Paper Menu >>
Journal Menu >>
 Advances in Materials Physics and Chemistry, 2012, 2, 229-232 doi:10.4236/ampc.2012.24B058 Published Online December 2012 (http://www.SciRP.org/journal/ampc) In Vitro Study of Carbamate-Linked Cationic Lipid for Gene Delivery Against Cervical Cancer Cells Defu Zhi1, Shuibao Zhang1, Yinan Zhao1, Shaohui Cui1, Bing Wang1, Huiying Chen1, Defu Zhi2, Defeng Zhao2 1SEAC-ME Key Laboratory of Biochemistry Engineering, Dalian Nationalities University, Dalian, China 2State Key Laboratory of Fine Chemicals, Dalian University of Technology, Dalian, China Email: zsb@dlnu.edu.cn, zhaodfg@chem.dlut.edu.cn Received 2012 ABSTRACT Design and synthesis of a carbamate-linked cationic lipid DDCTMA (N-[1-(2,3-didodecylcarbamoyloxy)propyl]-N,N,N-trimethyl- ammonium iodide) as gene delivery carriers was described in this work. The transfection efficiency of cationic liposome increased dramatically with the increase in the content of DOPE. In addition, the transfection efficiency of some of cationic lipoplexes was superior or parallel to that of two commercial transfection agents, Lipofectamine2000 and DOTAP. The carbamate-linked cationic lipid DDCTMA/DOPE may be a promising gene carrier that has high transfection efficiency as well as low cytotoxicity. Keywords: Cationic Lipids; DNA Condensation; Gene Delivery; Transfection Efficiency 1. Introduction As an important non-viral gene vector, cationic lipids have attracted increasing attention because they have many potential advantages compared with other non-viral vectors, including significant simplicity and ease of production, good repeatability, potential commercial value, and wide range of clinical applica- tion and safety [1]. Felgner et al. [2] have demonstrated that cationic lipid—DOTMA (N-[1-(2, 3-dioleoyloxy)propyl]-N,N, N-trimethylammonium chloride) was an effective gene vector in the study of complex transfection, since then numerous cati- onic lipids with different structures have been synthesized and used for the delivery of nucleic acids into cells during the last 25 years [3]. Cationic lipids have three basic chemical functional domains: a hydrophilic headgroup, a hydrophobic domain, and a linker bond that tethers the cationic headgroup and hydrophobic tail domain [4]. Linker bonds of cationic lipids have large influence on the transfection efficiency, biodegradability and the stability of cationic lipids. Most of the linker bonds in some synthesized lipids are ether, ester, or amide bonds, which are either too stable to be biodegraded and thus cause cytotoxicity (e.g., ether linkers) or prone to decompose during systemic circulation (e.g., ester linkers). In contrast with these strategies, in a previous paper [5] we developed two carbamate-linked lipids (DDC- TMA and DDCEDMA) bearing quaternary ammonium head- group and identical length of hydrocarbon chains for the pur- pose of taking advantage of the pH sensitivity of the carbamate bond (Scheme 1), which has high transfection efficiency in Hela and Hep-2 cells as well as low cytotoxicity. To advance the study of this lipid for gene delivery we investigated the influence of the N/P ratios on the characteristics of the DDCTMA/DOPE/DNA complex compared with two commer- cial transfection agents, Lipofectamine2000 and DOTAP. 