Advances in Bioscience and Biotechnology
Vol.4 No.3A(2013), Article ID:29273,9 pages DOI:10.4236/abb.2013.43A055
Transposable elements in Escherichia coli antimicrobial resistance
![]()
1Department of Biochemistry, King Abdulaziz University, Jeddah, KSA
2King Fahd Medical Research Center, King Abdulaziz University, Jeddah, KSA
3Department of Animal and Veterinary Sciences, American University of Beirut, Beirut, Lebanon
4Special Infectious Agents Unit—Biosafety Level 3, King Fahd Medical Research Center, King Abdulaziz University, Jeddah, KSA
5Medical Laboratory Technology Department, Faculty of Applied Medical Sciences, King Fahd Medical Research Center, King Abdulaziz University, Jeddah, KSA
6Infection Control and Antimicrobial Drug Resistance, University of Benghazi, Benghazi, Libya
7Infection Control and Environmental Health Unit, King Abdulaziz University Hospital, King Abdulaziz University, Jeddah, KSA
8Lebanese American University, Beirut, Lebanon
Email: *sharakeh@gmail.com
Received 4 January 2013; revised 6 February 2013; accepted 25 February 2013
Keywords: Transposable Elements; E. coli; Antimicrobial Resistance; Integrons
ABSTRACT
Transposable elements are capable of switching their positions on the genome thereby causing gene arrangements and contributing to genome evolution. The aim of this review is to specifically discuss the role of transposable elements in transferring antimicrobial resistance genes in E. coli, thus contributing to increase in virulence and conferring the possibility of multidrug resistance. Different types of transposable elements such as transposons and integrons and their profound influence on E. coli antimicrobial resistance are the focus of this review.
1. INTRODUCTION
In 1983 Barbara McClintock earned her Nobel prize after discovering jumping genes or transposons in maize. At the time of the discovery of transposons, there was much speculation about their credibility because they conferred elasticity to the genome, much contradictory to the genetic stability proven by Gregor Mendel [1]. But now, almost seven decades later, owing to the astonishing advances in molecular techniques, the structure and function of the transposons and their role in genomic evolution have been proven beyond any doubt [2].
2. TRANSPOSABLE ELEMENT(S)
{(TE(s)} are DNA sequences that are capable of changing their positions within the genome, creating new mutations and may change the cell’s genome size and often result in the duplication of the individual TE [2]. In general, they are considered as junk DNA and they make up a large proportion of the C-value of the eukaryotic cell and are useful in research to modify the DNA inside the living organisms [3]. TEs possess two main characteristics that differentiate them from other genetic elements. As they are mobile, they are capable of changing their genetic environment and in doing so they alter the genetic environment of the locus into which they insert [4]. Since they have the intrinsic ability to multiply during the transposition process, they are automatically amplified, with a large copy number, restricted only by the carrying capacity of their environment (i.e. the genome) [1]. Transposons often persist in genomes over millions of years. This requires an exquisite balance between replication and suppression of their activity. In fact, transposons often have specific targeting mechanisms that exploit “safe havens” in the genome, such as noncoding or transcriptionally repressed regions, that help them survive for longer periods [5].
Several types of transposable elements are present. One of those is mobile genetic elements which includes two classes based on their mechanism of transposition, and can best be described as either “copy and paste” (for Class I TEs) or “cut and paste” (for Class II TEs) [6].
2.1. Class I (Retro-Transposons)
They function by copying themselves from DNA to RNA by transcription and vice versa by reverse transcription. The resulting DNA copy will subsequently be inserted into the genome at a different position. The reverse transcriptase enzyme will initiate the reverse transcription, which is often coded for by the TE itself. Retro-transposons behave very similarly to retroviruses, such as HIV [6].
2.2. Class II (DNA Transposons)
In this case, an RNA intermediate is not needed for the cut-and-paste transposition mechanism of Class II TEs which are catalyzed by various types of transposase enzymes [4]. Some of those transposases do not need any specific target site to bind. In contrast, others can only bind to specific sequence targets. The transposase produce a staggered cut at the target site, forming sticky ends. The transposase will then remove the DNA transposon and ligates it into the target site [6]. The DNA polymerase fills in the resulting gaps from the sticky ends and DNA ligase closes the sugar-phosphate backbone. The final outcome is target site duplication and the production of short direct repeats at the insertion sites of DNA transposons. As a consequence, gene duplications can result and is an important evolutionary process [7].
The cut and paste mechanism is not always needed for all DNA transposons. In certain instances, replicative transposition is noted upon the replication of the transposon to a new target site. There is always the possibility for TEs to lose their ability to synthesize either reverse transcriptase or transposase through mutation without losing their ability to jump through the genome [8]. In bacteria, transposons have an additional function often to confer antibiotic resistance through the presence of an additional gene on a plasmid. There is always the possibility for transposons to jump from chromosomal DNA to plasmid DNA and vice versa, producing antibiotic or in many instances multi drug resistance genes belonging to the Tn family in bacteria [9]. In case the transposable elements do not have the additional genes, they are known as insertion sequences. Some of the common insertion sequence elements in E. coli are listed in Table 1.
Transposable elements are one of the major forces that drive prokaryote genome evolution. Analyses of TE evolutionary dynamics revealed extensive variability in TE density between prokaryote genomes, even closely related [10]. To explain this variability, a model of recurrent invasion/proliferation/extinction cycles has been proposed by Cerveau et al. [10], wherein they examined different parameters that influence these cycles in two of the simplest TE classes: insertion sequences and group II introns. In particular, they have focused on TE transTable 1. E. coli insertion sequences.
