: It was highly possible that the maximal dose of candesartan provided more effective renoprotection in hypertensive type 2 dia- betic patients initially treated with lower doses of candesartan. Keywords: Albuminuria; Angiotensin II Receptor Blocker (ARB); Candesartan; Hypertension; Type 2 Diabetes 1. Introduction Diabetes is a chronic disease that occurs either when the pancreas does not produce enough insulin or when the body cannot effectively use the insulin it produces. Dia- betes without proper treatment can cause many complica- tions, such as diabetic retinopathy, nephropathy and neu- ropathy. Diabetes, as well as hypertension, dyslipidemia and obesity, is believed to be a strong risk factor for cardiovascular disease [1]. Especially, the existence of albuminuria, a characteristic of diabetic early nephro- pathy, is considered to be an important risk factor for increased cardiovascular events in diabetic patients [2]. Recently, the number of new cases of dialysis due to diabetic nephropathy has been increasing in a linear fa- shion in Japan [3], implying that prevention of the de- velopment of diabetic nephropathy is absolutely impera- tive. It is generally considered that the prevention of dia- betic nephropathy requires strict blood pressure (BP) control as well as good control of the blood glucose level and an appropriate protein-restricted diet. At present, the Japanese Society of Hypertension Guidelines for the Ma- nagement of Hypertension (JSH 2009) recommend that the target systolic and diastolic BP in diabetic patients are set at <130 mmHg and <80 mmHg, respectively [4], and <125 mmHg and <75 mmHg in patients with more than 1 g/day of proteinuria, respectively. However, the rate of achievement of the target BP seems to be low and is not be satisfactory in clinical settings. The renin-angiotensin system (RAS) plays a critical role in the pathogenesis of chronic kidney disease (CKD) as well as hypertension and cardiovascular events [5], and inhibition of RAS by angiotensin II receptor blockers (ARB) is known to confer a protective action not only on renal function, but also on cardiovascular organs in dia- betic [6] as well as non-diabetic [7] hypertensive patients with CKD. On the other hand, it is unknown whether the renoprotective effect of ARBs, especially in diabetics, is dose-dependent or not. Regarding this point, the incipient to overt, angiotensin II blocker, telmisartan, investiga- tion on type 2 diabetic nephropathy (INNOVATION) *Disclosure Statement: The Authors declare that there is no conflict of interest. C opyright © 2013 SciRes. OJEMD  Y. OIKAWA ET AL. 253 study demonstrated a higher remission rate of microal- buminuria in Japanese type 2 diabetic patients with al- buminuria administered telmisartan as compared to pla- cebo [8]. However, when the telmisartan-treated subjects were divided into high-dose and low-dose groups, no significant difference in the remission rate of microalbu- minuria was observed between the two. The Candesartan Antihypertensive Survival Evalua- tion in Japan (CASE-J) trial recently revealed a protect- tive effect of candesartan against cardiovascular events comparable to that of amlodipine and an anti-diabetic effect [9]. In addition, sub-analysis of the CASE-J trial also showed a renoprotective effect of candesartan in