2. Materials and Methods 2.1. Instruments and Reagents Most chemicals were obtained from Sinopharm Holding Co. Ltd. (Shanghai, China). 3-Chloro-1,2-propanediol was pur- chased from Johnson Matthey (Hong Kong, China). N,N-car- bonyldiimidazole (CDI) was purchased from Medpep Co. Ltd. (Shanghai, China). Cell culture media and fetal bovine sera (FBS) were purchased from Invitrogen Corporation (Carlsbad, CA, USA). Dulbecco’s modified Eagle’s medium (DMEM) was purchased from Sigma Co. Ltd. (USA). Lipofectamine 2000 reagent was purchased from Invitrogen Corporation (Shanghai). DOTAP reagent was purchased from Roche Diagnostics GmbH. Scheme 1. Synthetic routes of cationic lipid. Reagents and condi- tions: (a) 2.5 equiv. 33% dimethylamine in aqueous solution, 1.0 equiv. sodium hydroxide, 4 h at 50 ºC, (70%); (b) 2.1 equiv. CDI in toluene solution, 3 h at 40 ºC with N2; (c) 2.1 equiv. alkylamine in toluene solution, 3 h at 40 ºC with N2, (50-60%); (d) 40 equiv. halo- genated hydrocarbons, 24 h, 80 ºC (95%). Copyright © 2012 SciRes. AMPC  D. F. ZHI ET AL. 230 2.2. Characterization FTIR spectral studies were carried out with a Thermo Nicolet 370 DTGS (USA) spectrometer in the range between 4000 and 500 cm-1. All powder samples were compressed into KBr pel- lets for the FTIR measurements. ESI-MS was detected by SHIMADZU LCMS-2010EV (Japan) in methanol (MeOH) depending on sample solubility at room temperature. Electros- pray ionization was achieved by application of a potential of 3.5 kV to a stainless needle. A Harvard apparatus syringe pump system was set at 3.0 mL/min. Nitrogen as a nebulizer gas was delivered to the spectrometer by a nitrogen line. HPLC-ELSD was detected by SHIMADZU LCMS-2010EV (Japan) and SFD-ZAM3000 (Schambeck SFD GmbH, Germany). Nuclear magnetic resonance (NMR) spectrum was recorded on a Varian Mercury plus 400-MHz NMR (USA) spectrometer in chloro- form (CDCl3) depending on sample solubility at room tem- perature. 2.3. Synthesis and Characterization of Cationic Lipid (DDCTMA) Synthesis and characterization of cationic lipid DDCTMA were as reported earlier [5]. The synthesis of cationic lipid DDCTMA is shown in Scheme 1. 2.4. Preparation of Liposomes and Plasmid DNA A solution of cationic lipid (1mg) in chloroform (1mL) was evaporated under a stream of nitrogen, and the residual solvent was removed under vacuum overnight. Liposomes were pre- pared by resuspending the lipid in distilled water (1mL) at 55°C and sonicating them to clarity at this temperature for 2 h in a closed vial. Plasmid pGL3 coding for luciferase gene was purchased from Clontech (USA). Plasmid DNA was isolated using a BBI DNA purification kit. The DNA concentration was determined by measuring UV absorbance at 260nm and 280nm, and the purity was confirmed by agarose gel electrophoresis and OD260/280 measurement. 