Food and Nutrition Sciences
Vol. 2 No. 6 (2011) , Article ID: 6609 , 9 pages DOI:10.4236/fns.2011.26075
Influence of Jet-Cooking Corn Bran on Its Antioxidant Activities, Phenolic Contents and Viscoelastic Properties
![]()
USDA, Agricultural Research Service, National Center for Agricultural Utilization Research, Functional Foods Research Unit, Peoria, USA.
Email: George.Inglett@ars.usda.gov
Received April 29th, 2011; revised May 28th, 2011; accepted June 6th, 2011.
Keywords: corn bran, phenolic contents, antioxidant activity, jet-cooking, water holding capacity, viscoelastic properties
ABSTRACT
Corn bran was subjected to high-shear and jet-cooking with or without alkaline treat-ment. The highest antioxidant activity was found in the soluble solids from jet-cooked corn bran without alkaline treatment. Jet-cooking under alkaline conditions resulted in a soluble fraction having the highest phenolic content but without increasing antioxidant activity. Phenolic contents of soluble solids were significantly higher than the insoluble solids. A colorimetric method using spectrophotometer was suitable to determine total phenolic content, whereas LC-ESI-MS technology was used for identifying important individual phenolic acids, namely caffeic, coumaric and ferulic acid. The insoluble solids from alkaline treatment had the highest water holding capacity and interesting viscoelastic properties. These results suggested that jet-cooking corn bran may be a useful processing procedure for creating phytochemical and functional products.
1. Introduction
Corn bran has received a renewed interest as a phytochemical source. Recent studies found that corn bran contain polyamine conjugates and related hydroxycinnamic acids [1,2]. Corn bran is also rich in several functional lipid constituents, including unsaturated fatty acids, tocopherols and phytosterols, and dietary fiber and carotenoid pigments [3]. Corn bran contains ferulic acid which has bioactivity related to the medicinal functions of R. A. sinensis, one of the most commonly used traditional Chinese medicine [4]. Traditional extraction method for ferulic acid was to reflux R. A. sinensis for 4 - 5 h in 70% ethanol [5].
Corn bran fiber can provide a good source of dietary fiber. Women with high intake of cereal fiber showed a 34% lower risk of coronary heart disease events when compared to women with low cereal fiber intake [6]. Cereal dietary fiber may produce these effects through multiple physiological mechanisms that include binding and eliminating cholesterol, modulation of hormonal activity, stimulation of immune system, and facilitating toxicant transit through the digestive tract. A two-step fractionation process with enzymatic hydrolysis effectively enriched arabinoxylan content [7]. Corn fiber gum (CFG) can be obtained by the extraction of corn bran using an alkaline hydrogen peroxide process [8].
This research was conducted to explore pro-cessing techniques for enhancing the bioactive and physical properties of corn bran for human health benefits and increasing its market value. The influence of jet-cooking and alkaline treatments on corn bran fractions was studied for phenolic contents, antioxidant activities, and physical properties of the fractions.
2. Materials and Methods
2.1. Source of Corn Bran
Corn bran used in this study was supplied by Bunge Milling Company (Danville, IL).
2.2. Processing Procedures
This procedure was modified from previous experience with corn bran by degrading and dispersing cellular components into soluble and insoluble portions [9-12].
2.2.1. Jet-Cooking with Alkaline Treatment (T1)
1) Three-hundred grams of corn bran and 15 g of lime (CaO, Sigma) were gradually added to 1.2 L of hot water (80˚C), and mixed using a Polytron (PT6000, Kinematica AG, Littau, Switzerland) at 8000 rpm for 5 min. The pH was adjusted to 12 with 50% sodium hydroxide, and mixed at again 8000 rpm for 5 another min.
2) The slurry was mixed with 1.5 L water, and then jet-cooked (Penford Corp., Cedar Rapids, IA) at 65 psi, 140˚C, and 1.2 L/min flow rate. The slurry was jet-cooked a second time, and pH was adjusted to 7 with acetic acid, and followed by centrifugation at 3000 rpm (1500 g) for 15 min.
3) The supernatant and insoluble fractions were drumdried at 135˚C (Model 20; Drum Dryer and Flaker Company, South Bend, IN), and then ground to pass through a 150 µm (100 mesh) sieve using a grind (Fritsch, Serial No. 14.102/2196, Idar-Oberstein, Germany).
2.2.2. Jet-Cooking without Alkaline Treatment (T2)
T2 was treated the same as T1 except no alkaline treatment made before jet-cooking.
2.2.3. Control (TC)
The unprocessed corn bran was used as control compared to jet-cooked treatments.
2.3. Proximate Composition
Moisture contents were determined according to approved method 44 - 15.02 [13]. Protein was determined by the combustion method with a protein correction factor of %N × 6.25 [14]. Oil was extracted using a Soxhlet apparatus and quantified gravimetrically [15]. Ash was determined gravimetrically after combustion at 590˚C for 12 h (Thermolyne, Sybron Corp., Milwaukee, WI, USA).