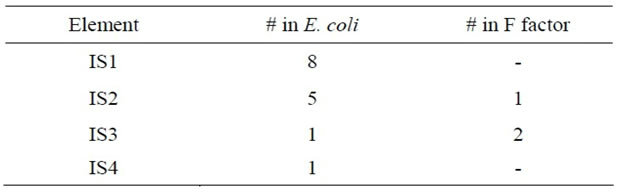
position efficiency (mechanisms and regulation), ability to transfer horizontally (through plasmids and phages), and impact on genome evolution (gene activation/inactivation and structural variation).
Currently, there is an intense interest in studying the evolution of TEs and their effects on the bacterial genome. There may be a universal common ancestor for all TEs or may have evolved in an independent manner from one another and spread to other kingdoms through horizontal gene transfer [11]. Some of them may benefit the host while others are considered like viruses as selfish DNA parasites. As a matter of fact, some TEs share similarities in their genome to viruses, indicating possible common ancestor as viruses. Bacteria have developed a specific mechanism of gene deletion to prevent excessive TE activity, which may lead to the destruction of its chromosome. TEs may have the ability to transpose to conjugative plasmids and some have integrons which are genetic elements able to catch and express genes from other sources. Integrons have the integrase enzyme that can integrate gene cassettes. Around 40 antibiotic resistance genes have already been identified on cassettes, and hence have virulence genes. The objective of this review is to delineate the involvement of transposable elements in E. coli antimicrobial resistance.
3. ANTIMICROBIAL RESISTANCE IN E. COLI
Antimicrobial drugs have played an important role in alleviating illness and death associated with infectious diseases in animals and humans. However, selective pressure exerted by antimicrobial drug use and also irrational use of antibiotics also have been the major factors behind the emergence and spread of drug-resistance traits among pathogenic and commensal bacteria [12]. In addition, resistance has developed after invention of every major class of antimicrobial drugs, varying in time from as short as 1 year (penicillin) to >10 years (vancomycin) [13].
The development of multi-drug resistance in E. coli is of major concern worldwide. Although prevalence rates for resistant E. coli strains are significantly distinct for various populations and environments and the impact of resistance to antimicrobial drugs is ubiquitous [14]. Erb et al., [15] reviewed the emergence trends in E. coli antimicrobial resistance from 1970 to 2006 and observed that the prevalence of resistance to specific antibiotics was highly variable in different populations and in different countries, ranging from 0% to 100%. The prevalence of resistance to a wide range of antibiotics reported in studies from Middle and South America, Spain, and Turkey was higher than that reported in the USA and Central Europe. Moreover, a tendency towards higher prevalence rates of resistance in recent years was observed.
Widespread antibiotic resistance has been recognized in E. coli isolates from human, animal and environmental sources [16]. A very recent study by Adachi et al., [17] showed the occurrence of fluoroquinolones and fluoroquinolone-resistance genes in E. coli in the aquatic environment in Japan. This is the first report of plasmid-mediated quinolone resistance determinant qnrS1 in the aquatic environment. Another recent study by Ryu et al., [18] on antimicrobial resistance and resistance genes in E. coli strains isolated from commercial fish and seafood in Korea showed that Class 1 and Class 2 integrons were associated with conferring resistance of these strains to a wide range of antibiotics. Another bothersome area is the infectious E. coli of animal origin that are multi drug resistant. Because of the intensive use of antimicrobial agents in food animal production, meat is frequently contaminated with antimicrobial-resistant E. coli. Humans can be colonized with E. coli of animal origin and because of resistance to commonly used antimicrobial agents, these bacteria may cause infections for which limited therapeutic options are available. This may lead to treatment failure and can have serious consequences for the patient. Furthermore, E. coli of animal origin may act as a donor of antimicrobial resistance genes for other pathogenic E. coli [16]. There are numerous reports that address most recent findings and indicate the severity and magnitude of antimicrobial resistance in E. coli from a wide spectrum of environments and enumerating them here would neither be possible nor pertinent. The aim of this review is to focus on the role of transposable elements and the underlying molecular mechanisms that aid in development of versatility in E. coli in acquiring and disseminating antimicrobial resistance.
4. THE ROLE OF MOBILE GENETIC ELEMENTS IN E. COLI ANTIMICROBIAL RESISTANCE
Bacterial evolution has been largely influenced by the high plasticity of bacterial genomes, thus leading to their adaptation to most ecosystems. This ability to exchange and rearrange genomic sequences to gain new traits has been extensively documented with the development of resistance to antibiotics [19]. Today, the increasing rate of antibiotic resistant bacteria is a major public health issue [20]. During the last decade, several studies have highlighted the environmental resistance as a source of resistance genes of clinical interest [21-24].
A variety of genetic elements are strongly associated with the exchange and transfer of antimicrobial resistance genes among gram-negative bacteria. Transposons can transfer resistance genes intracellularly. On the other hand, integrons are genetic elements that can acquire several resistance genes in the form of gene cassettes. These genetic elements sometimes co-exist and work cooperatively. For example, integrons, which are not mobile by themselves, can acquire an ability to transfer when they are incorporated into transposons [25]. In this section, we are reviewing the individual contribution of integrons and transposons in acquiring antimicrobial resistance and genomic evolution of E. coli.
5. TRANSPOSONS IN E. COLI
E. coli is known to harbour a wide range of transposons such as Tn3, Tn5, Tn7, Tn9 and Tn10, each conferring resistance to antibiotics, namely, Tn3 to ampicillin, Tn5 to kanamycin, Tn7 to trimethoprim, streptothricin, spectinomycin, streptomycin, Tn9 to chloramphenicol and Tn10 to tetracycline respectively [26]. Some of the common E. coli transposons and their antibiotic resistance are listed in Table 2. The Tn5 transposon is known to harbour a gene cassette of three genes conferring resistance to three antibiotics: the ble gene for bleomycin resistance, the neo gene for neomycin and the str gene for streptomycin resistance [27].
