Natural Science
Vol.5 No.7A(2013), Article ID:34187,8 pages DOI:10.4236/ns.2013.57A002
Single sperm selection and DNA fragmentation analysis: The case of MSOME/IMSI
![]()
1Department of Biology, Genetics Unit, University Autónoma of Madrid, Madrid, Spain; *Corresponding Author: jaime.gosalvez@uam.es
2Ginemed Clinic, República Argentina Av, Sevilla, Spain
Copyright © 2013 Jaime Gosálvez et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 28 May 2013; revised 28 June 2013; accepted 5 July 2013
Keywords: MSOME/IMSI; Assisted reproductive technology; Fertility; Sperm DNA fragmentation; Male factor; Semen processing
ABSTRACT
Sperm of poor quality may affect syngamy after fertilization, embryo development up to the blastocyst stage and reproductive outcome. Subsequently, sperm selection based on morphological characteristics and sperm DNA quality may help to partially avoid these problems. Today, highly efficient sperm selection based on morphological characteristics can be attained using the motile sperm organelle morphology (MSOME) examination, and the spermatozoa selected can be used for ICSI through a fertilization strategy known as intra-cytoplasmic morphologically selected sperm injection (IMSI). The aim of this investigation was to develop a simple methodology to assess sperm DNA fragmentation in single spermatozoa following MSOME/ IMSI, to test the hypothesis that morphologically normal spermatozoa, with an absence of vacuolization, is free of DNA damage. The results indicated that MSOME/IMSI-selected sperm, combined with the Sperm Chromatin Dispersion test (SCD; Oligo-Halosperm), can be reliably used to assess sperm DNA damage in selected single spermatozoa (75% average efficiency), thereby establishing a direct relationship between a good morphological pattern on the sperm and a good DNA quality. Furthermore, results showed spermatozoa presenting a normal morphology and no traces of vacuolization to be fully free of DNA damage. However, traces of vacuolization and more severe morphological alterations were accompanied by significant increases in the proportion of sperm containing a damaged DNA molecule. Interestingly, subtle morphological differences observed between normal and non-vacuolated and normal but vacuolated sperm exhibited significant differrences in the ability of the SCD-Oligo-Halosperm treated sperm to expand DNA fibers following protein depletion.
1. INTRODUCTION
Correlations between reproductive outcome and sperm morphology and/or other semen characteristics such as motility, concentration, membrane stability, mitochondrial activity or DNA fragmentation have been a recurring topic in reproductive research [1-4]. Among the sperm characteristics can be assessed; sperm morphology has traditionally played a crucial role in the diagnosis of male fertility potential [4-8]. Similarly, having received greater interest from researchers in recent years, the assessment of sperm DNA quality is another parameter recommended for the evaluation of ejaculate quality in patients with compromised fertility [9,10].
Recently, new techniques based principally on light microscopy and image manipulation in selected spermatozoa, have allowed for the visualization of living sperm with greater detail than that achieved through standard phase contrast microscopy. The interest in these techniques lies in their ability to achieve extremely high magnification (ca. 6600x) of a specific spermatozoon, allowing for real-time assessment of its morphological characteristics. As a result, embryologists may analyze the sperm in greater detail and, subsequently, target particular spermatozoa free of morphological anomalies for intracytoplasmic sperm injection (ICSI). In the motile sperm organelle morphology examination (MSOME) technique originally performed for the efficient identifycation of motile spermatozoa [11-14], MSOME’s ability to detect subtle morphological features such as midpiece-related abnormalities, traces of cytoplasmic droplets, abnormally-sized heads and vacuole presence in the sperm head has drawn the attention of researchers [13,15]. Known also as intracytoplasmic morphologically selected sperm injection (IMSI), this technique allows embryologists to observe the sperm in greater detail, select the targeted sperm and, using the same technique, inject the selected sperm into the oocyte. Some studies suggest that the use of this technique not only results in the selection of higher-quality sperm, but also in higher rates of pregnancy and lower rates of miscarriage when compared to conventional ICSI (in which confidence levels during the selection of the optimum sperm morphology are supposedly compromised by the lower resolution under the microscope). In Bartoov, et al. [11,16], fertilization using IMSI was reported to improve pregnancy rates in couples with repeated ICSI failures. After comparing 50 couples in two different groups, one undergoing IMSI and the other ICSI, the observed pregnancy rate in IMSI couples was found to be nearly double that of the conventional ICSI couples. Furthermore, significant improvement in implantation, pregnancy and birth rates has also been reported in IMSI in patients having made two or more previously unsuccessful ICSI attempts [17].
If these findings are to be believed, it is highly likely that the selection of good sperm morphology is linked to high-quality sperm DNA. However, the direct correlation between an essential sperm characteristic, such as DNA, and sperm morphology has yet to be studied in depth. This is mainly due to the fact that the selection of individual spermatozoa is a limiting factor in the establishment of a direct correlation between both parameters because of technical problems for a simultaneous visualization. However, there are indirect indications of an association between spermatozoa with high levels of morphological abnormalities, particularly vacuolization, and high levels of DNA damage [18,19].
This study was conducted with the aim of designing a simple system allowing for the direct assessment of sperm DNA fragmentation (SDF) from a single spermatozoa selected using MSOME/IMSI. The possibility of developing such a methodology could also be relevant for the assessment of baseline SDF levels in patients with oligospermia or severe oligoastheno-terato-zoospermia presenting low sperm counts, or even in sperm samples obtained directly from testicular sperm aspiration (TESA).