2.4. Total Phenolic Contents and Antioxidant Activity
2.4.1. Sample Extraction
One-hundred mg of corn bran or treated bran fractions was suspended in 10 mL of water, 10% ethanol, 50% ethanol, and 2% calcium hydroxide respectively, and mixed on a vortex mixer. Tubes were then placed in a boiling water bath for 5 min, and then centrifuged at 1500 g for 10 min. The supernatant was analyzed for phenolics content and antioxidant activity.
2.4.2. Total Phenolic Content Using Colorimetric Method
Total phenolic content was determined by the FolinCiocalteau colorimetric method as described pre-viously with minor modifications [16,17]. To 100 mL of extract, 7.9 mL of deionized water and 0.5 mL of Folin-Ciocalteau reagent (Sigma Aldrich, St Louis, MO) were added, mixed on a vortex mixer, and 1.5 mL of 1.85 M Na2CO3 added after 15 min. Absorbance of samples was measured at 765 nm wavelength after 2 h using gallic acid as a standard. Results were expressed as mg of gallic acid equivalents per g of sample on dry basis.
2.4.3. Antioxidant Activity Measurements
Antioxidant activities were determined as described with modifications by reacting 1 mL of the extracts with 1 mL of 200 mM 2,2-diphenyl-1-picryl-hydrazyl (DPPH) [18, 19]. Absorbance was measured at 515 nm wavelength after a 40 min reaction in dark. Tubes were centrifuged for 10 min at 3000 rpm (X 1462 g) prior to reading the absorbance at 515 nm. Results were expressed as mM of 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox) equivalents per g of sample on dry basis.
2.5. Identification of Phenolic Acid Using LC-ESI-MS Analysis
2.5.1. Sample Extraction
Samples (250 mg) were mixed with 3 mL of methanol, capped, wrapped with sealing tape, and incubated for 72 h at room temperature. Samples were then sonicated for 15 min at 25˚C and allowed to stand at room temperature for 1 - 2 h. An aliquot was removed from the vial and filtered (0.45 µm) for HPLC analysis.
2.5.2. Analytical Methodology
The method used was similar with a previous method [20]. Samples were run on a stand-alone Shimadzu 10A HPLC system (SCL-10A system controller, two LC-10A pumps, CTO-10A column oven, and SIL-10A autoinjector). Peaks were monitored using a Hewlet-Packard 1040A photodiode array detector running under the HP Chemstation software version A.02.05. The column used was an Inertsil ODS-3 reverse phase C-18 column (5 µM, 250 × 4.6 mm, with a Metaguard guard column, from Varian). For phenolic analysis, the initial conditions were 20% methanol and 80% 0.01 M phosphoric acid in water, at a flow rate of 1 ml per min. The effluent was monitored at 285 nm on the VWD. After injection (typically 15 µL), the column was held at the initial conditions for 2 min, then developed to 100% methanol in a linear gradient over 53 min. Three point standard curves based on nanomoles injected were prepared from injections of pure standards of coumaric acid, caffeic acid, sinapic acid and ferulic acid obtained from Sigma Chemical Co (St. Louis, MO).
2.5.3. LC-ESI-MS Analysis
Samples were run on a Finnigan-Thermoquest LCQ LC-MS system (AS3000 autoinjector, P4000 HPLC pump, UV6000 PDA detector, LCQ ion-trap mass spectrometer and a nitrogen generator) (San Jose, CA) all running under the Xcaliber 1.3 software system. The MS was run with the ESI probe in the negative mode. The column was a 3 mm × 150 mm Inertsil reverse phase C-18, ODS 3, 3 µ column (Metachem, Torrance, CA) with a Metaguard guard column. The source inlet temperature was set at 300˚C and the sheath gas rate was set at 80 arbitrary units. The MS was optimized for the detection of phenolic acids by using the autotune feature of the software while infusing a solution of ferulic acid in with the effluent of the column and tuning on an atomic mass unit of 193 [M-H]–. The solvent systems included A (water with 0.25% acetic acid) and B (methanol with 0.25% acetic acid). The column was equilibrated with 20% B at a flow rate of 0.3 ml per min. After injection the column was held at the initial conditions for 2 min, and then developed with a linear gradient to 100% B over 50 min. The column effluent was monitored at 285 nm in the PDA detector. The software package was set to collect mass data between 150 - 1000 AMUs. Generally the most significant sample ions generated under these conditions were [M-H]–.