Among the numerous and important classes of transposons are the conjugative transposons. As their name implies, conjugative transposons are mobile elements that possess the genetic machinery to facilitate their own transfer between bacterial cells. Referring back at some pioneering work in this field about two decades ago, several prototypes of conjugative transposons have been described [35]. An earlier misconception was that these transposons were largely found only on Gram-positive bacteria but later on it was observed that the transposon groups Tn916 and Tn1545 transfer readily to a wide
Table 2. Common E. coli transposons.

variety of gram-positive and gram-negative bacteria in the laboratory. In one report, Enterococcus faecalis readily transferred plasmid-borne Tn916 to Alcaligenes eutrophus, Citrobacter freundii, and E. coli, conferring selectable levels of tetracycline resistance [1]. It has been assumed that acquiring antimicrobial resistance mediated by transposons bestows upon the bacteria a fitness impact enabling it to survive better under selection pressure. E. coli is known to have acquired resistance to chloramphenicol, tetracycline and kanamycin from genes carried on transposons [36]. Therefore, as is evidenced by the above reports, starting from earlier antibiotics such as tetracycline, kanamycin and chloramphenicol to the new generation trimethoprim and sulfonamides, a wide array of transposons are involved in imparting resistance [37]. The broad-spectrum antibacterial trimethoprim is heavily used worldwide, most commonly in combination with sulphonamides. Trimethoprim inhibits the enzyme dihydrofolate reductase (Dfr). E. coli most commonly becomes resistance acquiring one of over 30 known dfr genes encoding resistant variants, most of which belong to the dfrA category [38]. Recently, Labar et al., [39] found the regional dissemination of a trimethoprim-resistance gene cassette via a successful transposable element. They observed a 50% increase in the prevalence of trimethoprim resistance among fecal E. coli from healthy Nigerian students between 1998 and 2005, a trend to increase that continued in 2009. In this study 16.3% isolates carried transposons conferring trime-thoprim resistance, indicating transposition as a principal mechanism for disseminating dfrA7 among E. coli from Nigeria and Ghana.
With the recent innovations in technology for analyzing the molecular genetics of bacterial pathogens, understanding of the role of transposons in the dissemination of antimicrobial resistance has expanded significantly. Although a lot of attention has been focused on transposon mediated transfer of antibiotic resistance, the wide spread involvement of integrons in conferring multi drug resistance is being strongly asserted in recent studies. Integrons are genetic elements that can collect several resistance genes in the form of gene cassettes. These genetic elements sometimes co-exist and work cooperatively. For example, integrons, which are not mobile by themselves, can obtain an ability to transfer when they are incorporated into transposons. It thus becomes very important to focus on this class of transposable elements, which seem to have relied initially on transposons to mobilise them, but have rapidly evolved in their ability to rapidly disseminate wider range of resistance to various antimicrobials in E. coli. Hence the next section is aimed at documenting some of the recent evidences for integron mediated antimicrobial resistance in E. coli from different environments.
6. ROLE OF INTEGRONS IN ACTIMICROBIAL RESISTANCE
Integrons contain an intI gene encoding a site-specific recombinase belonging to the integrase family and a recombination site attI. A gene cassette includes an open reading frame and, at the 3’-end, a recombination site attC. Integration or excision of cassettes occurs by a sitespecific recombination mechanism catalyzed by the integrase [40]. At least six classes of integrons have been determined according to their intl gene. Classes 1, 2 and 3 are the most studied and are largely associated with the dissemination of antibiotic resistance. Most gene cassettes encode antibiotic resistant determinants [41]. Integrons seem to play a major role in the spread of multidrug resistance in Gram-negative bacteria but recently they have been described in Gram-positive bacteria [20]. Moreover, the finding of super-integrons with gene cassettes coding for other determinants (biochemical functions, virulence factors) in different Gram negative bacteria suggests that integrons are probably involved in bacterial genome evolution [40]. More than 100 gene cassettes may be present, in contrast to resistance integrons where less than ten cassettes are present. Many species harbour super-integrons, which are species-specific, whereas particular resistance integrons can be found in a variety of species. In the last few years, a variety of new gene cassettes have been described. Many of these encode resistance against newer generation antibiotics such as cephalosporins and carbapenems [39].
7. TYPES OF INTEGRONS
With relation to acquisition of antimicrobial resistance, two major groups of integrons have been described: “chromosomal integrons” (CIs), and “mobile integrons” (MIs). They are located on the chromosome of hundreds of bacterial species; in fact, in silico analysis showed that 17% of sequenced bacterial genomes exhibit such genetic arrangements [38]. MIs are not self-transposable elements but are found on mobile genetic elements such as plasmids and transposons, which aid in their dissemination among bacteria. MIs contain a limited number of GCs (less than 10 GCs) [42]. The GCs described to date in these MIs usually encode antibiotic-resistance determinants. MIs are therefore sometimes also called “resistant integrons” (RIs) or “multidrug resistance integrons” (MRIs).
7.1. Integrons and Antibiotic Resistance in E. coli from Various Environments
Antibiotic pressure has probably played a key role in the MIs selection and dissemination in bacteria. More than 130 GCs conferring resistance to antibiotics and more than 60 GCs of unknown functions have been described in MIs [43]. Genes involved in resistance to almost all antibiotic families are embedded in GCs, including betalactams, aminoglycosides, trimethoprim, macrolides, chloramphenicol, fosfomycin, lincosamides, rifampicin and quinolones. Studies have suggested that MIs were more prevalent in bacterial communities subjected to direct or indirect antibiotic pressure in clinical, agricultural, and environmental settings [44-48]. Recent in vitro studies have shown that antibiotics are able to induce integrase transcription, both in CIs and MIs, via the SOS response. The SOS response is a global regulatory network controlled by the transcriptional repressor LexA and induced by stress leading to direct or indirect DNA damage, such as damage resulting from exposure to some widely used antibiotics such as fluoroquinolones, beta-lactams, trimethoprim, aminoglycosides [49-51]. The activation of the SOS response in bacteria results in integrase over expression, which leads to the raise of GCs recombinetion events.