2. MATERIAL AND METHODS
A comprehensive analysis was performed on three different normozoospermic individuals facilitating identification and selection of non-vacuolated spermatozoa with normal morphology. The selection and analysis of 100 single spermatozoa obtained from 100 different trials was performed. Also performed were 30 additional trials selecting four spermatozoa per single capture using MSOME/IMSI. For sperm selected by MSOME/IMSI, a Petri dish (WillCo-dish, WillCo-Wells B.V., Amsterdam, The Netherlands) was prepared as described in Antinori, et al. [20]; namely, with 4 μl droplets of aqueous mounting media composed of 7% polyvinylpyrrolidone (MediCult, Jyllinge, Denmark). Additionally, a 4 μl droplet (selection droplet) of Ham’s F-10 medium with HEPES (Irvine Scientific, Santa Ana, CA, USA) was created to preserve selected spermatozoa. Sperm selection was based on the judgment of an embryologist selecting the most normal-looking spermatozoon available under 200 /400x magnification. Observation and selection was performed using an inverted microscope (Leica AM 6000; Leica Microsystems, Wetzlar, Germany), equipped with differential interference contrast (Nomarski) optics, a PlanAPO 100x Oil/1.50 objective lens, and a 0.55 numerical aperture condenser lens.
All captured spermatozoa were included in a single microdroplet and processed for the Sperm Chromatin Dispersion test (SCD-Oligo-Halosperm; Halotech DNA, Madrid, Spain) for sperm DNA damage assessment. However, to achieve the stated objectives, a slight modification of the basic methodology recommended by the manufacturers, particularly concerning staining procedures, was carried out. Namely, the gelled aliquot provided in the kit containing low-melting-point agarose in 100 µl Eppendorf tubes was placed in a water bath of 90˚C to 100˚C for 5 minutes to melt the agarose. Once liquefied, the temperature was equilibrated for 5 minutes in a water bath of 37˚C. The slide provided was then heated to 37˚C. This is a particularly important step insofar as the volume of agarose used to retain the selected spermatozoa is low and can be instantly gelled at room temperature. The recommended volume of melted agarose (10 µl) was then placed on the preheated slide in the centre of the circle as depicted on the slide (Figure 1(a)).
The slide, in turn, was then placed on the micromanipulator and the selected sperm were injected in the centre of the agarose microdroplet, close to the slide surface. Once the sperm had been injected, the microdroplet was covered using a 20 × 20 mm coverslip which had also been preheated to 37˚C. The slide was refrigerated at 4˚C for 5 minutes to allow the agarose to set and produce a thin microgel containing the spermatoza. While gently pressing the cover to expand the microdroplet, effort was taken to avoid the microgel expanding out of the marked circular area. In general, it was observed that the sperm tended to be localized at peripheral areas of the flattened
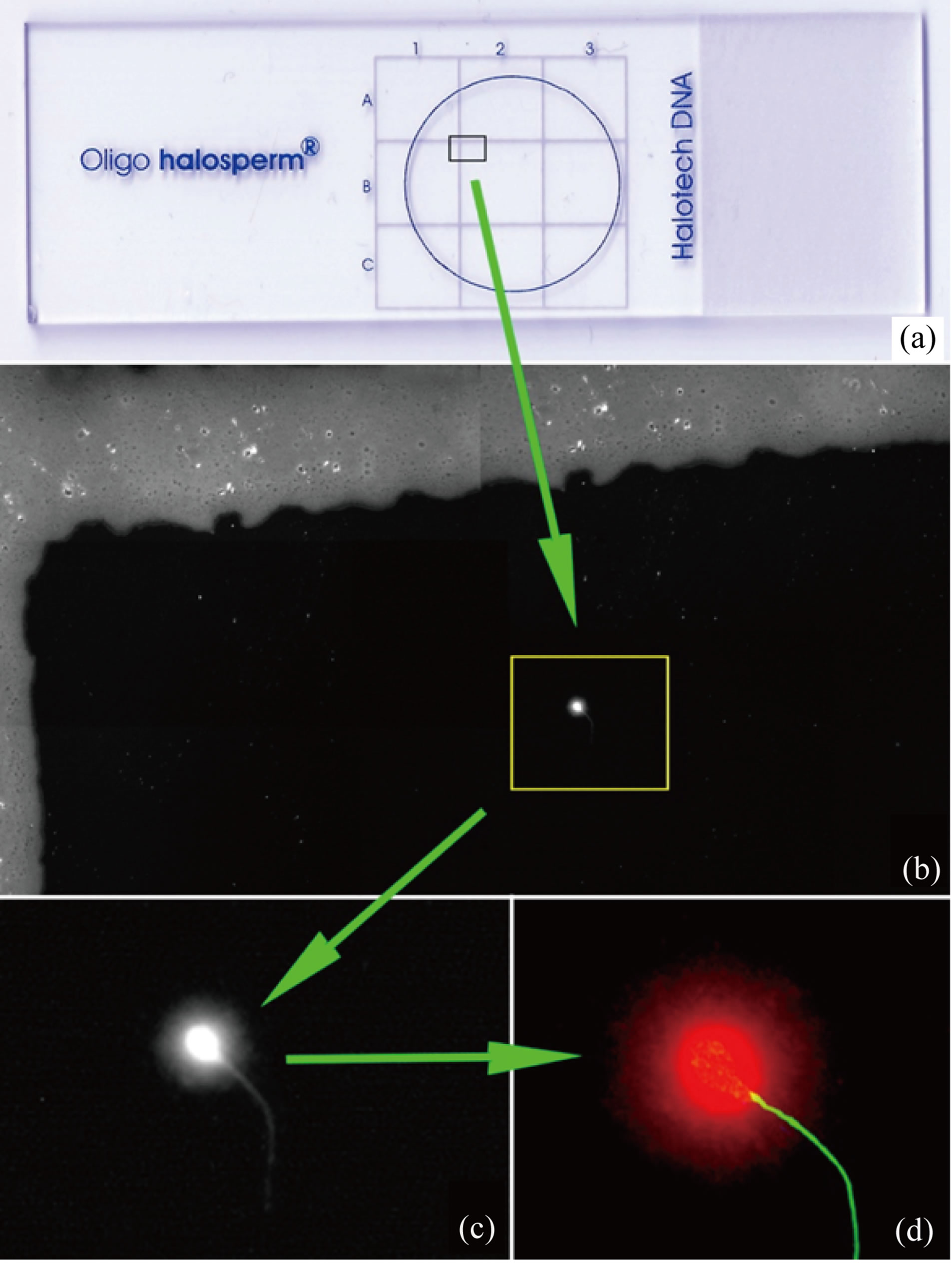
Figure 1. Single spermatozoa and different enlargement (a): sperm position on the slide; (b): x10 objective, cx40 objetive; (d): x63 objective) mapped on a single slide after selection with MSOME/IMSI and assessment of sperm DNA damage using the SCD-Oligo-Halosperm test.