2.6. Water-Holding Capacity
The method for water-holding capacity of corn bran and its fractions was modified according to a previous procedure [21]. Solids (2 g) with or without jet-cooking were mixed with 25 g of deionized water and vigorously mixed using a vortex to make a suspension, allowed to stand for 2 h, followed by centrifugation at 1,462 g for 15 min. The supernatant was decanted and weight of residue was measured. The water-holding capacity for each sample was measured in duplicate. Water-holding capacity was calculated by the following equation:
water holding capacity (%) = (sample weight after centrifugation–dry sample weight)/dry sample weight x 100
2.7. RVA Measurements
A Rapid Viscosity Analyzer RVA-4 (Foss North America, Eden Prairie, MN) was used for measuring pasting properties of unprocessed corn bran and jet-cooked corn bran with or without alkaline treatment. Sample (2.24 g, dry basis) was added to DI water in a RVA canister to make a suspension containing 8% solids. The suspensions were equilibrated at 30˚C for 1 min, heated to 95˚C at a rate of 6.0˚C/min, maintained at 95˚C for 5.5 min, and cooled to 50˚C at rate of 6.0˚C/min and held at 50˚C for 5 min. For all test measurements, a constant paddle rotating speed (160 rpm) was used throughout entire analysis except for the first 10 s to disperse sample (960 rpm). Each sample was analyzed in duplicate.
2.8. Statistical Analysis
Statistical differences were calculated using SAS software [22]. A general linear model analysis of variance followed by Tukey’s multiple comparison adjustment was used [23]. Significant differences were defined as p < 0.05.
3. Results and Discussion
3.1. Yield and Compositions
The soluble solids recovery from T1 (pH 12) was doubled (Table 1, 38.4 %) compared to soluble solids from T2 (pH 7, 19.4%), while the insoluble solids (61.6%) from T1 were approximately 20% lower than that in T2 (81.6%). T1 included alkali treatment that solubilized a substantial portion of hemicelluloses from plant cell wall materials [24]. Thus, the soluble solids in T1 were increased while insoluble solids were greatly decreased.
The protein content of soluble solids from T2 (Table 1, 6.62%) gave the highest among all the fractions. The soluble proteins were apparently a result of high temperature and shearing during jet-cooking. It suggested that high shearing and temperature using jet-cooking under neutral conditions does not degrade protein but increases protein solubility.
The oil contents in soluble (0.39%) and insoluble solids (0.17%) were decreased significantly from jet-cooking with alkaline treatment (T1) compared with the soluble (1.44%) and insoluble solids (0.62%) from T2 with

Table 1. Protein, oil, and ash contents.
out alkaline treatment, and TC starting material (1.02%, Table 1). The high pH could have resulted in the degradation of lipids in T1. The oil content of soluble solids from T2 was highest (1.44%) among all the samples, about 30% higher compare with starting corn bran (1.02%). Jet-cooking of T2 greatly modified the structure of corn bran by its high shear and high temperature, but did not degrade the oil and protein. Also, the oil content of soluble solids was significantly increased in T2 since the total weight was decreased by removing insoluble materials by centrifugation.
The ash contents of all treated fractions were higher than that of TC, unprocessed corn bran (Table 1). The ash contents (39.88%) from T1 were significantly higher than that of TC (1.09%). It was likely due to lime added in the beginning of the processing for T1, and could also provide a nutritional source of calcium.
The fatty acid composition was indicative of corn oil [25]. All samples contained a predominance of oleic acid, followed by linoleic acid (Table 2) with exception of insoluble solids from T2. The un-saturated/saturated ratio from T1 was higher than that from T2 and TC. Thus, T1 had less saturated fatty acids in the soluble solids compared to T2 and TC. The fatty acid content from T1 insoluble solids was below detention limit that may be due to the extremely low oil content (0.17%).
3.2. Total Phenolic Content
Phenolics are commonly categorized as phenolic acids, flavonoids, stilbenes, coumarins, and tannins. Phenolics are the products of secondary metabolism in plants, providing essential functions in the reproduction and growth of the plants, acting as defense mechanisms against pathogens, parasites, and predators, as well as contributing to the color of plant. In addition to their roles in plants, phenolic compounds in our diet may provide health benefits associated with reduced risk of chronic diseases [26].