Clinical, veterinary and environmental surveys have shown that bacteria harboring MIs are often associated with multidrug-resistant (MDR) phenotypes [52]. Recently, many studies have been conducted and have documented the involvement of integrons in acquisition of antibiotic resistance in E. coli, out of which some of the most recent findings are being discussed in this section. Integrons have been implicated in aiding antibiotic resistance in E. coli in a wide range of environments such as polluted water, poultry, food, hospitals, etc.
7.2. Integrons in E. coli from Polluted Waters
Chen et al., [53] carried out a study on Class I integrons in antibiotic resistance in E. coli strains isolated from the Minjiang River drainage basin in Fujian Province, which is one of China’s most intensive livestock and poultry production areas. According to this study, a total of 666 (24%) E. coli isolates carried at least one of the virulence genes elt, fedA, astA, fasA, estA, stx(2e), paa, and sepA. Forty-one percent of the isolates harbored class 1 integrons, and these isolates had a significantly higher probability of resistance to tobramycin, cefoperazone, cefazolin, ciprofloxacin, norfloxacin, azitromycin, and rifampin than isolates with no Class 1 integron detected. Frequencies of resistance to selected antibiotics were as high as or higher than those in fecal, wastewater, and clinical isolates in published surveys undertaken in China, North America, and Europe. Another recent study [54] conducted on E. coli isolates from the Dongjiang River in South China showed that and 87.5% of the isolates were resistant to at least three antibiotics. The presence of integrons (Classes 1 and 2) was frequently observed (82.3%) while no class 3 integron was found. Twentyone resistance genes of 14 gene cassette arrays and 10 different families of resistance genes were identified in these integrons. The above two recent studies have clearly documented the role of integrons in acquiring antimicrobial resistance.
Koczura et al., [55] studied the antimicrobial resistance of integron-harboring E. coli isolates from clinical samples, wastewater treatment plant and river water. They used multiplex PCR assay to determine the frequency of integron presence in E. coli isolates cultured from wastewater treatment plant (WWTP) (integrons were detected in 11% of E. coli isolates), river water upstream (6%) and downstream (14%) the discharge of WWTP, and clinical specimens (56%). Antimicrobial resistance of the integron-positive isolates, determined by disk diffusion method, varied between E. coli of different origin. Isolates from the downstream river, compared to those cultured from upstream river, were more frequently resistant to kanamycin, cephalotin, co-trimoxazole, trimethoprim, and fluoroquinolones. Moreover, they displayed broader resistance ranges, expressed as the number of classes of antimicrobials to which they were resistant.
7.3. Integrons in E. coli from Animal Sources and Poultry
Soufi et al., [56] studied the prevalence and diversity of integrons and associated resistance genes in E. coli isolates from poultry meat in Tunisia and observed a high rate of resistance to a wide range of antibiotics and were found to harbour various integrons. Sixty percent of isolates harbored integrons (Class 1, 30 isolates; Class 2, 5 isolates). Class 2 integrons contained in all cases the dfrA1-sat1-aadA1-orfX gene cassette arrangement. Nine gene cassette arrangements have been detected among Class 1 integrons, containing different alleles of dfrA (five alleles) and aadA (2 alleles) genes, which encode trimethoprim and streptomycin resistance, respectively. An uncommon gene cassette array (sat-psp-aadA2-cmlA1- aadA1-qacH-IS440-sul3) was identified in three class 1 integron-positive isolates. The study concluded that E. coli isolates of poultry origin could be a reservoir of antimicrobial-resistance genes and of integrons. Kadlec and Schwarz [57] conducted a study in a veterinary farm in Germany on identification of integron-associated resistance genes among E. coli isolates from horses, pigs and dogs. They reported the involvement of both Classes I and 2 integrons associated with resistance to a wide range of antibiotics. Another study [58] reported the association of tetracycline resistance with Class I integrons in E. coli isolates from beef cattle. All these findings show us the alarming incidence of multiresistant, integron harbouring E. coli in the veterinary environment.
7.4. Integrons in E. coli from Human Sources
Recently, Najibi et al., [59] studied the distribution of Class 1 integrons among enteropathogenic E. coli. They investigated the incidence of and resistance gene content of Class 1 integrons among enteropathogenic E. coli (EPEC) and non-EPEC and to investigate intraspecies genetic diversity of EPEC strains isolated from children with diarrhea in Iran. They reported that 82% EPEC isolates and 11 68.7% non-EPEC isolates harbored the int1 gene specific to the conserved integrase region of Class 1 integrons. A recent study [60], on EPEC in children with diarrhea in Iran, showed the presence of 61% of atypical E. coli isolates resistant to six different antibiotics, all harbouring Class 2 integrons. Highly resistant E. coli strains are not only found in humans with disease but have, also, been reported in the faeces of healthy humans. When Vinue et al., [61] studied the prevalence and diversity of integrons and associated resistance genes in faecal E. coli isolates of healthy humans in Spain, they found integrases associated with Class 1 and/or Class 2 integrons belonging to seven different gene cassettes, conferring resistance to a wide spectrum of antibiotics in 29% of the E. coli isolates. This leads to a worrisome speculation that individuals in the community could be a reservoir of integron-containing E. coli isolates and improper sanitation could easily cause the rapid dissemination of these resistant strains.