gel. After 5 minutes of refrigeration, the coverslip was gently removed. Prior to processing to assess sperm DNA fragmentation, the slide could be scanned under phase contrast microscopy to localize sperm position. For this experiment, a Leica DMRB (Leica Microsystems, Wetzlar, Germany) phase contrast microscope with 20x magnification lenses with XY holder motorisation was used and facilitated the identification of sperm localisation coordinates. In some cases, spermatozoa were not found on the slide, thus assessment of sperm DNA damage in the sample could not be performed.
The physical mapping of a single sperm on the slide after IMSI-SCD-Oligo-Halosperm is shown in Figure 1(a). Once the spermatozoa were localized and their position(s) fixed, the slide was treated first for 7 minutes with the kit-provided denaturing solution and second for 20 minutes with lysing solution. Following a washing in distilled water for 5 minutes, the slides were dehydrated in ethanol baths of increasing concentration (70%, 90%, and 100%) for 2 minutes each and then air-dried. For staining and DNA visualization, a double-staining protocol was followed using a 0.5x GelRed fluorochrome (Biotium, Inc., Hayward, CA, USA) in an H-1000 Vectashield mounting medium (Vector Laboratories, Inc., Burlingame, CA, USA). For flagellum staining, particularly for protein thiols and disulfide residues, 0.05µg/ml of disodium 2’,7’-dibromo-4-(hydroxymercury)-fluorescein was used (Sigma-Aldrich, St. Louis, MO, USA). As shown in Figure 1(d), the dual emission fluorochrome combination allowed for simultaneous visualizetion of DNA (represented in red) and proteins (represented in green) using a dual bandpass fluorescence filter block. Alternatively, single emission fluorescence could be observed using single band-pass fluorescence filter blocks. Using the same microscope as in spermatozoa mapping, spermatozoa were then observed with epifluorescence. Images were captured via a Photometrics cooled CCD camera (Roper Industries, Inc., Duluth, GA, USA) for 16-bit direct colour captures and a Leica DFC350 FX CCD camera for 12-bit grey captures to perform image analysis.
For image analysis, the public-domain image J software (http://rsb.info.nih, gov) was used. All images were captured following 5 seconds of fluorescence exposure using a Leica 60x PlanFluotar NA 1.3 dry objective. For sperm characterization, a fixed threshold protocol was used for all captures. After background subtraction and in order to define the morphological features of each spermatozoa, additional calculations were made for area, perimeter, circularity and Feret’s diameter; the latter of which measuring the distance between parallel lines tangent to the profile of an object and calculating the greatest distance possible between any two points along the boundary of a region of interest. Statistical analysis was performed using the Statistical Package for Social Sciences 17 (SPSS, Inc., Chicago, IL, USA) and significant difference was defined for a p-value of <0.05.
3. RESULTS
For this preliminary study, spermatozoa were classified as either morphologically Normal and Non-Vacuolated (NNV; Figure 2(a)) or abnormal (Figures 2(b)-(c)). Among the abnormal forms, two main classes were defined: Highly Abnormal (HAb) sperm head morphology (Figure 2(b)) and Normal head morphology with Vacuolization (NV; Figure 2(c)).
For single-sperm DNA fragmentation analysis, sperm selection was performed using one spermatozoon or four spermatozoa belonging to the same pre-established group. Simultaneous MSOME/IMSI-SDF testing performed on single spermatozoa was possible in 84 out of the 100 trials (84% success rate). In multiple MSOME/IMSI, sperm selection where 4 spermatozoa had been recruited and injected into the same microdroplet, a 100% success rate per slide for sperm visualization and analysis was possible, inasmuch as at least one sperm could be visualized per slide. The total success rate for sperm analysis