3.2.1. Effect of Alkaline Treatments
The phenolic contents from T1 in soluble solids (56.30, 61.38, 54.25 and 52.93 mg/g) were highest among all samples regardless solvent (Table 3). The phenolic contents from T1 in insoluble solids (28.76, 31.14, 29.7428.12 mg/g.) were about half compared to soluble solids in T1, but they were significantly higher than soluble and insoluble solids in T2 and TC for all solvents used with exception for 0.2% Ca(OH)2 extraction of insoluble so-
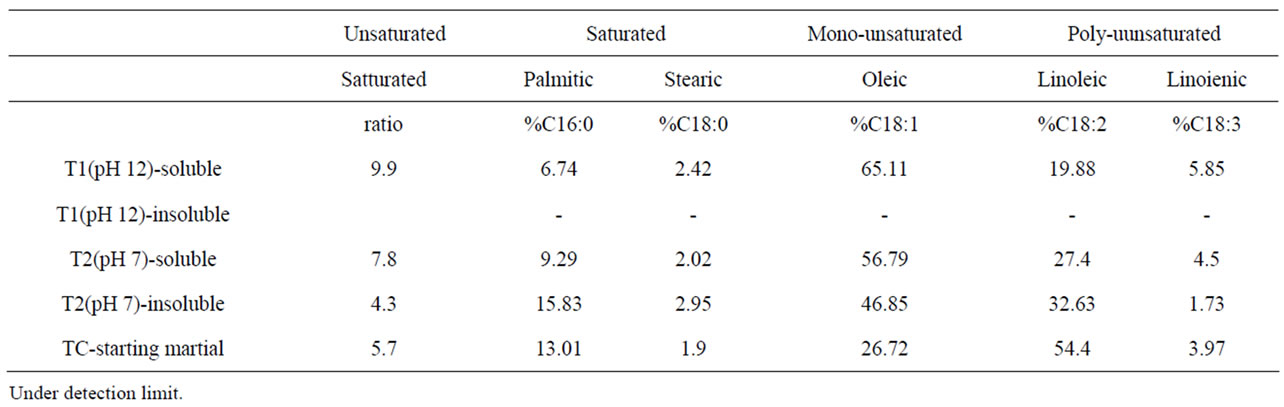
Table 2. Fatty acid compositions.

Table 3. Phenolic contents extracted by water, 10% and 50% ethanol.
lids from T2 (Table 3). The phenolic contents from T2 in soluble solids (7.18, 7.10, 6.85, 7.83 mg/g), using all solvents respectively, were less than 20% of that from soluble solids from T1 (56.30, 61.38, 54.25 and 52.93 mg/g). It indicated that alkaline conditions were effective in degrading the cell wall structure of corn bran in T1; hence, phenolic compounds, such as ferulic acid, could be released from ester bonds by alkaline conditions [27]. Overall, the phenolic contents were higher in soluble solids than insoluble solids regardless treatment with the exclusion of the extractions by 0.2% Ca (OH)2. Phenolic contents (7.18, 7.10, 6.85, and 7.83 mg/g) in soluble solids from T2 (pH 7) using different solvents, respectively, were higher than that (1.28, 2.79, 4.64, and 23.57 mg/g) from TC. Food processing, such as thermal processing, pasteurization, fermentation, and freezing, contributes to the release of bound phenolic acids [4]. In general, the phenolic contents of soluble and insoluble solids from T1 with alkaline treatment were significantly higher than those from T2 and TC. Our study indicated that high temperature and shearing by jet-cooking released some soluble phenolic compounds, but alkaline treatment and jet-cooking together was the most effective procedure to release phenolic compounds from corn bran.
3.2.2. Effect of Ethanol Concentration
Ten percent ethanol was chosen because it was generally the concentration used for preparing standards in a typical procedure [16]. The phenolic contents in insoluble solids from T2 and TC were significantly increased with increasing ethanol percentage (Table 3). It suggests that phenolic compounds from corn bran have a high solubility in ethanol. There were no significant differences found in insoluble and soluble solids from T1 by different concentrations of ethanol respectively (Table 3). It suggests that the effect of alkaline condition on phenolic solubility was more effective than ethanol concentration for extraction.
3.2.3. Effect of 0.2% Ca(OH)2 Extraction
Interestingly, dramatic increases in phenolic contents were found in solids from T2 and TC (23.57 mg/g) using 0.2% Ca(OH)2, compared to water, 10% and 50% ethanol extractions, respectively. There are bound phenolic content and free phenolic content and the contribution of bound phenolic to the total phenolic content was significantly higher than that of free and esterfied fractions [28]. The solids from T2 and TC were not involved with alkaline treatment. Therefore, bound phenolic compounds from T2 and TC were released using when 0.2% calcium hydroxide. There were no significant differences in phenolic contents for soluble and insoluble solids from T1 between water extraction and 0.2% calcium hydroxide extraction (56.30 mg/g vs. 52.93 mg/g, and 28.76 mg/g vs. 28.12 mg/g since the solids from T1 were processed under alkaline conditions previously. Probably most bound phenolic compounds in T1 were released from cell wall structures by alkaline treatment and found in free form after processing. Therefore, 0.2% calcium hydroxide extraction had no effect on results.
Our results confirmed that alkaline conditions are important for the release phenolic compounds. Also, it is possible to extract more phenolic com-pounds from insoluble solids without alkaline treat-ment and TC by using alkaline extraction such as 0.2% calcium hydroxide. Perhaps the 0.2% calcium hydroxide extraction could be an alternative and efficient choice without jet-cooking for extracting increased amount of phenolic compounds.