All the above mentioned reports draw our attention not only to the presence of antibiotic resistance conferring integrons in E. coli from a wide range of environments, but also highlight the vast molecular diversity of the integron gene cassettes present in these strains. The molecular diversity is a key factor that gives these strains a selective advantage for rapid dissemination in the environment.
8. INSERTION SEQUENCE COMMON REGION ELEMENT (ISCR)
Having discussed in detail the role of transposable elements in antimicrobial resistance, one is bound to highlight the recent concept of insertion sequence common region element (ISCR) and their role as a gene capture system in organisms. ISCR elements differ from the insertion sequences by lacking terminal inverted repeats (IRs), do not generate directly repeated sequence on insertion and are thought to be transposed by a mechanism defined as rolling-circle (RC) transposition [62]. ISCR elements are closely related to an unusual family of insertion sequences called the IS91 family. ISCR elements, as a novel gene-capturing system, are capable of mobilizing any piece of adjacent DNA sequences [63]. This powerful gene mobilization mechanism serves as a highly mobile vector or milieu in transferring antibiotic resistance genes between different species of bacteria
[64]. Nineteen members of the ISCR family have been discovered until now in many Gram-negative pathogens. The majority of these elements are found to be closely associated with antimicrobial resistance genes that are not necessary components of the host genome, suggesting that ISCR elements may be responsible for the rapid transmission of bacterial multi-drug resistance in the environment [65]. ISCR elements are now recognized as powerful antibiotic resistance gene capture and movement systems that also possess the ability to construct extended clusters of antibiotic resistance genes on plasmids as well as on chromosomes [66]. As a result, there is a lot of focus on these elements as major contributors to bacterial antimicrobial resistance and research in this area in the future would definitely provide deeper insights into this versatile phenomenon of genome evolution.
9. CONCLUDING REMARKS
E. coli, usually a commensal bacterium of humans and animals is a highly versatile and ubiquitous bacterium. Pathogenic variants cause a wide range of infections such as intestinal and extraintestinal infections, including gastroenteritis, urinary tract infection, meningitis, peritonitis and septicemia [67]. Surveillance data show that resistance in E. coli is consistently highest for antimicrobial agents that have been in use the longest time in human and veterinary medicine [68]. The past two decades have witnessed major increases in emergence and spread of multidrug-resistant bacteria and increasing resistance to newer compounds such as fluoroquinolones and certain cephalosporins [14]. Intensive molecular analyses have shown multiple mechanisms in which E. coli attain the antimicrobial resistance, the chief contributions to this genomic fitness being horizontal gene transfer in terms of conjugative plasmids, transducing phages and transposable elements carrying resistance genes. Of the various contributing genetic elements, this review aims at assessing the recent information available on the role of transposable elements in dissemination of multidrug resistance traits to E. coli. The transposons and integrons, owing to their genomic plasticity have contributed a great deal to the fitness quotient and robustness of E. coli to survive in varying environments. The information presented here is an indication of the magnitude of genomic variation that has resulted due to the transposable elements and also fortifies the role of these elements in genomic evolution, thoroughly justifying the predictions of Barbara McClintok that trasnposons will play a major role in genomic diversity and evolution.
REFERENCES
- Hua-Van, A., Rouzic, A.L., Boutin, T.S., Filee, J. and Capy, P. (2011) The struggle for life of the genome’s selfish architects. Biology Direct, 6, 19. doi:10.1186/1745-6150-6-19
- Shapiro, J.A. (2010) Mobile DNA and evolution in the 21st century. Mobile DNA, 1, 4. doi:10.1186/1759-8753-1-4
- Bao, W., Jurka, M.G., Kapitonov, V.V. and Jurka, J. (2009) New superfamilies of eukaryotic DNA transposons and their internal divisions. Molecular Biology and Evolution, 26, 983-993. doi:10.1093/molbev/msp013
- Wicker, T., Sabot, F., Hua-Van, A., Bennetzen, J.L., Capy, P., Chalhoub, B., Flavell, A., Leroy, P., Morgante, M., Panaud, O., Paux, E., SanMiguel, P. and Schulman A.H. (2007) A unified classification system for eukaryotic transposable elements. Nature Reviews Genetics, 8, 973-982. doi:10.1038/nrg2165
- Martin, S.L. and Garfinkel, D.J. (2003) Survival strategies for transposons and genomes. Genome Biology, 4, 313. doi:10.1186/gb-2003-4-4-313
- Kapitonov, V.V. and Jurka, J. (2008) A universal classification of eukaryotic transposable elements implemented in Repbase. Nature Reviews Genetics, 9, 411-412. doi:10.1038/nrg2165-c1
- Madigan, M.T., Martinko, J.M. and Brock, T.D. (2006) Brock biology of microorganisms. Pearson Prentice Hall, Upper Saddle River.
- Feschotte, C. and Pritham, E.J. (2007) DNA transposons and the evolution of eukaryotic genomes. Annual Review of Genetics, 41, 331-368. doi:10.1146/annurev.genet.40.110405.090448
- Wagner, A. (2006) Periodic extinctions of transposable elements in bacterial lineages: Evidence from intragenomic variation in multiple genomes. Molecular Biology and Evolution, 23, 723-733. doi:10.1093/molbev/msj085
- Cerveau, N., Leclerq, S., Bouchon, D. and Cordaux, R. (2011) Evolutionary dynamics and genomic impact of prokaryote transposable elements. In: Pontarotti, P., Ed., Springer, Heidelberg and New York, 291-312.