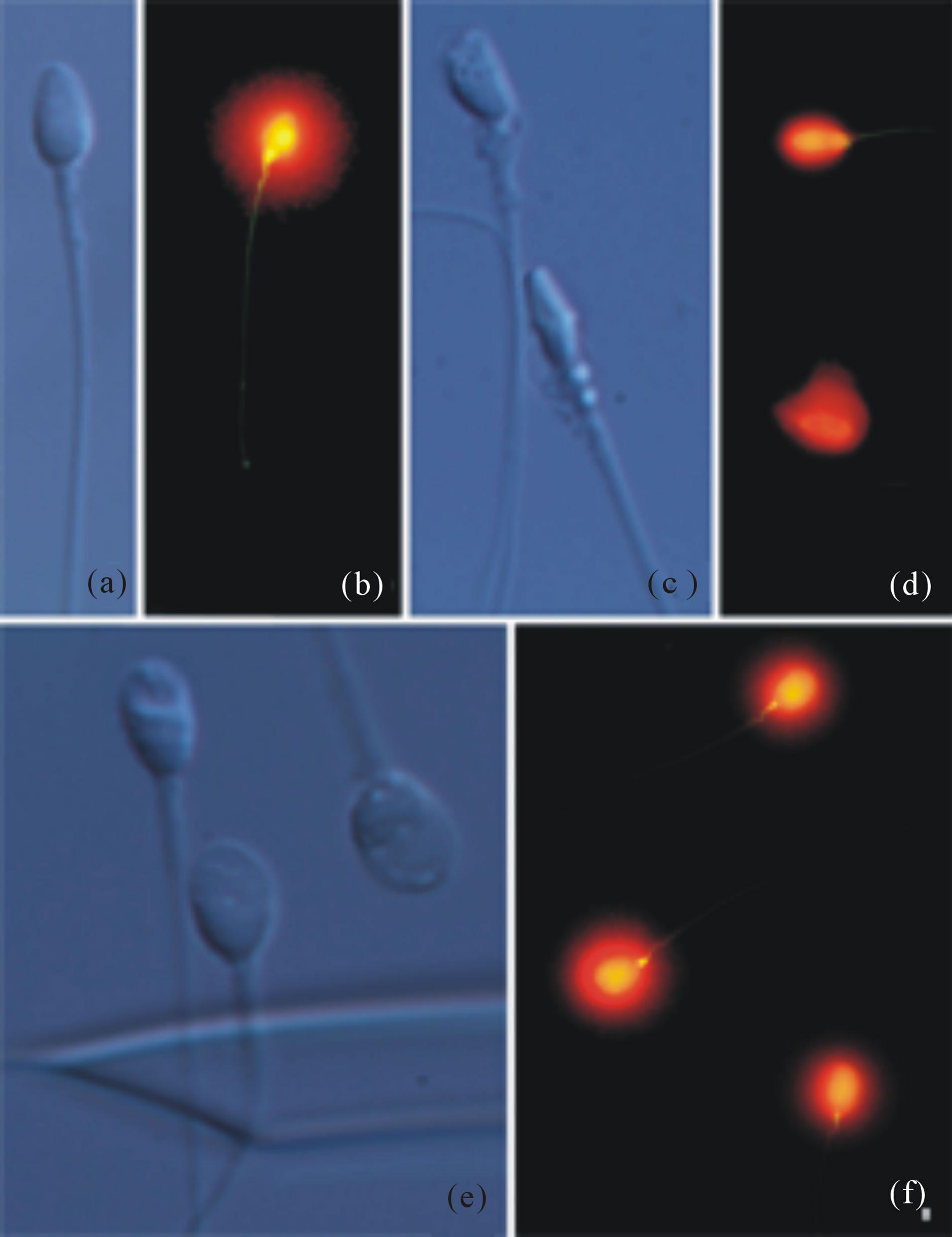
Figure 2. Visualisation of sperm morphology using MSOME /IMSI and DNA fragmentation assessment in selected spermatozoa. Normal spermatozoa showing absence of vacuoles (a) and absence of DNA damage in the same spermatozoa (b). Two highly abnormal spermatozoa (c) and presence of DNA fragmentation in both cases (d). Three spermatozoa showing presence of vacuolization (e) and lack of DNA fragmentation after the sperm the SCD-Oligo-Halosperm test (f).
was therefore approximately 70%, with 120 MSOME/ IMSI spermatozoa selected and 85 used in final analysis.
In NNV sperm following SCD-Oligo-Halosperm, chromatin dispersion halo morphology is generally quite large and symmetrical (Figure 1(b)), and demonstrates homogeneous and distinctive morphological features when compared to AV (Figure 2(d); Figure 3 plot). As regular chromatin dispersion halos are interpreted as representing a non-fragmented DNA molecule, none of these selected spermatozoa exhibited traces of DNA damage, as indicated in Table 1. Similarly, as the absence of such halos indicates DNA fragmentation, 74% of selected HAb spermatozoa were identified as demonstrating visible DNA damage (Figure 2(d) and Table 1). In general, the chromatin dispersion halos of NV spermatozoa tended to be smaller than those observed in NNV spermatozoa (comparison of Figure 2(f) with Figure 2(b) and Figure 3 plot). The level of SDF observed in this class was also relatively low (10%).
Image analysis to calculate the area, circularity, pe-rimeter and Feret’s diameter of 58 spermatozoa following SCD-Oligo-Halosperm was performed using the NNV, NV and HAb as clustering criteria. Descriptive statistics are shown in Figures 3(a)-(d).
Statistically significant differences were observed when area (non-parametric Kruskal-Wallis, χ2 = 47.3; P < 0.00), perimeter (non-parametric Kruskal-Wallis, χ2 = 42.0; P < 0.00), circularity (non-parametric KruskalWallis, χ2 = 33.8; P < 0.00) and Feret’s diameter (nonparametric Kruskal-Wallis, χ2 = 44.4; P < 0.00) were compared among the different pre-established groups. In three-dimensional plots of the different spermatozoa analysed (Figure 4) using area, circularity and Feret’s diameter values, NNV (black dots) are clustered differently than NV (white dots), while a certain degree of overlap is nevertheless present. Both, however, are clearly different from HAb (grey dots). Interestingly, the spermatozoa presenting a fully normal morphology with absence of vacuoles demonstrated the highest area, perimeter and Feret’s diameter values, but the lowest values for circularity.
4. DISCUSSION
The results obtained in this study show that MSOME /IMSI combined with the SCD-Oligo-Halosperm test can be reliably used to assess sperm DNA damage in selected single spermatozoa, therefore, corroborate the existence of a direct relationship between sperm morphological patterns and DNA quality. Additionally, the present investigation shows that MSOME/IMSI-selected sperm, targeted for their normal morphology and lack of traces of vacuolization, are fully free of DNA damage. However, traces of vacuolization and highly severe morphological alterations in the sperm are correlated with minor and/or large increases in the possibility of selecting sperm containing a damaged DNA molecule. With regard to the subtle morphological differences observed between NNV and NV sperm, these differences are also registered in halo morphology following the SCD-Oligo-Halosperm test and image analysis. This fact likely indicates the interference of the morphological alterations with halo formation or, alternatively, subtle deleterious sperm chromatin changes that, putatively, may partially interfere with fertilization or pregnancy.