3.2.4. The Comparison of Gallic Acid and Ferulic Acid as Standards for Phenolic Analysis
Ferulic acid plays a significant role in the plant cell walls because it forms bonding between polysaccharides and proteins [27]. Ferulic acid is a well known antioxidant with potential for food and medical applications [29]. Gallic acid is commonly used for phenolic content test. This study was the first report using ferulic acid as standard to determine phenolic content in corn bran. The statistical significant differences were found in soluble solids of T1, insoluble solids of T2, and TC between the results using gallic acid or ferulic acid as standard (Table 4). The results from this study provided an alternative choice for using ferulic acid as standard to test phenolic content.
3.3. Identification of Phenolic Acids Using HPLC
To further indentify phenolic acids in each of the corn bran fractions, HPLC was used to detect caffeic, coumaric and ferulic acid, three of the most common phenolic acids in corn [30]. The coumaric, caffeic, ferulic and sinapic acids are mainly present in the bound form, linked to cell wall components such as cellulose, lignin, and proteins through ester bonds [26]. Their structures are similar, except on carbon 3 of the phenolic ring, ferulic acid has methyl ether, caffeic acid has a hydroxyl group, and coumaric acid has hydrogen. The extra hydroxyl group on caffeic acid makes it more water-soluble than coumaric and ferulic acids [12]. The soluble solids from T1 had the highest coumaric content (0.933 mg/g) and ferulic acid content (7.153 mg/g) among all the samples tested (Table 5). Consistent with the total phenolics data, in general, the soluble fractions after treatment had higher phenolic acid levels than the insoluble fractions in T1. The trend for coumaric and ferulic acid content using LC-ESI-MS were in agreement with the total phenolic contents measured by spectrophotometer. The results were comparable with a previous study that considerable

Table 4. Phenolic contents of 50% ethanol extracts.
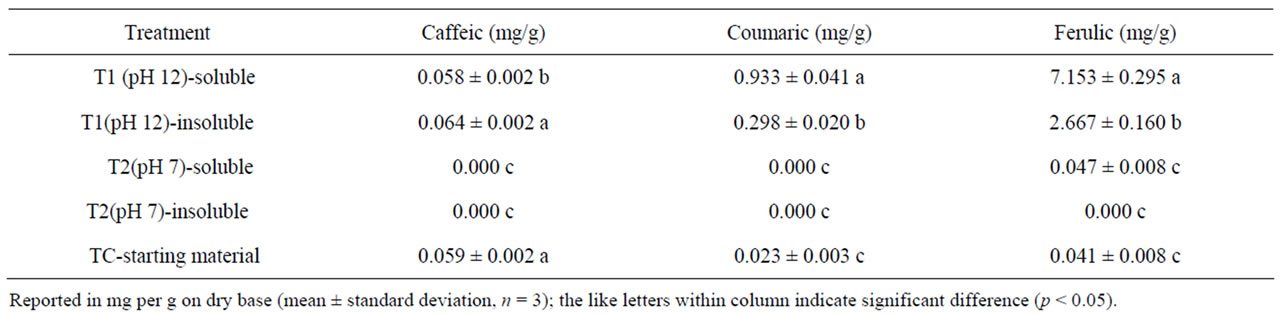
Table 5. Phenolic acid contents using LC-ESI-MS.
amounts of hydroxycinnamic acid (up to 0.015% of mainly ferulic acid) and lipid (up to 0.043%) were released with 1.5 N methanolic KOH [31]. The higher coumaric and ferulic acid levels were found in the soluble fraction from alkaline treatment while the soluble fraction without alkaline treatment contained much lower level of these phenolic acids. These phenolic acids were liberated through base-catalyzed hydrolysis during processing in T1 suggesting that T1 was more effective at liberating water-soluble phenolic acids than T2. Our results indicated that colorimetric method using spectrophotometer was suitable to determine total phenolic acid compounds while LC-ESI-MS was excellent for identifying the individual phenolic compound.
3.4. Antioxidant Activity
The antioxidant activities of soluble solids from T2 (6.49, 5.68, and 6.87 μmol/g) were the highest among all fractions regardless of extracting solvent and treatment (Table 6). It was not expected that the soluble solids from T1 would have the highest phenolic content but without increasing antioxidant activities, regardless of solvent. T1 included harsher conditions (high temperature, pressure, and high pH) than T2 which may have destroyed some antioxidant activity of the phenolics. Perhaps those phenolics having a greater number of hydroxyl-type substituents may be more easily degraded under the alkaline extraction conditions whereas some phenolics without antioxidant activity appear to be more stable than those with antioxidant activity [32].
Significant differences in antioxidant activities were observed between water and 50% ethanolic extracts. Over all, antioxidant activities were increased with increasing ethanol percentages. Fifty % ethanol extracts showed significantly higher anti-oxidant activity in all cases. Trolox has lower water solubility (0.5 mg/1 mL) than that of gallic acid (11.5 mg /1 mL, Merck Index), indicating that the phenolic compounds exhibiting antioxidant activities had high solubility in ethanol. T2 gave enhanced antioxidant activities in the soluble fraction compared to TC (Table 6).