hypertensive patients with CKD [10]. However, the dose effect of renoprotection by candesartan in diabetics re- mains to be elucidated. To investigate whether the reno- protective effect of candesartan in diabetes is dose-de- pendent or not, we increased the prescribed amount of candesartan from lower doses to the maximal dose in Japanese type 2 diabetic patients with hypertension and albuminuria, and evaluated the time course of a variety of clinical parameters including urine albumin excretion (UAE) level. 2. Subjects and Method 2.1. Study Design This study was an uncontrolled longitudinal observa- tional study; i.e. a case series study. Fifty-six Japanese type 2 diabetic patients with hyper- tension, whose BP did not reach the target systolic/dia- stolic BP (<130/80 mmHg) despite administration of 4 or 8 mg/day of candesartan for more than 6 months, were recruited at Eiju General Hospital. Of these patients, 26 were shown to have albuminuria without overt proteinu- ria, and their baseline UAE level in a single spot-urine collection ranged from 31.2 to 620.0 mg/gCr (creatinine). After the need for an increase in dose to reach the target BP level was approved by the 26 patients, the dose of candesartan (4 mg/day or 8 mg/day) was increased to the maximal dose in Japan, i.e. 12 mg/day. Venous blood samples were obtained from the patients regardless of fasting or non-fasting state and a variety of clinical pa- rameters were investigated before, at 6 months and 12 months after the increase in dose. Exclusion criteria were as follows; type 1 diabetes, patients with stage I (UAE level <30 mg/gCr), IIIb (urine protein level ≥1 g/day or estimated glomerular filtration rate (eGFR) <60 mL/ min/1.73 m2), IV (increased serum creatinine level above normal range) or V (receiving dialysis) diabetic neph- ropathy, serum potassium level >5.5 mEq/L, unconsent- ing patients, overt bacteriuria, severe autonomic neu- ropathy including orthostatic hypotension, seated systolic BP/diastolic BP ≥180/100 mmHg, severe liver dysfunc- tion and definable chronic kidney disease other than dia- betic nephropathy. Hemoglobin A1c (HbA1c) was expressed as a NGSP (National Glycohemoglobin Standardization Program) equivalent value in this study; i.e., HbA1c (NGSP equi- valent value) (%) = HbA1c (Japan Diabetes Society value) (%) + 0.4%. 2.2. Statistical Analysis Data are expressed as mean S.D. Continuous variables were compared using paired t-test. A P value less than 0.05 was considered to be statistically significant. 3. Results 3.1. Patients’ Baseline Characteristics The mean age of the 26 patients (21 male, 5 female) with albuminuria was 60.3 years, their mean BMI was 25.9 kg/m2 and their mean systolic/diastolic BP was 145.3/ 85.6 mmHg at enrollment (Table 1). Of the 26 patients, 4 mg/day and 8 mg/day of candesartan had been initially prescribed to 15 patients and 11 patients, respectively. Ca channel antagonists were administered in 57.7% (15/26) of the patients, whereas one patient was treated with an -blocker and no patient was treated with a -blocker or diuretic. Approximately 70% (18/26) of patients were treated with a sulfonylurea, 46.2% (12/26) with an - glucosidase inhibitor, 38.5% (10/26) with a biguanide, and 26.9% (7/26) with a thiazolidine against type 2 dia- betes. Insulin treatment was given in 15.4% (4/26) of patients. During this study, the dose of sulfonylurea was increased in one patient, and the dose of thiazolidine was increased in another one. However, there was no change in the formulation of any drugs other than candesartan or that of insulin therapy during the observation period. 