Nevertheless, the results found here clearly indicate that, while 100% of NNV sperm show an absence of DNA damage, at least 10% of NV sperm may present a damaged DNA molecule. This raises the question of if, with standard sperm selection using standard systems under regular phase contrast microscopy, we inadvertently increase the probability of selecting deficient spermatozoa with low-quality DNA. The observations made in this experiment also appear to support the data reported by some authors claiming that MSOME/IMSI,
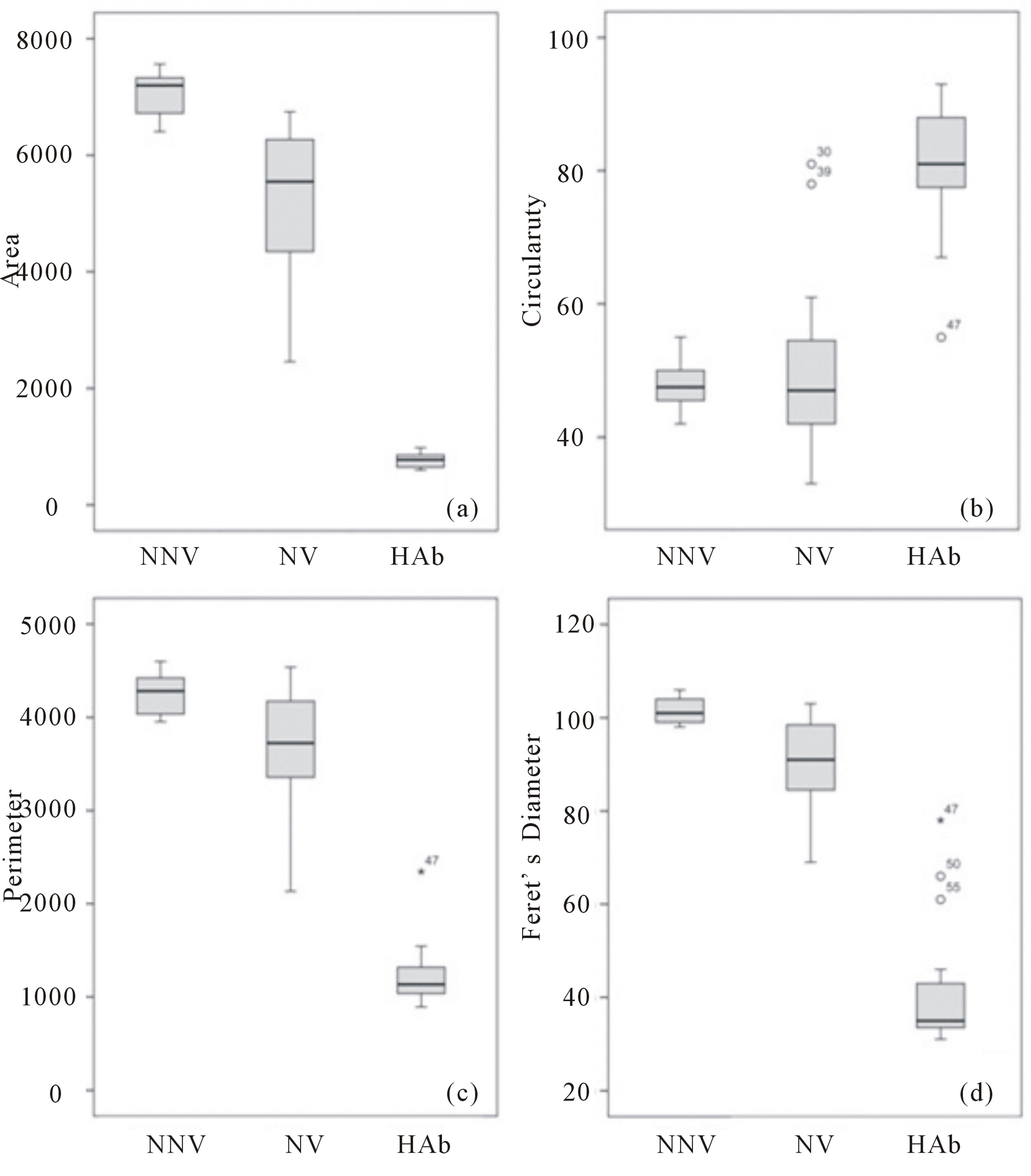
Figure 3. Descriptive statistics using a box-and-whisker plot for Normal Non-Vacuolated (NNV), Normal Vacuolated (NV) and Highly Abnormal (HAb) spermatozoa processed with SCD-Oligo-Halosperm with regard to (a) area, (b) circularity, (c) perimeter and (d) Feret’s diameter.