3.5. Water Holding
Jet-cooking increased water holding capacities from 254% (TC) to about 300 % for jet-cooked solids without pH adjustment (T2). As the pH values were adjusted to 12 prior to jet-cooking (T1), the water holding capacity of insoluble solids was considerably increased to 524 % (Figure 1). The results of water holding capacities were consistent with the data obtained from peak viscosity using Rapid viscosity analysis (Figure 2). The results were consistent with the previous research that thermomechanical shear during steam jet-cooking led to significant increases in the water absorption, water solubility, and swelling power of the barley flour [33] and earlier research on corn bran [9,10].
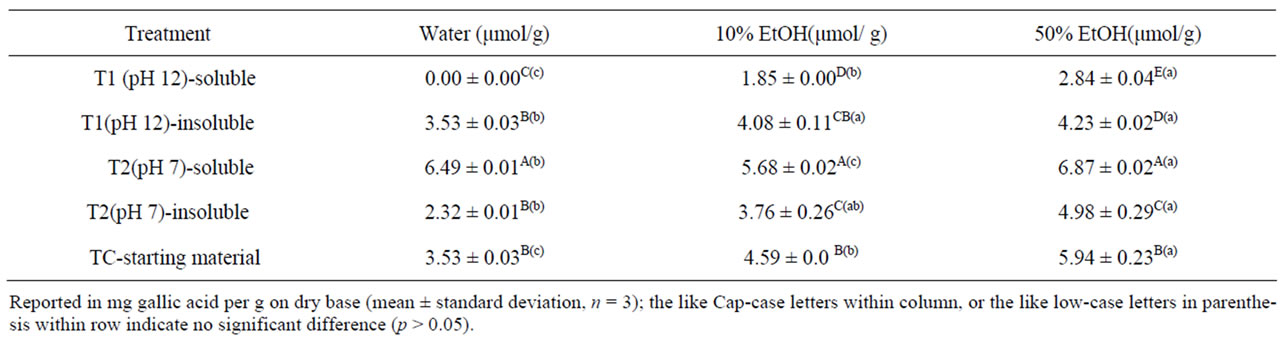
Table 6. Antioxidant activities of water, 10% and 50% ethanol extracts.
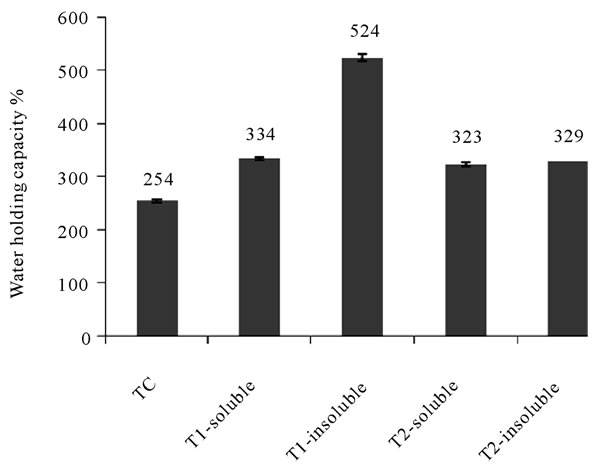
Figure 1. Water holding capacities of jet-cooked corn bran at different pH values compared with unprocessed corn bran.
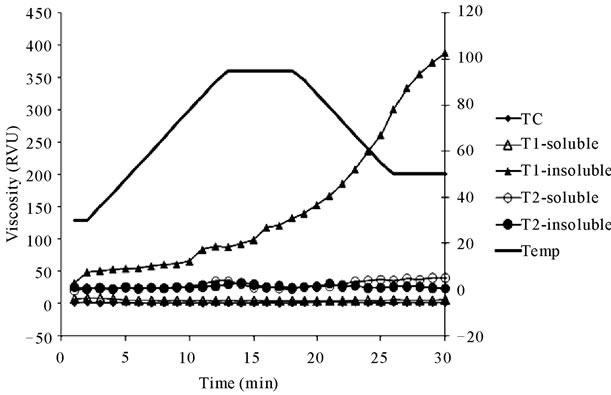
Figure 2. RVA pasting profiles of jet-cooked barley flours at different pH values compared with unprocessed corn bran.
3.6. Rapid Viscosity Analysis
The insoluble solids from T1 demonstrated higher viscosity compared with processed samples from T2 and TC (Figure 2). Apparently, the high pH degraded cell wall structures in T1, creating more small molecules that resulted in increasing thickness and viscosity. The pasting curve from minimum viscosity after peak to final viscosity, referred to as the setback region, was significantly increased for the insoluble solids with alkaline treatment. A large setback value is generally associated with syneresis, or weeping, during freeze/thaw cycles [34]. This special property along with high antioxidant activities could be an important factor in making functional corn bran products.