- C. Feschotte (2008) Transposable elements and the evolution of regulatory networks. Nature Reviews Genetics, 9, 397-405. doi:10.1038/nrg2337
- Aarestrup, F.M., Wegener, H.C. and Collignon, P. (2008) Resistance in bacteria of the food chain: Epidemiology and control strategies. Expert Review of Anti-Infective Therapy, 6, 733-750. doi:10.1586/14787210.6.5.733
- Levy, S.B. and Marshall, B. (2004) Antibacterial resistance worldwide: Causes, challenges and responses. Nature Medicine, 10, S122-129. doi:10.1038/nm1145
- von Baum, H. and Marre, R. (2005) Antimicrobial resistance of Escherichia coli and therapeutic implications. International Journal of Medical Microbiology, 295, 503- 511. doi:10.1016/j.ijmm.2005.07.002
- Erb, A., Sturmer, T., Marre, R. and Brenner, H. (2007) Pre- valence of antibiotic resistance in Escherichia coli: Overview of geographical, temporal, and methodological variations. European Journal of Clinical Microbiology & Infectious Diseases, 26, 83-90. doi:10.1007/s10096-006-0248-2
- Hammerum, A.M. and Heuer, O.E. (2009) Human health hazards from antimicrobial-resistant Escherichia coli of animal origin. Clinical Infectious Diseases, 48, 916-921. doi:10.1086/597292
- Adachi, F., Yamamoto, A., Takakura, K.I. and Kawahara, R. (2013) Occurrence of fluoroquinolones and fluoroquinolone-resistance genes in the aquatic environment. Science of the Total Environment, 444C, 508-514. doi:10.1016/j.scitotenv.2012.11.077
- Ryu, S.H., Park, S.G., Choi, S.M., Hwang, Y.O., Ham, H.J., Kim, S.U., Lee, Y.K., Kim, M.S., Park, G.Y., Kim, K.S. and Chae, Y.Z. (2012) Antimicrobial resistance and resistance genes in Escherichia coli strains isolated from commercial fish and seafood. International Journal of Medical Microbiology, 152, 14-18. doi:10.1016/j.ijfoodmicro.2011.10.003
- Stalder, T., Barraud, O., Casellas, M., Dagot, C. and Ploy, M.C. (2012) Integron involvement in environmental spread of antibiotic resistance. Front Microbiol, 3, 119. doi:10.3389/fmicb.2012.00119
- Davies, J. and Davies, D. (2010) Origins and evolution of antibiotic resistance. Microbiology and Molecular Biology Reviews, 74, 417-433. doi:10.1128/MMBR.00016-10
- Wright, G.D. (2010) Antibiotic resistance in the environment: A link to the clinic? Current Opinion in Microbiology, 13, 589-594. doi:10.1016/j.mib.2010.08.005
- Martinez, J.L. (2008) Antibiotics and antibiotic resistance genes in natural environments. Science, 321, 365-367. doi:10.1126/science.1159483
- D’Costa, V.M., McGrann, K.M., Hughes, D.W. and Wright, G.D. (2006) Sampling the antibiotic resistome. Science, 311, 374-377. doi:10.1126/science.1120800
- Aminov, R.I. and Mackie, R.I. (2007) Evolution and ecology of antibiotic resistance genes. FEMS Microbiology Letters, 271, 147-161. doi:10.1111/j.1574-6968.2007.00757.x
- Harada, S. (2012) Genetic elements involved in the acquisition of antimicrobial resistance genes in bacteria. Nihon Rinsho, 70, 329-332.
- Roberts, A.P., Chandler, M., Courvalin, P., Guedon, G., Mullany, P., Pembroke, T., Rood, J.I., Smith, C.J., Summers, A.O., Tsuda, M. and Berg, D.E. (2008) Revised nomenclature for transposable genetic elements. Plasmid, 60, 167-173. doi:10.1016/j.plasmid.2008.08.001
- Reznikoff, W.S. (2003) Tn5 as a model for understanding DNA transposition. Molecular Microbiology, 47, 1199- 1206. doi:10.1046/j.1365-2958.2003.03382.x
- Kopecko, D.J. and Cohen, S.N. (1975) Site specific recA— Independent recombination between bacterial plasmids: Involvement of palindromes at the recombinational loci. Proceedings of the National Academy of Sciences of the United States of America, 72, 1373-1377. doi:10.1073/pnas.72.4.1373
- Berg, D.E., Davies, J., Allet, B. and Rochaix, J.D. (1975) Transposition of R factor genes to bacteriophage lambda. Proceedings of the National Academy of Sciences of the United States of America, 72, 3628-3632. doi:10.1073/pnas.72.9.3628
- Barth, P.T., Datta, N., Hedges, R.W. and Grinter, N.J. (1976) Transposition of a deoxyribonucleic acid sequence encoding trimethoprim and streptomycin resistances from R483 to other replicons. Journal of Bacteriology, 125, 800- 810.
- Gottesman, M.M. and Rosner, J.L. (1975) Acquisition of a determinant for chloramphenicol resistance by coliphage lambda. Proceedings of the National Academy of Sciences of the United States of America, 72, 5041-5045. doi:10.1073/pnas.72.12.5041
- Foster, T.J., Howe, T.G. and Richmond, K.M. (1975) Trans- location of the tetracycline resistance determinant from R100-1 to the Escherichia coli K-12 chromosome. Journal of Bacteriology, 124, 1153-1158.