2.5. DNA Binding Assay DNA-lipid complexes were formed by mixing 2 μg of plasmid DNA (0.1 μg/μL in 10mM Hepes buffer, pH 7.4) with varying amounts of cationic lipid so that the final lipid/DNA charge ratios were maintained at 0.5/1 to 8/1 in a total volume of 50 μL. Complexes were incubated for 30 min at room temperature after which 20 μL of each lipoplex was loaded on a 1.2% aga- rose gel and subjected to electrophoresis. The samples were electrophoresed at 90 V for 1h, and the bands were visualized with ethidium bromide staining. 2.6. Transfection Assay Human cervical cancer cells (7721) were obtained from ATCC (American Type Culture Collection Shanghai Representative Office) and seeded in 100μl of growth medium (RPMI1640) without antibiotics. The cells were incubated at 37 ºC in a fully humidified atmosphere containing 5% CO2, up to 80% conflu- ence prior to use. To measure transfection efficiency, liposomes and 0.5μg plasmid DNA were diluted in 25μl DMEM without serum, respectively, and mixed gently. Five minutes after dilution, the diluted liposomes were added to the diluted DNA and mixed together with vortex. The mixture was held for 20 min at room temperature to enable the lipoplex formation. The original cell culture media were replaced with the lipoplex solution contain- ing the transfection lipoplexes (prepared as described above) and phosphate buffered saline (PBS) for each well. They were incubated at 37ºC in a humidified incubator with 5% CO2 for 4-6 h, then cells were washed by PBS or DMEM once and the medium was exchanged with fresh and complete DMEM cul- ture media and cells were further cultured for 48 h, prior to analysis. Finally, the transfection efficiency of some of the cationic liposomes, as % relative light units (RLU) was meas- ured by luciferase assay. 3. Results and Discussion We have described the synthesis of lipid DDCTMA and re- ported some of its physicochemical characteristics and transfec- tion efficiencies in vitro and cytotoxicity using Hela and Hep-2 cell lines [5]. Herein, we not only investigated its DNA-binding ability and transfection efficiencies using 7721 cell lines but also researched on the influence of the N/P ratios on the char- acteristics of the DDCTMA/DOPE/DNA complex compared with two commercial transfection agents, Lipofectamine2000 and DOTAP. 3.1. Properties of cationic liposomes In order to determine the effect of DOPE and cationic lipo- somes to pGFP-N2 plasmid charge ratios (N/P ratios), pGL3 plasmid complexes were prepared by adjusting the stoichiome- try of cationic liposomes and plasmid (N/P, 0.5/1, 1/1, 1.5/1, 2/1, 3/1, 4/1, 6/1and 8/1), using liposomes prepared from cati- onic lipid (DDCTMA) and a co-lipid (DOPE) at different molar ratios (N/D, 2/1, 1/1 and 1/2). As shown in Figure 1, the DNA-binding ability of cationic liposome increased with an increase in the N/P ratio, indicating that these liposomes have an ability to form a lipoplex with plasmid DNA. When the N/P ratio of DDCTMA/DOPE (2/1) liposome/DNA was greater than 2, liposome and DNA bound tightly and completely. When N/P was equal to 4, the band of DDCTMA/DOPE (1/1) liposome/DNA was fainter than the N/P ratios of 6/1 and above. However, DDCTMA/DOPE (1/2) liposome was found to be the weakest in DNA binding, as the band of plasmid DNA disappeared at the N/P ratio of 8/1. Ob- viously, the DNA-binding ability of cationic lipid decreased with an increase in the content of DOPE. It is postulated that the increase in the content of DOPE induces significant changes in morphologies and structural parameters of the lipoplexes (such as, particle size and ζ-potential as showed in Table 1) and hence influences their DNA-binding ability [6]. 3.2. In Vitro Transfection In constructing gene complex vectors, the molar ratio of cati- onic lipid to co-lipid may play an important role in influencing the phase transition temperature of cationic liposomes and the Copyright © 2012 SciRes. AMPC 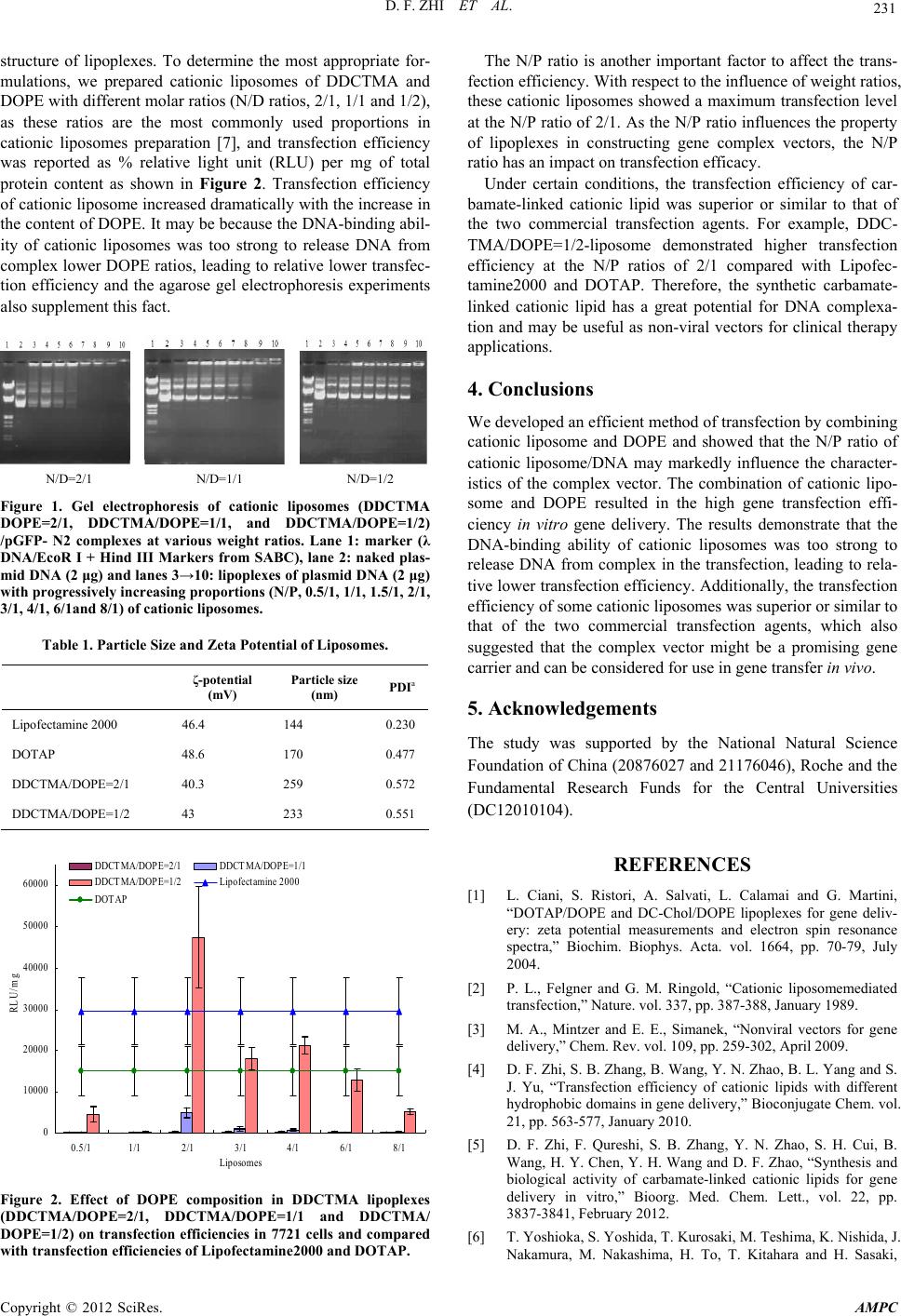 D. F. ZHI ET AL. 231 structure of lipoplexes. To determine the most appropriate for- mulations, we prepared cationic liposomes of DDCTMA and DOPE with different molar ratios (N/D ratios, 2/1, 1/1 and 1/2), as these ratios are the most commonly used proportions in cationic liposomes preparation [7], and transfection efficiency was reported as % relative light unit (RLU) per mg of total protein content as shown in Figure 2. Transfection efficiency of cationic liposome increased dramatically with the increase in the content of DOPE. It may be because the DNA-binding abil- ity of cationic liposomes was too strong to release DNA from complex lower DOPE ratios, leading to relative lower transfec- tion efficiency and the agarose gel electrophoresis experiments also supplement this fact. N/D=2/1 N/D=1/1 N/D=1/2 Figure 1. Gel electrophoresis of cationic liposomes (DDCTMA DOPE=2/1, DDCTMA/DOPE=1/1, and DDCTMA/DOPE=1/2) /pGFP- N2 complexes at various weight ratios. Lane 1: marker (λ DNA/EcoR I + Hind III Markers from SABC), lane 2: naked plas- mid DNA (2 μg) and lanes 3→10: lipoplexes of plasmid DNA (2 μg) with progressively increasing proportions (N/P, 0.5/1, 1/1, 1.5/1, 2/1, 3/1, 4/1, 6/1and 8/1) of cationic liposomes. Table 1. Particle Size and Zeta Potential of Liposomes. ζ-potential (mV) Particle size (nm) PDIa Lipofectamine 2000 46.4 144 0.230 DOTAP 48.6 170 0.477 DDCTMA/DOPE=2/1 40.3 259 0.572 DDCTMA/DOPE=1/2 43 233 0.551 0 10000 20000 30000 40000 50000 60000 0.5/11/1 2/1 3/14/1 6/1 8/1 Liposomes RLU/m g DDCTMA/DOPE=2/1 DDCT MA/DOPE=1/1 DDCTMA/DOPE=1/2 Lipofectamine 2000 DOTAP Figure 2. Effect of DOPE composition in DDCTMA lipoplexes (DDCTMA/DOPE=2/1, DDCTMA/DOPE=1/1 and DDCTMA/ DOPE=1/2) on transfection efficiencies in 7721 cells and compared with transfection efficiencies of Lipofectamine2000 and DOTAP. The N/P ratio is another important factor to affect the trans- fection efficiency. With respect to the influence of weight ratios, these cationic liposomes showed a maximum transfection level at the N/P ratio of 2/1. As the N/P ratio influences the property of lipoplexes in constructing gene complex vectors, the N/P ratio has an impact on transfection efficacy. Under certain conditions, the transfection efficiency of car- bamate-linked cationic lipid was superior or similar to that of the two commercial transfection agents. For example, DDC- TMA/DOPE=1/2-liposome demonstrated higher transfection efficiency at the N/P ratios of 2/1 compared with Lipofec- tamine2000 and DOTAP. Therefore, the synthetic carbamate- linked cationic lipid has a great potential for DNA complexa- tion and may be useful as non-viral vectors for clinical therapy applications. 