3.2. UAE Level Was Improved by Increase in Candesartan to Maximal Dose UAE level was investigated before (at baseline), at 6 months and 12 months after an increase in dose of can- desartan from 4 or 8 mg/day to 12 mg/day. As shown in Table 1 and Figure 1, UAE level slightly decreased be- tween baseline and 6 months, but there was no signifi- cant difference in UAE level between the two time points. Ultimately, UAE level was significantly decreased at 12 months as compared to baseline and 6 months. 3.3. BP Was Significantly Decreased by Increase in Candesartan to Maximal Dose The increase in dose of candesartan to 12 mg/day re- sulted in significant decreases in systolic and diastolic BP both at 6 and 12 months as compared to the baseline systolic and diastolic BP, though there was no significant Copyright © 2013 SciRes. OJEMD 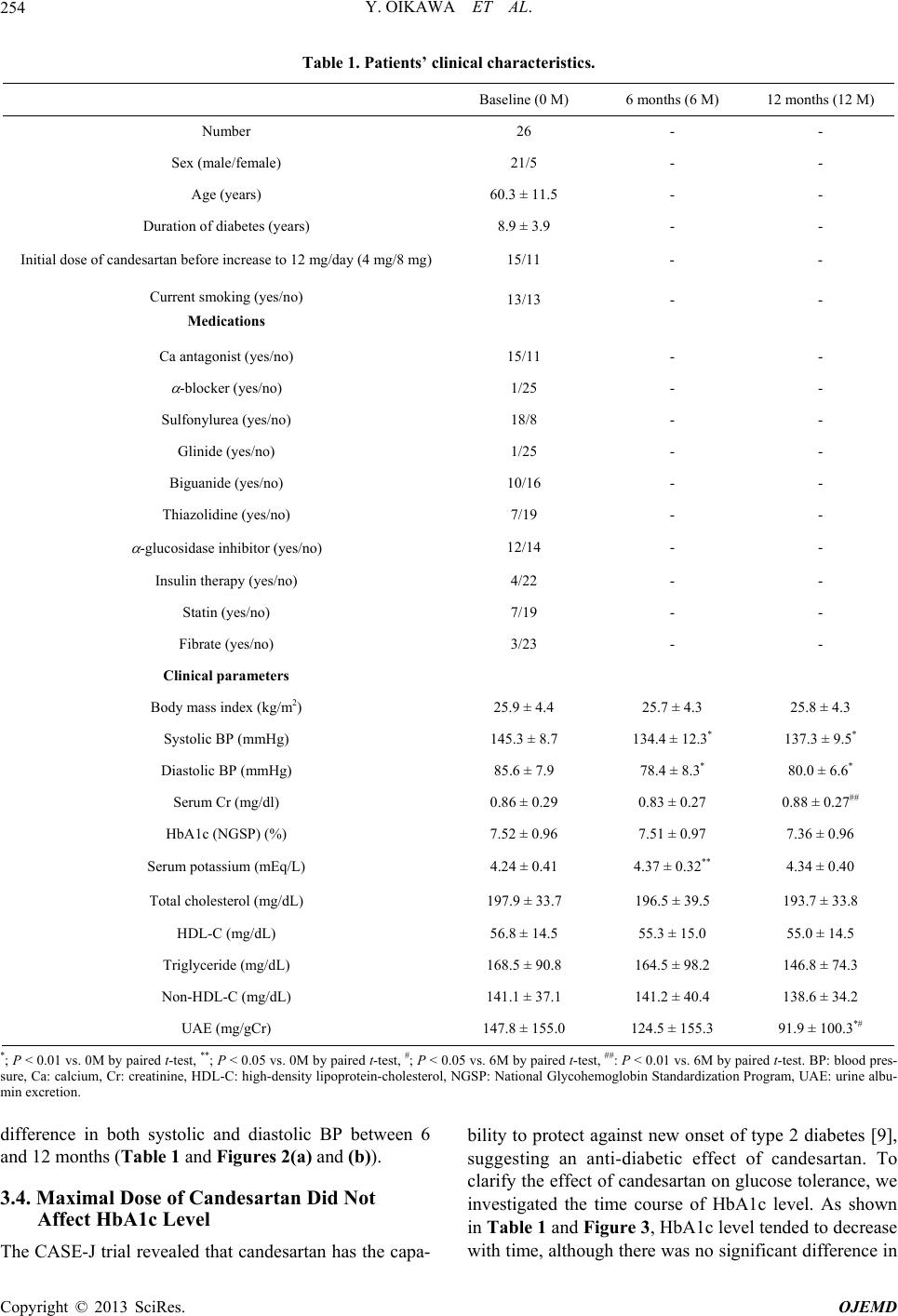 Y. OIKAWA ET AL. Copyright © 2013 SciRes. OJEMD 254 Table 1. Patients’ clinical characteristics. Baseline (0 M) 6 months (6 M) 12 months (12 M) Number 26 - - Sex (male/female) 21/5 - - Age (years) 60.3 ± 11.5 - - Duration of diabetes (years) 8.9 ± 3.9 - - Initial dose of candesartan before increase to 12 mg/day (4 mg/8 mg) 15/11 - - Current smoking (yes/no) Medications 13/13 - - Ca antagonist (yes/no) 15/11 - - -blocker (yes/no) 1/25 - - Sulfonylurea (yes/no) 18/8 - - Glinide (yes/no) 1/25 - - Biguanide (yes/no) 10/16 - - Thiazolidine (yes/no) 7/19 - - -glucosidase inhibitor (yes/no) 12/14 - - Insulin therapy (yes/no) 4/22 - - Statin (yes/no) 7/19 - - Fibrate (yes/no) 3/23 - - Clinical parameters Body mass index (kg/m2) 25.9 ± 4.4 25.7 ± 4.3 25.8 ± 4.3 Systolic BP (mmHg) 145.3 ± 8.7 134.4 ± 12.3* 137.3 ± 9.5* Diastolic BP (mmHg) 85.6 ± 7.9 78.4 ± 8.3* 80.0 ± 6.6* Serum Cr (mg/dl) 0.86 ± 0.29 0.83 ± 0.27 0.88 ± 0.27## HbA1c (NGSP) (%) 7.52 ± 0.96 7.51 ± 0.97 7.36 ± 0.96 Serum potassium (mEq/L) 4.24 ± 0.41 4.37 ± 0.32** 4.34 ± 0.40 Total cholesterol (mg/dL) 197.9 ± 33.7 196.5 ± 39.5 193.7 ± 33.8 HDL-C (mg/dL) 56.8 ± 14.5 55.3 ± 15.0 55.0 ± 14.5 Triglyceride (mg/dL) 168.5 ± 90.8 164.5 ± 98.2 146.8 ± 74.3 Non-HDL-C (mg/dL) 141.1 ± 37.1 141.2 ± 40.4 138.6 ± 34.2 UAE (mg/gCr) 147.8 ± 155.0 124.5 ± 155.3 91.9 ± 100.3*# *; P < 0.01 vs. 0M by paired t-test, **; P < 0.05 vs. 0M by paired t-test, #; P < 0.05 vs. 6M by paired t-test, ##: P < 0.01 vs. 6M by paired t-test. BP: blood pres- sure, Ca: calcium, Cr: creatinine, HDL-C: high-density lipoprotein-cholesterol, NGSP: National Glycohemoglobin Standardization Program, UAE: urine albu- min excretion. difference in both systolic and diastolic BP between 6 and 12 months (Table 1 and Figures 2(a) and (b)). 