Table 1. Distribution of DNA fragmentation in the three preestablished subclasses of spermatozoa according to morphology: NNV (Normal Non-Vacuolated), HAb (Highly Abnormal) and NV (Normal Vacuolated).
due to its ability to detect small morphological abnormalities and other defects in human spermatozoa that could otherwise be selected through ICSI, may increase the pregnancy rate [17,20]. In Wilding, et al. [19], it was reported that, following MSOME/IMSI analysis, 64.8% of the spermatozoa observed would not have been suitable for ICSI procedures due to morphological alterations. The same study also found a correlation between morphological alterations observed through MSOME /IMSI and DNA fragmentation as assessed with the TUNEL assay. Also in this investigation, if we compare
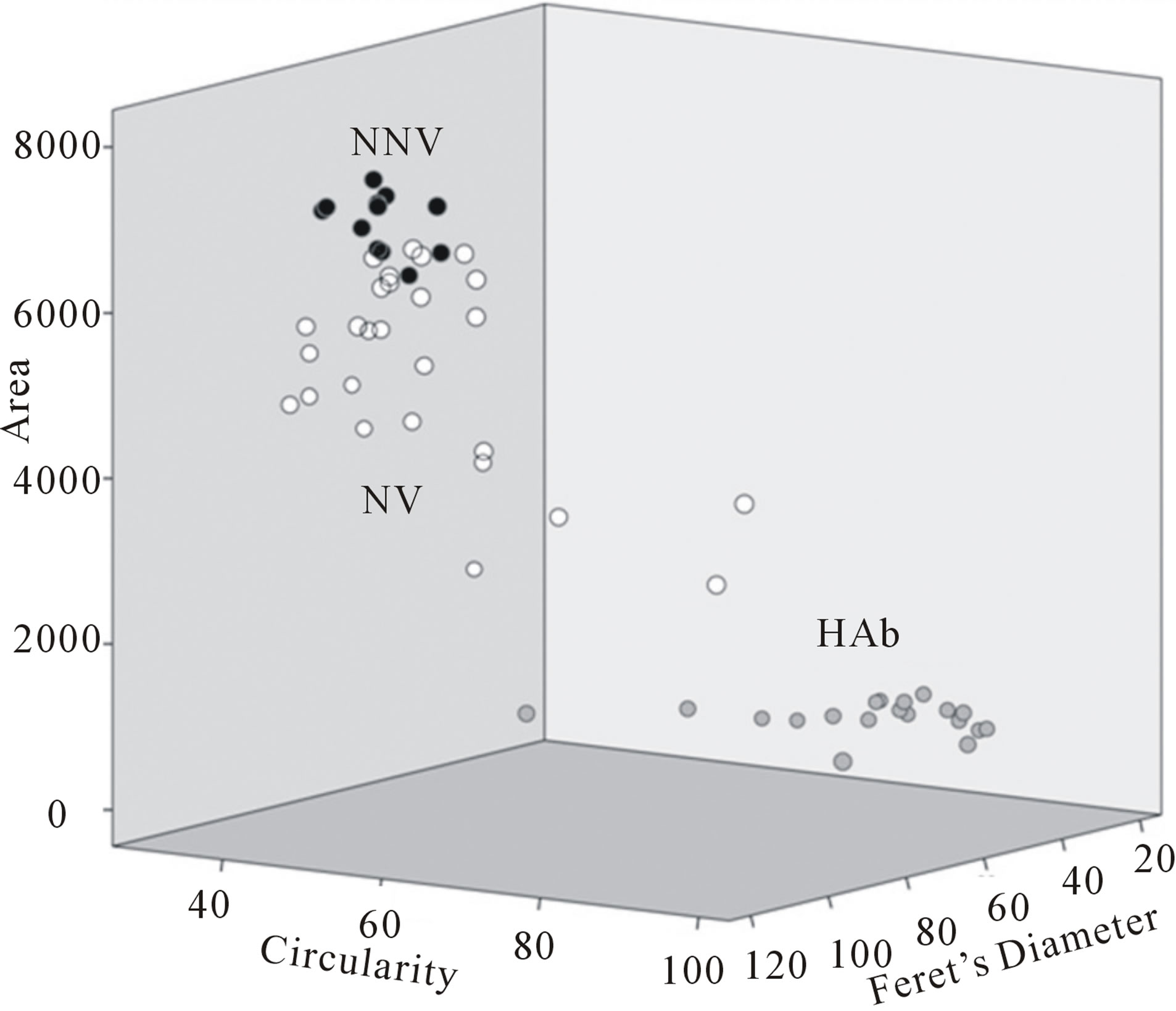
Figure 4. Differential plotting of area, circularity and Feret’s diameter in 42 different spermatozoa. Normal Non-Vacuolated (NNV; black dots), Highly Abnormal (HAb ; Grey dots) and Normal Vacuolated (NV; white dots).
positive/negative spermatozoa for TUNEL, in groups around 100 spermatozoa selected using MSOME/IMSI, there is a significant increase for DNA damage in all the groups where abnormal MSOME/IMSI were included.
In a clear congruence with the results in this experimental approach, Franco Jr. et al. [18] suggested the existence of a correlation between spermatozoa with low levels of vacuolization and the presence of high or low levels of DNA damage. Although, in this case, the correlation was established indirectly, the results can be seen as parallel to those obtained here directly; that is, the lack of any DNA damage detected in sperm selected according to strict criteria for good head and tail morphology and the absence of vacuolization. What seems evident is that effective sperm selection through the application of technical alternatives such a MSOME/IMSI, Physiologic ICSI (PICSI) using the capacity of some sperm to react with hyaluronic acid, or even, under more extreme conditions such as semen sex-sorting using flow cytometry, tend to reduce the frequency of spermatozoa presenting a damaged DNA molecule in the selected subpopulation [18,21-23], an effect which should theoretically result in improved pregnancy rates.