- Oka, A., Sugisaki, H. and Takanami, M. (1981) Nucleotide sequence of the kanamycin resistance transposon Tn903. Journal of Molecular Biology, 147, 217-226. doi:10.1016/0022-2836(81)90438-1
- So, M. and McCarthy, B.J. (1980) Nucleotide sequence of the bacterial transposon Tn1681 encoding a heat-stable (ST) toxin and its identification in enterotoxigenic Escherichia coli strains. Proceedings of the National Academy of Sciences of the United States of America, 77, 4011-4015. doi:10.1073/pnas.77.7.4011
- Rice, L.B. (1998) Tn916 family conjugative transposons and dissemination of antimicrobial resistance determinants. Antimicrobial Agents and Chemotherapy, 42, 1871- 1877.
- Enne, V.I., Delsol, A.A., Davis, G.R., Hayward, S.L., Roe, J.M. and Bennett, P.M. (2005) Assessment of the fitness impacts on Escherichia coli of acquisition of antibiotic resistance genes encoded by different types of genetic element. Journal of Antimicrobial Chemotherapy, 56, 544-551. doi:10.1093/jac/dki255
- Reznikoff, W.S. (2008) Transposon Tn5. Annual Review of Genetics, 42, 269-286. doi:10.1146/annurev.genet.42.110807.091656
- Cambray, G., Guerout, A.M. and Mazel, D. (2010) Integrons. Annual Review of Genetics, 44, 141-166. doi:10.1146/annurev-genet-102209-163504
- Labar, A.S., Millman, J.S., Ruebush, E., Opintan, J.A., Bishar, R.A., Aboderin, A.O., Newman, M.J., Lamikanra, A. and Okeke, I.N. (2012) Regional dissemination of a trimethoprim-resistance gene cassette via a successful transposable element. PLoS One, 7, Article ID: e38142. doi:10.1371/journal.pone.0038142
- Fluit, A.C. and Schmitz, F.J. (2004) Resistance integrons and super-integrons. Clinical Microbiology and Infection, 10, 272-288. doi:10.1111/j.1198-743X.2004.00858.x
- Ploy, M.C., Lambert, T., Couty, J.P. and Denis, F. (2000) Integrons: An antibiotic resistance gene capture and expression system. Clinical Chemistry and Laboratory Medicine, 38, 483-487. doi:10.1515/CCLM.2000.070
- Naas, T., Mikami, Y., Imai, T., Poirel, L. and Nordmann, P. (2001) Characterization of In53, a class 1 plasmidand composite transposon-located integron of Escherichia coli which carries an unusual array of gene cassettes. Journal of Bacteriology, 183, 235-249. doi:10.1128/JB.183.1.235-249.2001
- Partridge, S.R., Tsafnat, G., Coiera, E. and Iredell, J.R. (2009) Gene cassettes and cassette arrays in mobile resistance integrons. FEMS Microbiology Reviews, 33, 757- 784. doi:10.1111/j.1574-6976.2009.00175.x
- Skurnik, D., Menac’h A.L., Zurakowski, D., Mazel, D., Courvalin, P., Denamur, E., Andremont, A. and Ruimy, R. (2005) Integron-associated antibiotic resistance and phylogenetic grouping of Escherichia coli isolates from healthy subjects free of recent antibiotic exposure. Antimicrobial Agents and Chemotherapy, 49, 3062-3065. doi:10.1128/AAC.49.7.3062-3065.2005
- Luo, Y., Mao, D., Rysz, M., Zhou, Q., Zhang, H., Xu, L. and Pedro, J.J.A. (2010) Trends in antibiotic resistance genes occurrence in the Haihe River, China. Environmental Science & Technology, 44, 7220-7225. doi:10.1021/es100233w
- Kristiansson, E., Fick, J., Janzon, A., Grabic, R., Rutgersson, C., Weijdegard, B., Soderstrom, H. and Larsson, D.G. (2011) Pyrosequencing of antibiotic-contaminated river sediments reveals high levels of resistance and gene transfer elements. PLoS One, 6, Article ID: e17038. doi:10.1371/journal.pone.0017038
- Daikos, G.L., Kosmidis, C., Tassios, P.T., Petrikkos, G., Vasilakopoulou, A., Psychogiou, M., Stefanou, I., Avlami, A. and Katsilambros, N. (2007) Enterobacteriaceae bloodstream infections: Presence of integrons, risk factors, and outcome. Antimicrobial Agents and Chemotherapy, 51, 2366-2372. doi:10.1128/AAC.00044-07
- Barlow, R.S., Fegan, N. and Gobius, K.S. (2009) Integron- containing bacteria in faeces of cattle from different production systems at slaughter. Journal of Applied Microbiology, 107, 540-545. doi:10.1111/j.1365-2672.2009.04240.x
- Guerin, E., Cambray, G., Sanchez-Alberola, N., Campoy, S., Erill, I., Da Re, S., Gonzalez-Zorn, B., Barbe, J., Ploy, M.C. and Mazel, D. (2009) The SOS response controls integron recombination. Science, 324, 1034. doi:10.1126/science.1172914
- Cambray, G., Sanchez-Alberola, N., Campoy, S., Guerin, E., Da Re, S., Gonzalez-Zorn, B., Ploy, M.C., Barbe, J., Mazel, D. and Erill, I. (2011) Prevalence of SOS-mediated control of integron integrase expression as an adaptive trait of chromosomal and mobile integrons. Mobile DNA, 2, 6. doi:10.1186/1759-8753-2-6
- Baharoglu, Z., Bikard, D. and Mazel, D. (2010) Conjugative DNA transfer induces the bacterial SOS response and promotes antibiotic resistance development through integron activation. PLOS Genetics, 6, Article ID: e1001165. doi:10.1371/journal.pgen.1001165