3.4. Maximal Dose of Candesartan Did Not Affect HbA1c Level The CASE-J trial revealed that candesartan has the capa- bility to protect against new onset of type 2 diabetes [9], suggesting an anti-diabetic effect of candesartan. To clarify the effect of candesartan on glucose tolerance, we investigated the time course of HbA1c level. As shown in Ta ble 1 and Figure 3, HbA1c level tended to decrease with time, although there was no significant difference in 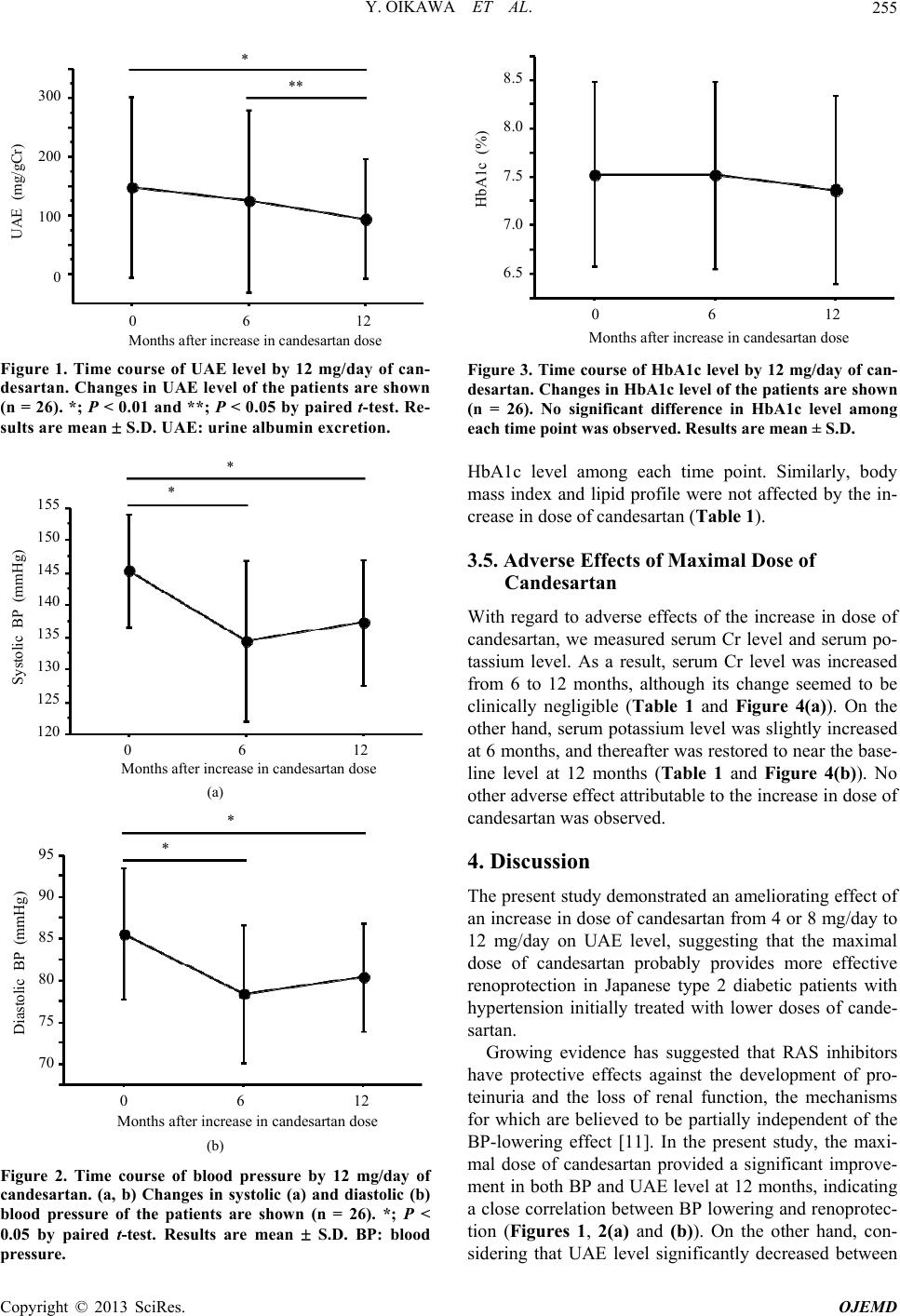 Y. OIKAWA ET AL. 255 300 200 100 0 0 6 12 UAE (mg/gCr) Months after increase in candesartan dose * ** Figure 1. Time course of UAE level by 12 mg/day of can- desartan. Changes in UAE level of the patients are shown (n = 26). *; P < 0.01 and **; P < 0.05 by paired t-test. Re- sults are mean S.D. UAE: urine alb umin excre tion. 0 6 12 Months after increase in candesartan dose * 155 150 145 140 135 130 125 120 Systolic BP (mmHg) * (a) 0 6 12 Months after increase in candesartan dose 95 90 85 80 75 70 Diastolic BP (mmHg) * * (b) Figure 2. Time course of blood pressure by 12 mg/day of candesartan. (a, b) Changes in systolic (a) and diastolic (b) blood pressure of the patients are shown (n = 26). *; P < 0.05 by paired t-test. Results are mean S.D. BP: blood pressure. 0 6 12 Months after increase in candesartan dose 8.5 8.0 7.5 7.0 6.5 HbA1c (%) Figure 3. Time course of HbA1c level by 12 mg/day of can- desartan. Changes in HbA1c level of the patients are shown (n = 26). No significant difference in HbA1c level among each time point was observed. Results are mean ± S.D. HbA1c level among each time point. Similarly, body mass index and lipid profile were not affected by the in- crease in dose of candesartan (Table 1). 3.5. Adverse Effects of Maximal Dose of Candesartan With regard to adverse effects of the increase in dose of candesartan, we measured serum Cr level and serum po- tassium level. As a result, serum Cr level was increased from 6 to 12 months, although its change seemed to be clinically negligible (Table 1 and Figure 4(a)). On the other hand, serum potassium level was slightly increased at 6 months, and thereafter was restored to near the base- line level at 12 months (Table 1 and Figure 4(b)). No other adverse effect attributable to the increase in dose of candesartan was observed. 4. Discussion The present study demonstrated an ameliorating effect of an increase in dose of candesartan from 4 or 8 mg/day to 12 mg/day on UAE level, suggesting that the maximal dose of candesartan probably provides more effective renoprotection in Japanese type 2 diabetic patients with hypertension initially treated with lower doses of cande- sartan. Growing evidence has suggested that RAS inhibitors have protective effects against the development of pro- teinuria and the loss of renal function, the mechanisms for which are believed to be partially independent of the BP-lowering effect [11]. In the present study, the maxi- mal dose of candesartan provided a significant improve- ment in both BP and UAE level at 12 months, indicating a close correlation between BP lowering and renoprotec- tion (Figures 1, 2(a) and (b)). On the other hand, con- sidering that UAE level significantly decreased between Copyright © 2013 SciRes. OJEMD 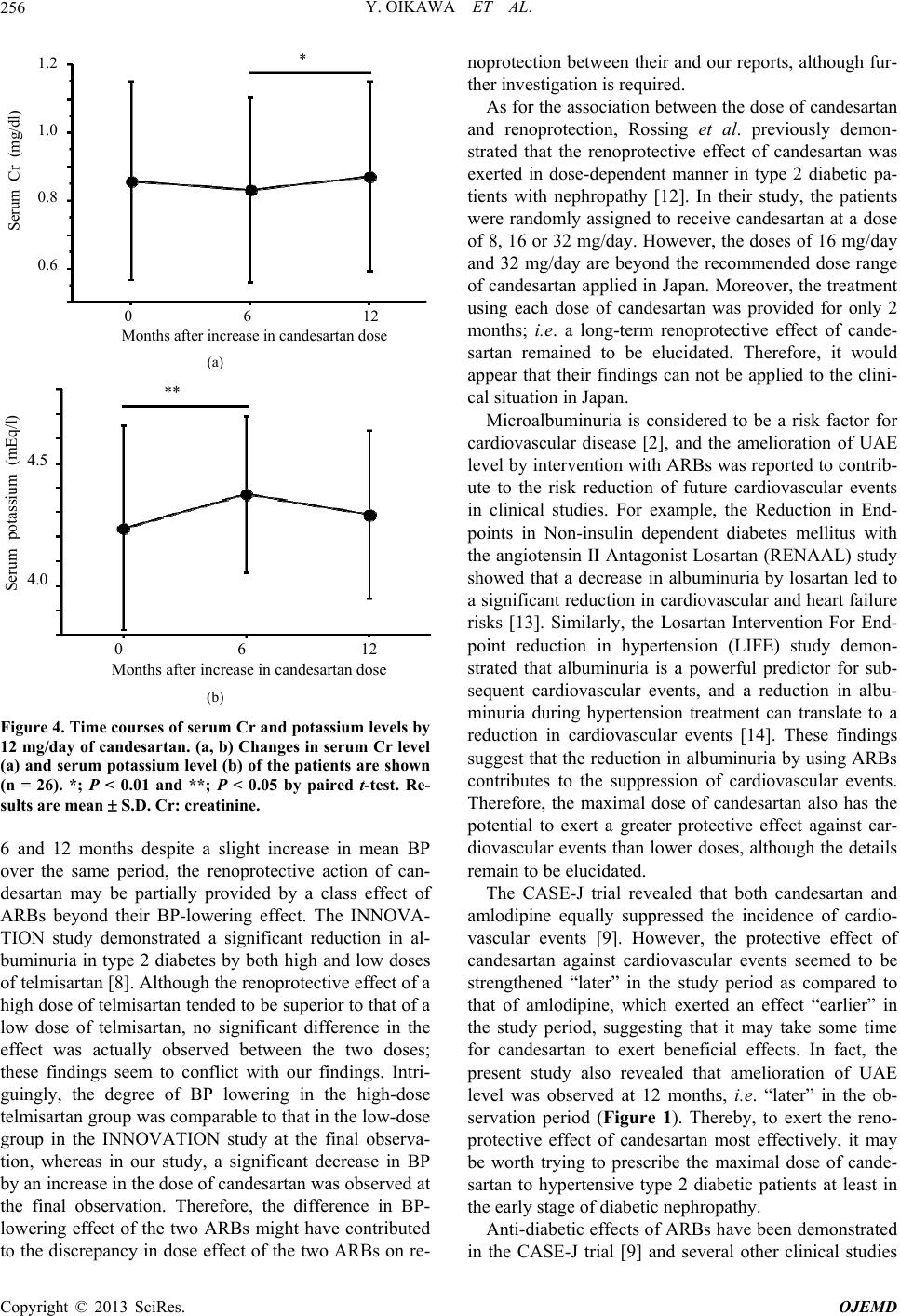 Y. OIKAWA ET AL. 256 1.2 1.0 0.8 0.6 Serum Cr (mg/dl) 0 6 12 Months after increase in candesartan dose * (a) 4.5 4.0 Serum potassium (mEq/l) ** 0 6 12 Months after increase in candesartan dose (b) Figure 4. Time courses of serum Cr and potassium levels by 12 mg/day of candesartan. (a, b) Changes in serum Cr level (a) and serum potassium level (b) of the patients are shown (n = 26). *; P < 0.01 and **; P < 0.05 by paired t-test. Re- sults are mean S.D. Cr: cre a tinine. 