4. Conclusions
This study demonstrated that jet-cooked corn bran under alkaline conditions (T1) produced more soluble solids with increased total phenolic content and high water holding capacities along with interesting viscoelastic properties but without increasing antioxidant activities. Jet-cooking corn bran without alkali (T2) increased the soluble solids that contained higher phenolic content with increased antioxidant activity and water holding capacity compared with control (TC). This study suggested that corn bran is an important source of phytochemicals including phenolics having antioxidant activities. The jetcooking technology may increase the value of corn bran by creating new photochemical and functional products.
REFERENCES
- R. A. Moreau, A. Nuñez and V. Singh, “Diferuloylputrescine and P-Coumaroyl-Feruloylputrescine, Abundant Polyamine Conjugates in Lipid Extracts of Maize Kernels,” Lipids, Vol. 36, No. 8, 2001, pp. 839-844. doi:10.1007/s11745-001-0793-6
- M. J. Kim, S. M. Kim, K. R. Im and K.-S. Yoon, “Effect of Hydroxycinnamic Acid Derivatives from Corn Bran on Melanogenic Protein Expression,” Journal of the Korean Society for Applied Biological Chemistry, Vol. 53, No. 4, 2010, pp. 422-426.
- A. Y. A. Plate and D. D. Gallaher, “The Potential Health Benefits of Corn Components and Products,” Cereal Foods World, Vol. 50, No. 6, 2005, pp. 305-314.
- V. Dawanto, X. Z. Wu and R. H. Liu, “Processed Sweet Corn has Higher Antioxidant Activity,” Journal of Agricultural and Food Chemistry, Vol. 50, 2002, pp. 4959-4964. doi:10.1021/jf0255937
- Z. Liu, J. Wang, P. Shen, C. Wang and Y. Shen, “Microwave-Assisted Extraction and High-Speed Counter-Current Chromatography Purification of Ferulic Acid from Radix Angelicae Sinensis,” Separation and Purification Technology, Vol. 52, No. 1, 2006, pp.18-21. doi:10.1016/j.seppur.2006.03.009
- A. Wolk, J. E. Manson, M. J. Stampfer, G. A. Colditz, F. B. Hu, F. E. Speizer, C. H. Hennekens, and W. C. Willett, “Long-Term Intake of Dietary Fiber and Decreased Risk of Coronary Heart Disease among Women,” Journal of the American Medical Association, Vol. 281, No. 21, 1999, pp. 1998-2004. doi:10.1001/jama.281.21.1998
- B. Wang, B. Cheng and F. Hao, “Enrich Arabinnoxylan in corn Fiber for Value-Added Products,” Biotechnology Letters, Vol. 30. No. 2, 2007, pp. 275-279. doi:10.1007/s10529-007-9537-9
- M. P. Yadav, D. B. Johnston, Jr., A. T. Hotchkiss and K. B. Hicks, “Corn Fiber Gum: A Potential Gum Arabic Replacer for Beverage Flavor Emulsification,” Food Hydrocolloids, Vol. 21, No. 7, 2007, pp. 1022-1030. doi:10.1016/j.foodhyd. 2006.07.009
- G. E. Inglett, “Development of a Dietary Fiber Gel for Calorie-Reduced Foods,” Cereal Foods World, Vol. 42, No. 5, 1997, pp. 382-385.
- G. E. Inglett, “Soluble Hydrocolloid Food Additives and Method of Making,” US Patent No. 6,060,519. 1998.
- D. N. Kim, I. Y. Bae, G. E. Inglett and S. Lee, “Effects of Hydrothermal Treatment on the Physicochemical, Rheological and Oil-Resistant Properties of Rice Flour,” Journal of Texture Studies, Vol. 40, No. 2, 2009, pp. 192-207. doi:10.1111/j.1745-4603.2009.00176.x
- G. E. Inglett, D. Chen, D. J. Rose and M. Berhow, “High-Shear, Jet-Cooking, and Alkali Treatment of Corn Distillers Dried Grains to Obtain Products with Enhanced Protein, Oil and Phenolic Antioxidants,” Food Science and Technology International, Vol. 16, No. 4, 2010, pp. 297-304. doi:10.1177/1082013209353821
- AACC International, “Approved Methods of Analysis,” 11th Edition, AACC International, St. Paul, 2010.
- American Association of Cereal Chemists, “Crude Protein–Combustion Method,” In: “American Association of Cereal Chemists Official Methods,” 9th Edition, St. Paul, 1995, pp. 46-30.
- American Oil Chemists’ Society, “Official Methods and Recommended Practices of the American Oil Chemists Society,” 4th Edition, Champaign, 1993, pp. 3-44.
- A. L. Waterhouse, “Determination of Total Phenolics,” In R. E. Wrolstad, Ed., Current Protocols in Food Analytical Chemistry, John Wiley & Sons, Inc., New York, 2001.