However, and despite the high hopes of researchers with regard to pregnancy rates in the early phases of MSOME/IMSI use, positive net results are not so clear. On the one hand, MSOME/IMSI appears to improve embryo development, as well as laboratory and clinical outcomes of sperm microinjection in the same infertile couples, presenting male infertility and poor embryo development, when compared to previous ICSI attempts [24]. Thus, the results indicate that a positive clinical outcome from in vitro fertilization in couples with previous poor embryo development was possible, a finding which would be congruent with more effective DNA quality selection. On the other hand, however, it has also been found that, in other cases of couples having suffered repeated implantation failures using conventional ICSI, MSOME/IMSI did not provide a significantly improved clinical outcome [25]. Furthermore, in a prospective randomized study [26] comparing the clinical outcome of 87 IMSI cycles with 81 conventional ICSI cycles, a similar tendency was observed with respect to the pregnancy rate. That said, when MSOME/IMSI versus ICSI were compared, in general, tendencies of the former for higher rates of implantation (28.9% to 19.5%), clinical pregnancy (54.0% to 44.4%) and live births (43.7% to 38.3%) were also observed. Thus, it is quite likely that only the comparative prospective randomized study of a large number of cases will, in the end, be able to shed some light on the possible clinical benefits of this technique. For the moment, however, suffice it to say of MSOME /IMSI that no negative effects have ever been demonstrated, and that good sperm morphology and high-quality sperm DNA are ensured through the use of these criteria.
Finally, it is interesting to note that NNV spermatozoa selected exhibit large and symmetrical chromatin dispersion halos in the SCD-Oligo-Halosperm test. It may be speculated that this particular morphology is the result of an ordered protein depletion occurring in highly ordered sperm chromatin. The morphological characteristics of the halo—including area, perimeter, circularity and Feret’s diameter—are significantly different in sperm with traces of vacuolization, and extremely so in those spermatozoa presenting bad morphology and damaged DNA. According to the model of sperm chromatin organization presented by Ward and his co-workers [27-30], it seems that chromatin relaxation following the controlled protein depletion used in SCD-Oligo-Halosperm may originate in the DNAse hypersensitive sites and matrix attachment regions (MARs) connecting the different protamine toroids (see Figure 1 in [31]). These MARs, could be candidates to bind more sensitive proteins to be removed and, following SCD-Oligo-Halosperm, an ordered release of connected, yet relaxed toroids, could form well-ordered halos of dispersed chromatin. The symmetry on the haloes would be the result of a highly-ordered and orthodox sperm chromatin structure. Morphological alterations can be related to deficient chromatin packing and/or protamination, producing haloes of smaller size after protein depletion. In any case, the differential sperm morphology achieved after protein depletion and its correlation with DNA damage could be potentially exploited to analyze if a high frequency of large and symmetrical haloes present in some patients, would be indicative of generally higher-quality sperm.
5. ACKNOWLEDGEMENTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. This research was supported with public funding (Spanish Ministry of Science and Technology (MCYT: BFU2010-16738)
REFERENCES
- Guzick, D.S., Overstreet, J.W., Factor-Litvek, P., Brazil, C.K., Nakajima, S.T., Coutifaris, C., Carson, S.A. et al. (2001) Comparison of sperm morphology, motility, and concentration in fertile and infertile men. New England Journal of Medicine, 345, 1388-1393. doi:10.1056/NEJMoa003005
- Schlegel, P.N. (2009) Evaluation of male infertility. Minerva Ginecologica, 61, 261-283.
- Zini, A. and Libman, J. (2006) Sperm DNA damage, clinical significance in the era of assisted reproduction. Canadian Medical Association Journal, 175, 495-500. doi:10.1503/cmaj.060218
- Castilla, J.A., Zamora, S., Gonzalvo, M.C., Luna Del Castillo, J.D., Roldan-Nofuentes, J.A., Clavero, A., et al. (2010) Sperm chromatin structure assay and classical semen parameters, systematic review. Reproductive Biomedicine Online, 20, 114-124. doi:10.1016/j.rbmo.2009.10.024
- Kruger, T.F., Acosta, A.A., Simmons, K.F., Swanson, R.J., Matta, J.F., Veeck, L.L., Morshedi, M., Brugo, S. (1987) New method of evaluating sperm morphology with predictive value for human in vitro fertilization. Urology, 30, 248-251. doi:10.1016/0090-4295(87)90246-9
- Kruger, T.F., Lombard, C.J. and Ombelet, W. (2001) Predictive value of normal sperm morphology in intrauterine insemination (IUI), a structured literature review. Human Reproduction Update, 7, 495-500. doi:10.1093/humupd/7.5.495
- Ombelet, W., Wouters, E., Boels, L., Cox A., Janssen, M., Spiessens, C., Vereecken, A., et al., (1997) Sperm morphology assessment, diagnostic potential and comparative analysis of strict or WHO criteria in a fertile and a subfertile population. International Journal of Andrology, 20, 367-372. doi:10.1046/j.1365-2605.1998.00079.x
- Van Waart, J., Kruger, T.F., Lombard, C.J., Ombelet, W. (2001) Predictive value of normal sperm morphology in intrauterine insemination (IUI), a structured literature review. Human Reproduction Update, 5,495-500. doi:10.1093/humupd/7.5.495
- Borini, A., Tarozzi, N., Bizzaro, D., Bonu, M.A., Fava, L., Flamigni, C. and Coticchio, G. (2006) Sperm DNA fragmentation, paternal effect on early post-implantation embryo development in ART. Human Reproduction, 21, 2876-2881. doi:10.1093/humrep/del251
- Evenson, D.P. (2013) Sperm chromatin structure assay (SCSA®). Methods in Molecular Biology, 927, 147-164. doi:10.1007/978-1-62703-038-0_14
- Bartoov, B., Berkovitz, A., Eltes, F., Kogosowski, A., Menezo, Y. and Barak, Y. (2002) Real-time fine morphology of motile human sperm cells is associated with IVF-ICSI outcome. Journal of Andrology, 23, 1-8.