6 and 12 months despite a slight increase in mean BP over the same period, the renoprotective action of can- desartan may be partially provided by a class effect of ARBs beyond their BP-lowering effect. The INNOVA- TION study demonstrated a significant reduction in al- buminuria in type 2 diabetes by both high and low doses of telmisartan [8]. Although the renoprotective effect of a high dose of telmisartan tended to be superior to that of a low dose of telmisartan, no significant difference in the effect was actually observed between the two doses; these findings seem to conflict with our findings. Intri- guingly, the degree of BP lowering in the high-dose telmisartan group was comparable to that in the low-dose group in the INNOVATION study at the final observa- tion, whereas in our study, a significant decrease in BP by an increase in the dose of candesartan was observed at the final observation. Therefore, the difference in BP- lowering effect of the two ARBs might have contributed to the discrepancy in dose effect of the two ARBs on re- noprotection between their and our reports, although fur- ther investigation is required. As for the association between the dose of candesartan and renoprotection, Rossing et al. previously demon- strated that the renoprotective effect of candesartan was exerted in dose-dependent manner in type 2 diabetic pa- tients with nephropathy [12]. In their study, the patients were randomly assigned to receive candesartan at a dose of 8, 16 or 32 mg/day. However, the doses of 16 mg/day and 32 mg/day are beyond the recommended dose range of candesartan applied in Japan. Moreover, the treatment using each dose of candesartan was provided for only 2 months; i.e. a long-term renoprotective effect of cande- sartan remained to be elucidated. Therefore, it would appear that their findings can not be applied to the clini- cal situation in Japan. Microalbuminuria is considered to be a risk factor for cardiovascular disease [2], and the amelioration of UAE level by intervention with ARBs was reported to contrib- ute to the risk reduction of future cardiovascular events in clinical studies. For example, the Reduction in End- points in Non-insulin dependent diabetes mellitus with the angiotensin II Antagonist Losartan (RENAAL) study showed that a decrease in albuminuria by losartan led to a significant reduction in cardiovascular and heart failure risks [13]. Similarly, the Losartan Intervention For End- point reduction in hypertension (LIFE) study demon- strated that albuminuria is a powerful predictor for sub- sequent cardiovascular events, and a reduction in albu- minuria during hypertension treatment can translate to a reduction in cardiovascular events [14]. These findings suggest that the reduction in albuminuria by using ARBs contributes to the suppression of cardiovascular events. Therefore, the maximal dose of candesartan also has the potential to exert a greater protective effect against car- diovascular events than lower doses, although the details remain to be elucidated. The CASE-J trial revealed that both candesartan and amlodipine equally suppressed the incidence of cardio- vascular events [9]. However, the protective effect of candesartan against cardiovascular events seemed to be strengthened “later” in