- L. Yu and K. Zhou, “Antioxidant Properties of Bran Extracts from ‘Platte’ Wheat Grown at Different Locations,” Food Chemistry, Vol. 90, No. 1-2, 2005, pp. 311-316. doi:10.1016/j.foodchem.2004.04.007
- W. Brand-Williams, M. E. Cuvelier and C. Berset, “Use of a Free Radical Method to Evaluate Antioxidant Activity,” LWT - Food Science and Technology, Vol. 28, No. 1, 1995, pp. 25-30. doi:10.1016/S0023-6438(95)80008-5
- I. Şensoy, R. T. Rosen, C.-T. Ho and M. V. Karwe, “Effect of Processing on Buckwheat Phenolics and Antioxidant Activity,” Food Chemistry, Vol. 99, No. 2, 2006, pp. 388-393. doi:10.1016/j.foodchem.2005.08.007
- M. A. Berhow, “ Modern Analytical Techniques for Flavonoid Determination,” In: B. S. Buslig and J. A. Manthey, Eds., Flavonoids in the Living Cell, Kluwer Academic/Plenum Publishers, New York, 2002, pp. 61-76.
- B. I. O. Ade-Omowaye, K. A. Taiwo, N. M. Eshtiaghi, A. Angersbach and D. Knorr, “Comparative Evaluation of the Effects of Pulsed Electric Field and Freezing on Cell Membrane Permeabilisation and Mass Transfer during Dehydration of Red Bell Peppers,” Innovative Food Science and Emerging Technologies, Vol. 4, No. 2, 2003, pp. 177-188. doi:10.1016/S1466-8564(03)00020-1
- SAS Institute Inc., “The SASÒ System for WindowsÒ,” Version 8e, Cary, 1999.
- J. W. Tukey, “Where should multiple com-parisons go next?” In: F. M. Hoppe, Ed., Multiple Comparisons, Selection, and Applications in Biometry, New York, 1993, pp. 187-208.
- L. W. Doner and K. B. Hicks, “Isolation of Hemicellulose from Corn Fiber by Alkaline Hydrogen Peroxide Extraction,” Cereal Chemistry, Vol. 74, No. 2 1997, pp. 176-181. doi:10.1094/CCHEM.1997.74.2.176
- P. J. White and E. J. Weber, “Lipids of the kernel,” In: P. J. White and L. A. Johnson, Eds, Corn: Chemistry and Technology, American Association of Cereal Chemists, St. Paul, 2003, pp. 355-406.
- R. H. Liu, “Whole Grain Phytochemicals and Health,” Journal of Cereal Science, Vol. 46, No. 3, 2007, pp. 207-219. doi:10.1016/j.jcs.2007.06.010
- P. Jankovska, J. Copikova, A. Sinitsya and M. Novotna, “The Effect of Ferulic Acid on the Structure of Plant Cell Wall and Its Determination,” Czech-Journal-of-FoodScience, Vol. 18, 2000, pp. 182-183.
- C. M. Liyana-Pathirana and F. Shahidi, “Importance of Insoluble-Bound Phenolics to Antioxidant Properties of Wheat,” Journal of Agricultural Food Chemistry, Vol. 54, No. 4, 2006, pp. 1256-1264. doi:10.1021/jf052556h
- H. D. Shin, S. McClendon, T. Le, F. Taylor and R. R. Chen, “A Complete Enzymatic Recovery of Ferulic Acid from Corn Residues with Extra-Cellular Enzymes from Neosartorya Spinosa NRRL185,” Biotechnology and Bioengineering, Vol. 95, No. 6, 2006, pp. 1108-1115. doi:10.1002/bit.21056
- P. Mattila, J.-M. Pihlava and J. Hellström, “Contents of Phenolic Acids, Alkyland Alkenylresorcinols, and Avenanthramides in Commercial Grain Products,” Journal of Agricultural and Food Chemistry, Vol. 53, No. 21, 2005, pp. 8290-8295. doi:10.1021/jf051437z
- M. P. Yadav, R. A. Moreau and K. B. Hick, “Phenolic Acids, and Proteins Associated with Purified Corn Fiber Arabinoxylans,” Journal of Agricultural and Food Chemistry, Vol. 55, No. 3, 2007, pp. 943-947. doi:10.1021/jf0624493
- A. Liazid, M. Palma, J. Brigui and C. G. Barroso, “Investigation on Phenolic Compounds Stability during Microwave-Assisted Extraction,” Journal of Chromatography A, Vol. 1140, 2007, No. 1-2, pp. 29-34. doi:10.1016/j.chroma.2006.11.040
- S. Y. Lee and G. E. Inglett, “Functional Characterization of Steam Jet-Cooked β-Glucan-Rich Barley Flour as an Oil Barrier in Frying Batters,” Journal of Food Science, Vol. 71, No. 6, 2006, pp. E308-E313. doi:10.1111/j.1750-3841.2006.00121.x
- Newport Scientific Pty. Ltd., “The Application Manual for the Rapid Visco Analyser,” NSW 2102, Warrriewood, 1998.