- Laroche, E., Pawlak, B., Berthe, T., Skurnik, D. and Petit, F. (2009) Occurrence of antibiotic resistance and class 1, 2 and 3 integrons in Escherichia coli isolated from a densely populated estuary (Seine, France). FEMS Microbiology Ecology, 68, 118-130. doi:10.1111/j.1574-6941.2009.00655.x
- Chen, B., Zheng, W., Yu, Y., Huang, W., Zheng, S., Zhang, Y., Guan, X., Zhuang, Y., Chen, N. and Topp, E. (2011) Class 1 integrons, selected virulence genes, and antibiotic resistance in Escherichia coli isolates from the Minjiang river, Fujian province, China. Applied and Environmental Microbiology, 77, 148-155. doi:10.1128/AEM.01676-10
- Su, H.C., Ying, G.G., Tao, R., Zhang, R.Q., Zhao, J.L. and Liu, Y.S. (2012) Class 1 and 2 integrons, sul resistance genes and antibiotic resistance in Escherichia coli isolated from Dongjiang River, South China. Environmental Pollution, 169, 42-49. doi:10.1016/j.envpol.2012.05.007
- Koczura, R., Mokracka, J., Jablonska, L., Gozdecka, E., Kubek, M. and Kaznowski, A. (2012) Antimicrobial resistance of integron-harboring Escherichia coli isolates from clinical samples, wastewater treatment plant and river water. Science of the Total Environment, 414, 680- 685. doi:10.1016/j.scitotenv.2011.10.036
- Soufi, L., Abbassi, M.S., Saenz, Y., Vinue, L., Somalo, S., Zarazaga, M., Abbas, A., Dbaya, R., Khanfir, L., Ben Hassen, A., Hammami, S. and Torres, C. (2009) Prevalence and diversity of integrons and associated resistance genes in Escherichia coli isolates from poultry meat in Tunisia. Foodborne Pathogens and Disease, 6, 1067-1073. doi:10.1089/fpd.2009.0284
- Kadlec, K. and Schwarz, S. (2008) Analysis and distribution of class 1 and class 2 integrons and associated gene cassettes among Escherichia coli isolates from swine, horses, cats and dogs collected in the BfT-GermVet monitoring study. Journal of Antimicrobial Chemotherapy, 62, 469-473. doi:10.1093/jac/dkn233
- Wu, R.B., Alexander, T.W., Li, J.Q., Munns, K., Sharma, R. and McAllister, T.A. (2011) Prevalence and diversity of class 1 integrons and resistance genes in antimicrobial-resistant Escherichia coli originating from beef cattle administered subtherapeutic antimicrobials. Journal of Applied Microbiology, 111, 511-523. doi:10.1111/j.1365-2672.2011.05066.x
- Najibi, S., Bakhshi, B., Fallahzad, S., Pourshafie, M.R., Katouli, M., Sattari, M., Alebouyeh, M. and Tajbakhsh, M. (2012) Distribution of class 1 integrons among enteropathogenic Escherichia coli. Canadian Journal of Microbiology, 58, 637- 643. doi:10.1139/w2012-035
- [61] Bakhshi, B., Fallahzad, S. and Pourshafie, M.R. (2012) The occurrence of atypical enteropathogenic Escherichia coli strains among children with diarrhea in Iran. Journal of Infection and Chemotherapy. doi:10.1007/s10156-012-0526-0
- [62] Vinue, L., Saenz, Y., Somalo, S., Escudero, E., Moreno, M.A., Ruiz-Larrea, F. and Torres, C. (2008) Prevalence and diversity of integrons and associated resistance genes in faecal Escherichia coli isolates of healthy humans in Spain. Journal of Antimicrobial Chemotherapy, 62, 934- 937. doi:10.1093/jac/dkn331
- [63] Toleman, M.A., Bennett, P.M. and Walsh, T.R. (2006) ISCR elements: Novel gene-capturing systems of the 21st century? Microbiology and Molecular Biology Reviews, 70, 296-316. doi:10.1128/MMBR.00048-05
- [64] Toleman, M.A., Bennett, P.M. and Walsh, T.R. (2006) Common regions e.g. orf513 and antibiotic resistance: IS91- like elements evolving complex class 1 integrons. Journal of Antimicrobial Chemotherapy, 58, 1-6. doi:10.1093/jac/dkl204
- [65] Deng, Y., Zeng, Z., Liu, J. and Chen, Z. (2009) Insertion sequence common region element: A novel gene-capturing system in bacteria—A review. Acta microbiologica Sinica, 49, 987-993.
- [66] Call, D.R., Singer, R.S., Meng, D., Broschat, S.L., Orfe, L.H., Anderson, J.M., Herndon, D.R., Kappmeyer, L.S., Daniels, J.B. and Besser, T.E. (2010) BlaCMY-2-positive IncA/C plasmids from Escherichia coli and Salmonella enterica are a distinct component of a larger lineage of plasmids. Antimicrobial Agents and Chemotherapy, 54, 590-596. doi:10.1128/AAC.00055-09
- [67] Toleman, M.A. and Walsh, T.R. (2010) ISCR elements are key players in IncA/C plasmid evolution. Antimicrobial Agents and Chemotherapy, 54, 3534. doi:10.1128/AAC.00383-10
- [68] Tadesse, D.A., Zhao, S., Tong, E., Ayers, S., Singh, A., Bartholomew, M.J. and McDermott, P.F. (2012) Antimicrobial drug resistance in Escherichia coli from humans and food animals, United States, 1950-2002. Emerging Infectious Diseases, 18, 741-749. doi:10.3201/eid1805.111153
- [69] FDA (2010) National antimicrobial resistance monitoring system enteric bacteria (NARMS): 2008 executive report. http://www.fda.gov/AnimalVeterinary/SafetyHealth/AntimicrobialResistance/NationalAntimicrobialResistanceMonitoring System/default.htm
NOTES
*Corresponding author.