the study period as compared to that of amlodipine, which exerted an effect “earlier” in the study period, suggesting that it may take some time for candesartan to exert beneficial effects. In fact, the present study also revealed that amelioration of UAE level was observed at 12 months, i.e. “later” in the ob- servation period (Figure 1). Thereby, to exert the reno- protective effect of candesartan most effectively, it may be worth trying to prescribe the maximal dose of cande- sartan to hypertensive type 2 diabetic patients at least in the early stage of diabetic nephropathy. Anti-diabetic effects of ARBs have been demonstrated in the CASE-J trial [9] and several other clinical studies Copyright © 2013 SciRes. OJEMD  Y. OIKAWA ET AL. 257 using losartan [15], irbesartan [16] and valsartan [17], thus far. In general, angiotensin II, a potent vasocon- strictor, has the capability to impair insulin action and induce glucose intolerance [18,19]. Therefore, inhibition of angiotensin II action is currently believed to contribute to an improvement in insulin sensitivity or -cell respon- siveness to glucose. Although the present study failed to reveal a lowering effect on HbA1c level of the maximal dose of candesartan, the mean HbA1c level tended to slightly decrease between baseline and 12 months (Fig- ure 3). It would appear that the obscure anti-diabetic effect may be attributable to the small sample size; a lar- ger scale study would thereby help to demonstrate the beneficial effect of candesartan on glucose tolerance. Limitations of our data include not only the small sample size and gender imbalance, but also the observa- tional nature of the case series study; i.e. a control group continuing initial lower doses of candesartan was not set for comparison. In addition, albuminuria was character- ized by determination of UAE level in a single spot-urine collection at baseline and each time point of follow-up; however, a previous study demonstrated a close correla- tion between spot-urine UAE level and measurements of albuminuria from overnight or 24-hour urine collection [20]. Considering the variability in determination of UAE level, only one measurement at each time point would weaken the relationship between UAE level and outcome measures. To solve this problem, a larger number sample size would be required. It should be noted that our patients had been initially treated with lower doses (4 or 8 mg/day) of candesartan without any adverse effects before the increase in dose, possibly contributing to the minimal change in serum Cr level and serum potassium level without any severe ad- verse effects in the present study. Thus, we would like to emphasize that the maximal dose of candesartan should be prescribed to diabetic patients after evaluating the to- lerability of candesartan beforehand by using lower doses of candesartan. 