4. Conclusions We developed an efficient method of transfection by combining cationic liposome and DOPE and showed that the N/P ratio of cationic liposome/DNA may markedly influence the character- istics of the complex vector. The combination of cationic lipo- some and DOPE resulted in the high gene transfection effi- ciency in vitro gene delivery. The results demonstrate that the DNA-binding ability of cationic liposomes was too strong to release DNA from complex in the transfection, leading to rela- tive lower transfection efficiency. Additionally, the transfection efficiency of some cationic liposomes was superior or similar to that of the two commercial transfection agents, which also suggested that the complex vector might be a promising gene carrier and can be considered for use in gene transfer in vivo. 5. Acknowledgements The study was supported by the National Natural Science Foundation of China (20876027 and 21176046), Roche and the Fundamental Research Funds for the Central Universities (DC12010104). REFERENCES [1] L. Ciani, S. Ristori, A. Salvati, L. Calamai and G. Martini, “DOTAP/DOPE and DC-Chol/DOPE lipoplexes for gene deliv- ery: zeta potential measurements and electron spin resonance spectra,” Biochim. Biophys. Acta. vol. 1664, pp. 70-79, July 2004. [2] P. L., Felgner and G. M. Ringold, “Cationic liposomemediated transfection,” Nature. vol. 337, pp. 387-388, January 1989. [3] M. A., Mintzer and E. E., Simanek, “Nonviral vectors for gene delivery,” Chem. Rev. vol. 109, pp. 259-302, April 2009. [4] D. F. Zhi, S. B. Zhang, B. Wang, Y. N. Zhao, B. L. Yang and S. J. Yu, “Transfection efficiency of cationic lipids with different hydrophobic domains in gene delivery,” Bioconjugate Chem. vol. 21, pp. 563-577, January 2010. [5] D. F. Zhi, F. Qureshi, S. B. Zhang, Y. N. Zhao, S. H. Cui, B. Wang, H. Y. Chen, Y. H. Wang and D. F. Zhao, “Synthesis and biological activity of carbamate-linked cationic lipids for gene delivery in vitro,” Bioorg. Med. Chem. Lett., vol. 22, pp. 3837-3841, February 2012. [6] T. Yoshioka, S. Yoshida, T. Kurosaki, M. Teshima, K. Nishida, J. Nakamura, M. Nakashima, H. To, T. Kitahara and H. Sasaki, Copyright © 2012 SciRes. AMPC  D. F. ZHI ET AL. Copyright © 2012 SciRes. AMPC 232 “Cationic liposomes-mediated plasmid DNA delivery in murine hepatitis induced by carbon tetrachloride,” J. Liposome Res. vol. 19, pp. 141-147, June 2009. [7] S., Fletcher, A., Ahmad, E., Perouzel, M. R., Jorgensen and A. D. Miller, “A dialkanoyl analogue of DOPE improves gene transfer of lower-charged, cationic lipoplexes.” Org. Biomol. Chem., vol. 4, pp. 196-199, December 2006. |