- Berkovitz, A., Eltes, F., Ellenbogen, A., Peer, S., Feldberg, D. and Bartoov, B. (2006) Does the presence of nuclear vacuoles in human sperm selected for ICSI affect pregnancy outcome? Human Reproduction, 21, 1787-1790. doi:10.1093/humrep/del049
- Berkovitz, A., Eltes, F., Lederman, H., Peer, S., Ellenbogen, A., Feldberg, B. and Bartoov, B. (2006) How to improve IVF-ICSI outcome by sperm selection. Reproductive Biomedicine Online, 12, 634-638. doi:10.1016/S1472-6483(10)61191-1
- Cassuto, N.G., Bouret, D., Plouchart, J.M., Jellad, S., Vanderzwalmen, P., Balet, R., Larue, L., et al. (2009) A new real-time morphology classification for human spermatozoa, a link for fertilization and improved embryo quality. Fertility and Sterility, 92, 1616-1625. doi:10.1016/j.fertnstert.2008.08.088
- Chelli, M.H., Albert, M., Ray, P.F., Guthauser, B., Izard V., Hammoud, I., Selva, J., et al. (2010) Can intracytoplasmic morphologically selected sperm injection be used to select normal-sized sperm heads in infertile patients with macrocephalic sperm head syndrome? Fertilty and Sterility, 93, e1-e5.
- Bartoov, B., Berkovitz, A., Eltes, F., Kogosovsky, A., Yagoda, A., Lederman, H., Artzi, S., et al., (2003) Pregnancy rates are higher with intracytoplasmic morphologically selected sperm injection than with conventional intracytoplasmic injection. Fertilty and Sterility, 80, 1413-1419. doi:10.1016/j.fertnstert.2003.05.016
- Hazout, A., Dumont-Hassan, M., Junca, A.M., Cohen Bacrie, P. and Tesarik, J. (2006) High magnification ICSI overcomes paternal effect resistant to conventional ICSI. Reproductive Biomedicine Online, 12, 19-25. doi:10.1016/S1472-6483(10)60975-3
- Franco, J.G. Jr, Baruffi, R.L.R., Mauri, A.L., Petersen C.G., Oliveira J.B.A. and Vagnini, L. (2008) Significance of large nuclear vacuoles in human spermatozoa, implications for ICSI. Reproductive Biomedicine Online, 17, 42- 45.
- Wilding, M., Coppola, G., di Matteo, L., Palagiano, A., Fusco, E. and Dale, B. (2011) Intracytoplasmic injection of morphologically selected spermatozoa (IMSI) improves outcome after assisted reproduction by deselecting physiologically poor quality spermatozoa. Journal of Assisted Reproduction and Genetics, 28, 253-262. doi:10.1007/s10815-010-9505-5
- Antinori, M., Licata, E., Dani, G., Cerusico, F., Versaci, C., D’Angelo, D. and Antinori, S. (2008) Intracytoplasmic morphologically selected sperm injection: A prospective randomized trial. Reproductive Biomedicine Online, 16, 835-841. doi:10.1016/S1472-6483(10)60150-2
- Parmegiani, L., Cognigni, G.E., Bernardi, S., Troilo, E, Ciampaglia, W. and Filicori, M. (2010) ‘‘Physiologic ICSI’’, Hyaluronic acid (HA) favors selection of spermatozoa without DNA fragmentation and with normal nucleus, resulting in improvement of embryo quality. Fertility and Sterility, 93, 598-604. doi:10.1016/j.fertnstert.2009.03.033
- Gosálvez, J., Ramirez, M.A., López-Fernández, C., Crespo, F., Evans, K.M., Kjelland, M.E. and Moreno, J.F. (2011) Sex-sorted bovine spermatozoa and DNA damage: I. Static features. Theriogenology, 15, 197-205.
- Cassuto, N.G, Hazout, A., Hammoud, I., Balet, R., Bouret, D., Barak, Y., Sonia, S., Jellad, S. et al. (2012) Correlation between DNA defect and sperm-head morphology. Reproductive Biomedicine Online, 24, 211-218. doi:10.1016/j.rbmo.2011.10.006
- Knez, K., Zorn, B., Tomazevic T., Vrtacnik-Bokal, E. and Virant-Klun, I. (2011) The IMSI procedure improves poor embryo development in the same infertile couples with poor semen quality: A comparative prospective randomized study. Reproductive Biology and Endocrinology, 9, 123. doi:10.1186/1477-7827-9-123
- Oliveira, J.B., Massaro, F.C., Mauri, A.L., Petersen, C.G., Nicoletti, A.P., Baruffi, R.L. and Franco, J.G. Jr. (2009) Motile sperm organelle morphology examination is tricter than Tygerberg criteria. Reproductive Biomedicine Online, 18, 320-326. doi:10.1016/S1472-6483(10)60088-0
- Balaban, B., Yakin, K., Alatas, C., Oktem, O., Isiklar, A. and Urman, B. (2011) Clinical outcome of intracytoplasmic injection of spermatozoa morphologically selected under high magnification, a prospective randomized study. Reproductive Biomedicine Online, 5, 472-476. doi:10.1016/j.rbmo.2010.11.003
- Ward, W.S. and Coffey. D.S., (1991) DNA packaging and organization in mammalian spermatozoa, comparison with somatic cells. Biology of the Reproduction, 44, 569- 574. doi:10.1095/biolreprod44.4.569
- Ward, W.S. (1993) DNA loop domain tertiary structure in mammalian spermatozoa. Biology of the Reproduction, 48, 1193-1201. doi:10.1095/biolreprod48.6.1193
- Ward, W.S. (2010) Function of Sperm Chromatin Structural Elements in Fertilization and Development. Molecular Human Reproduction, 16, 30-36. doi:10.1093/molehr/gap080
- Ortega, M.A., Sil, P., and Ward, W.S. (2011) Mammalian Sperm Chromatin as a Model for Chromatin Function in DNA Degradation and DNA Replication. Systems Biology in Reproductive Medicine, 57, 43-49. doi:10.3109/19396368.2010.505679
- Ward, M.A. and Ward, W.S. (2004) A model for the function of sperm DNA degradation. Reproduction Fertility and Development, 16, 547-554. doi:10.1071/RD03072