5. Conclusion In conclusion, an increase in amount of candesartan from a lower dose to the maximal dose exerted an ameliorating effect on albuminuria as well as BP in hypertensive type 2 diabetic patients in the early stage of diabetic neph- ropathy. Using the maximal dose of candesartan at the earliest possible time may help to prevent not only loss of renal function, but also future cardiovascular events in hypertensive type 2 diabetic patients at high cardiovas- cular risk. 6. Acknowledgements This research received no specific grant from any fund- ing agency in the public, commercial, or not-for-profit sectors. REFERENCES [1] W. B. Kannel and D. L. McGee, “Diabetes and Glucose Tolerance as Risk Factors for Cardiovascular Disease: the Framingham Study,” Diabetes Care, Vol. 2, No. 2, 1979, pp. 120-126. doi:10.2337/diacare.2.2.120 [2] H. C. Gerstein, J. F. Mann, Q, Yi, B. Zinman, S. F. Din- neen, B. Hoogwerf, J. P. Hallé, J. Young, A. Rashkow, C. Joyce, S. Nawaz and S. Yusuf; HOPE Study Investigators, “Albuminuria and Risk of Cardiovascular Events, Death, and Heart Failure in Diabetic and Nondiabetic Individu- als,” JAMA, Vol. 286, No. 4, 2001, pp. 421-426. doi:10.1001/jama.286.4.421 [3] M. Hayashi, “Development of New Therapies, Including Regeneration of the Kidney, for Chronic Kidney Disea- ses,” Clinical and Experimental Nephrology, Vol. 10, No. 2, 2006, pp. 99-101. doi:10.1007/s10157-006-0418-7 [4] Japanese Society of Hypertension Committee, “The Japa- nese Society of Hypertension Guidelines for the Manage- ment of Hypertension (JSH 2009),” Hypertension Resear- ch, Vol. 32, No. 1, 2009, pp. 3-107. [5] V. Dzau, “The Cardiovascular Continuum and Renin-An- giotensin-Aldosterone System Blockade,” Journal of Hy- pertension—Supplement, Vol. 23, No. 1, 2005, pp. S9- S17. doi:10.1097/01.hjh.0000165623.72310.dd [6] E. J. Lewis, L. G. Hunsicker, W. R. Clarke, T. Berl, M. A. Pohl, J. B. Lewis, E. Ritz, R. C. Atkins, R. Rohde and I. Raz; Collaborative Study Group, “Renoprotective Effect of the Angiotensin-Receptor Antagonist Irbesartan in Pa- tients with Nephropathy Due to Type 2 Diabetes,” The New England Journal of Medicine, Vol. 345, No. 12, 2001, pp. 851-860. doi:10.1056/NEJMoa011303 [7] M. A. Pfeffer, K. Swedberg, C. B. Granger, P. Held, J. J. McMurray, E. L. Michelson, B. Olofsson, J. Ostergren, S and Yusuf and S. Pocock; CHARM Investigators and Committees, “Effects of Candesartan on Mortality and Morbidity in Patients with Chronic Heart Failure: the CHARM-Overall Programme,” Lancet, Vol. 362, No. 9386, 2003, pp. 759-766. doi:10.1016/S0140-6736(03)14282-1 [8] H. Makino, M. Haneda, T. Babazono, T. Moriya, S. Ito, Y. Iwamoto, R. Kawamori, M. Takeuchi and S. Katayama; INNOVATION Study Group, “Prevention of Transition from Incipient to Overt Nephropathy with Telmisartan in Patients with Type 2 Diabetes,” Diabetes Care, Vol. 30, No. 6, 2007, pp. 1577-1578. doi:10.2337/dc06-1998 [9] T. Ogihara, K. Nakao, T. Fukui, K. Fukiyama, K. Ueshi- ma, K. Oba, T. Sato and T. Saruta; Candesartan Anti-hy- pertensive Survival Evaluation in Japan Trial Group, “Ef- fects of Candesartan Compared with Amlodipine in Hy- pertensive Patients with High Cardiovascular Risks: Can- desartan Antihypertensive Survival Evaluation in Japan Trial,” Hypertension, Vol. 51, No. 2, 2008, pp. 393-398. doi:10.1161/HYPERTENSIONAHA.107.098475 [10] T. Saruta, K. Hayashi, T. Ogihara, K. Nakao, T. Fukui and K. Fukiyama; CASE-J Study Group, “Effects of Can- desartan and Amlodipine on Cardiovascular Events in Copyright © 2013 SciRes. OJEMD  Y. OIKAWA ET AL. Copyright © 2013 SciRes. OJEMD 258 Hypertensive Patients with Chronic Kidney Disease: Sub- analysis of the CASE-J Study,” Hypertension Research, Vol. 32, No. 6, 2009, pp. 505-512. doi:/10.1038/hr.2009.44 [11] F. Locatelli, L. Del Vecchio and A. Cavalli, “Inhibition of the Renin-Angiotensin System in Chronic Kidney Disea- se: A Critical Look to Single and Dual Blockade,” Ne- phron Clinical Practice, Vol. 113, No. 4, 2009, pp. c286- c293. doi:10.1159/000235946 [12] K. Rossing, P. K. Christensen, B. V. Hansen, B. Carsten- sen and H. H. Parving, “Optimal Dose of Candesartan for Renoprotection in Type 2 Diabetic Patients with Nephro- pathy: A Double-Blind Randomized Cross-Over Study,” Diabetes Care, Vol. 26, No. 1, 2003, pp. 150-155. doi:10.2337/diacare.26.1.150 [13] D. de Zeeuw, G. Remuzzi, H.H. Parving, W. F. Keane, Z. Zhang, S. Shahinfar, S. Snapinn, M. E. Cooper, W. E. Mitch and B. M. Brenner, “Albuminuria, a Therapeutic Target for Cardiovascular Protection in Type 2 Diabetic Patients with Nephropathy,” Circulation, Vol. 110, No. 8, 2004, pp. 921-927. doi:10.1161/01.CIR.0000139860.33974.28 [14] H. Ibsen, M. H. Olsen, K. Wachtell, K. Borch-Johnsen, L. H. Lindholm, C. E. Mogensen, B. Dahlöf, R. B. Devereux, U. de Faire, F. Fyhrquist, S. Julius, S. E. Kjeldsen, O. Lederballe-Pedersen, M. S. Nieminen, P. Omvik, S. Opa- ril and Y. Wan, “Reduction in Albuminuria Translates to Reduction in Cardiovascular Events in Hypertensive Pa- tients: Losartan Intervention for Endpoint Reduction in Hypertension Study,” Hypertension, Vol. 45, No. 2, 2005, pp. 198-202. doi:10.1161/01.HYP.0000154082.72286.2a [15] J. V. Lozano, J. L. Llisterri, J. Aznar and J. Redon; Spani- sh Working Group, “Losartan Reduces Microalbuminuria in Hypertensive Microalbuminuric Type 2 Diabetics,” Nephrology Dialysis Transplantation, Vol. 16, Suppl. 1, 2001, pp. 85-89. doi:10.1093/ndt/16.suppl_1.85 [16] P. Bramlage, D. Pittrow and W. Kirch, “The Effect of Irbesartan in Reducing Cardiovascular Risk in Hyperten- sive Type 2 Diabetic Patients: An Observational Study in 16,600 Patients in Primary Care,” Current Medical Re- search and Opinion, Vol. 20, No. 10, 2004, pp. 1625- 1631. doi:10.1185/030079904X3861 [17] S. E. Kjeldsen, S. Julius, G. Mancia, G. T. McInnes, T. Hua, M. A. Weber, A. Coca, S. Ekman, X. Girerd, K. Ja- merson, P. Larochelle, T. M. MacDonald, R. E. Sch- mieder, M. A. Schork, P. Stolt, R. Viskoper, J. Widimský and A. Zanchetti; VALUE Trial Investigators, “Effects of Valsartan Compared to Amlodipine on Preventing Type 2 Diabetes in High-Risk Hypertensive Patients: The VALUE Trial,” Journal of Hypertension, Vol. 24, No. 7, 2006, pp. 1405-1412. doi:10.1097/01.hjh.0000234122.55895.5b [18] R. H. Rao, “Pressor Doses of Angiotensin II Increase Hepatic Glucose Output and Decrease Insulin Sensitivity in Rats,” Journal of Endocrinology, Vol. 148, No. 2, 1996, pp. 311-318. doi:10.1677/joe.0.1480311 [19] T. Ogihara, T. Asano, K. Ando, Y. Chiba, H. Sakoda, M. Anai, N. Shojima, H. Ono, Y. Onishi, M. Fujishiro, H. Katagiri, Y. Fukushima, M. Kikuchi, N. Noguchi, H. Ab- uratani, I. Komuro and Y. Fujita, “Angiotensin II-In- duced Insulin Resistance Is Associated with Enhanced Insulin Signaling,” Hypertension, Vol. 40, No. 6, 2002, pp. 872-879. doi:10.1161/01.HYP.0000040262.48405.A8 [20] O. Eshøj, B. Feldt-Rasmussen, M. L. Larsen and E. F. Mogensen, “Comparison of Overnight, Morning and 24- Hour Urine Collections in the Assessment of Diabetic Mi- croalbuminuria,” Diabetic Medicine, Vol. 4, No. 6, 1987, pp. 531-533. doi:10.1111/j.1464-5491.1987.tb00924.x
|